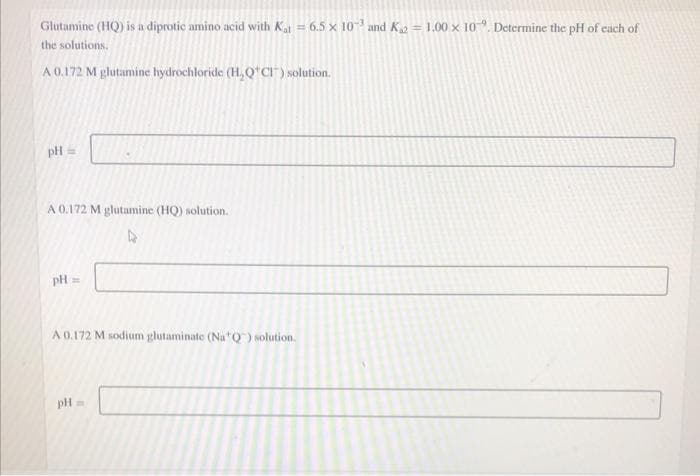
Glutamine (HQ) is a diprotic amino acid with Ka = 6.5 × 10 and K2 = 1.00 × 10". Determine the pH of cach of the solutions, A0.172 M glutamine hydrochloride (H,Q'cr) solution. pH =
Q: b. What type of reactions are these called? 2. Which reaction number above involves only one reac...
A:
Q: Nitric oxide, NO, is believed to react with chlorine according to the following mechanism: NO + Cl2...
A: Which of the following is/are and intermediate in the reaction?
Q: KBr + KCIO, + H,SO, Br, + KCI + K,SO, + H,C
A: Answer - Oxidation number method - The oxidation number method, also called oxidation states, keeps ...
Q: The pH of an aqueous solution of 0.429 M formic acid, HCOOH is ________. Calculate the pH of a 0.014...
A: 1) Given - Concentration of HCOOH = 0.429M Ka of HCOOH - 1.8×10-4 To calculate = PH of formic acid ...
Q: Nitrogen and hydrogen react according to the reaction below to form ammonia: N2 (g) + 3 H2 (g) ⇌ 2 N...
A: the equilibrium concentration of NH3 can be calculated as
Q: . Sodium carbonate is combined with calcium nitrate. a. Write the molecular, ionic equation, and net...
A:
Q: Answer the following questions about each of the hydroxy ketones: 1-hydroxybutan-2-one (A) and 4-hyd...
A: Complete solution is given below in next step in jpeg format
Q: a) CH3MGBR + CdBr2 b) CSH5NA + BeBr2 c) PHCH2OCH3 + Li
A: ANSWER : The reaction are known as Metal Exchange Reaction in which one metal get exchanged with oth...
Q: c) Br2 hv d) HO Br
A:
Q: AG =
A:
Q: You have 100 mL of solvent available to do an extraction. Which will give a better separation? O A. ...
A:
Q: Calculate the velocity of a neutron whose de Broglie wavelength is 500 pm. Search for the mass of a ...
A: given :- de Broglie wavelength = 500 pm to be calculated :- velocity of neutron
Q: The gas-phase reaction of NO with F2 to form NOF and F has an activation energy of E = 6.30 kJ/mol a...
A:
Q: Look at the following Lewis Dot Structure: 2- :S:
A:
Q: The hydroxide ion concentration, [OH-], of an aqueous solution of 0.429 M methylamine (a weak base w...
A:
Q: Direction: Identify each compound as Empirical or Molecular. Write your answer on the space provided...
A: A formula which represents the simplest whole number ratio of elements present in the compound is c...
Q: 1. Provide the major product(s) for the following reactions. Be sure to indicate stereochemistry whe...
A:
Q: The following data were acquired in an iodination experiment involving acetone. Reaction times are f...
A: The iodination of acetone in aqueous solution is catalyzed by H+ ions: I2 + CH3COCH3 → H+ HI + CH3C...
Q: . At 30.0 °C, pure benzene has a vapor pressure of 121.8 mmHg. Dissolving 15.0 g of a non- volatile ...
A:
Q: Match Column A with Column B. V Distance traveled by the solvent A. Silica gel G Adsorbent used in T...
A: 1) The solvent front is defined as the distance that is traveled by the solvent on the paper' of pa...
Q: Answer the following comprehensively based on what you have learned on the properties of liquids and...
A: 1. a) Answer - According to the question - Surface tension - Surface tension is the tendency of liqu...
Q: Draw the structure of the following compounds. 1. 5-chloro-4-cyanopentanoic acid 2. 3-(cyclobutylmet...
A: Rules to draw a structure from IUPAC name • Locate the root word and write the principal chain. The...
Q: ... Question 13 of 39 Submit What is the correct IUPAC name for the compound shown here? Br | 3-)5-)...
A: ANS. 1-bromo-2,3-dimethylcyclopentane
Q: The hydroxide ion concentration in an aqueous solution at 25°C is 3.3x102 M. The hydronium ion conce...
A: Solution Given that hydroxide ion concentration = 3.3x10-2M Fi...
Q: In the presence of excess thiocyanate ion, SCN-, the following reaction is first order in iron(III)...
A:
Q: Question 25 of 39 Submit What is the correct systematic name for the compound shown here? CH2 /CH3 t...
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: Identify the reackion type for each of the Pollowing a) 8 H2s t 8cl2> Sg t Ib HCl b) MnSOy tH2s > Mn...
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost...
Q: Please don't copy consider the two diagrams showing the energies (boxes) of each of four A particles...
A: each possible outcome is called a "microstate". The combination of all microstates that give the sam...
Q: Categorize each alcohol below as primary, secondary or tertiary OH OH OH
A: Identify which type of alcohol is present--
Q: Phosphoric acid is often added to colas to give them the tart flavor people like. Coke Classic® has ...
A: H3PO4 dissociates as: H3PO4↔H++H2PO4- Ka1=6.9×10-3H2PO4-↔H++HPO42- ...
Q: What is the correct expected product from the following reaction? HO CH3 Br CH3 CH3 CH3 CH3 CH3 ọ-CH...
A: Unimolecular nucleophilic substitution reaction (SN1): This reaction proceeds into two steps. The fi...
Q: H NMR SPECTRUM H NMR Spectrum 200 Me. CDO, solutioni expansions hanges TMS ppm 20 10 9 8 (ppm) 2.2 p...
A: The analysis of 1H NMR spectrum of the above compound is given below
Q: A first order reaction is found to have a half- life of 1.2 seconds. This reaction is initiated with...
A: The rate constant (k) for a first-order reaction can be calculated from the half-life time t12. It c...
Q: NH a) Br 6)
A: In the option a,the two compounds are not isomer because isomers have same molecular formula but dif...
Q: A cylindrical glass tube of length 25.75 cm and radius 2 cm is filled with argon gas. The Empty tube...
A:
Q: QUESTION 1 If 47.88 mL of distilled water at 90.2 °C is added to 47.93 mL of distilled water, in a c...
A: We have to predict the heat capacity of calorimeter.
Q: Which of the reaction mechanisms is consistent with the energy diagram? fast O 2 A → B slow В C slow...
A: The energy diagram is given that provide graph of reaction mechanism. we have to predict the consis...
Q: 3) Is the following molecule a D or Lamino acid? 4) Draw the structure of Ala-Val-lle.
A:
Q: A reactant decomposes with a half-life of 149 s when its initial concentration is 0.312 M. When the ...
A:
Q: based on the following equation determine how many g of magnesium phosphate will be produced if you ...
A:
Q: What is the correct systematic name for the compound shown here? CH3 CH3
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: Calculate ∆U, ∆H, q, and w for compressing 2 mol of an ideal gas from 1 bar to 10 bars at 25°C given...
A: An isothermal process is a change of a thermodynamic system, in which the temperature remains consta...
Q: Ammonium phosphate ((NH) PO) is an important ingredient in many solid fertilizers. It can be made by...
A: The reaction of phosphoric acid with liquid ammonia is, H3PO4 + 3NH3 → (NH4)33PO4
Q: Calculate the pH of a 0.0470 M aqueous solution of the weak base methylamine (CH3NH2, Kb = 4.20×10-4...
A:
Q: A. Dilution of Stock Solution Concentration of STOCK solution you prepared (M) 0.8 Volume of STOCK s...
A: Introduction: This can be calculated from the equivalence formula, V1S1 = V2S2 (V and S are the volu...
Q: Data of NaOH Trial 1: mass=0.537g Volume =23.6ml Trial 2: mass=0.566g Volume = 26.8ml Trial 3: mass=...
A:
Q: What is the percent of a sucrose solution if 18.2 grams of sucrose is dissolved in 500 mL of water?
A: Given, Mass of sucrose = 18.2 g Volume in mL = 500 mL What is the percent of a sucrose solution = ...
Q: At 25 °C, the equilibrium partial pressures for the reaction A(g) + 2 B(g) = C(g)+D(g) were found to...
A: Given, A(g) + 2B(g) ⇌ C(g) + D(g) Equilibrium partial pressure for all the above species: PA = 4.22 ...
Q: Q2/B/A Compound (A) mass of (109.23) composed of 78.46% C, 8.46%H and 13%N mixed with 40.77gm O. fin...
A: Step 1 : Determination of mass Mass of mixture = 109.23 gm Carbon mass = 78.46 % of 109.23 ...
Q: Construct a (011) within a unit cell.
A: A lattice plane in a unit cell is those plane whose intersections with the lattice are periodic.
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

- A solution is made by dissolving 0.111 moles of N2H4 (Kb = 3.0 x 10-6 ) and 0.199 moles of N2H5Cl in water and diluting to 1.00 L. What is the pH of the resulting solution?What is the pH of the solution after mixing 0.188 g of Mg(OH)2 (MW=58.321 g/mol) with 18.1 mL of 0.0173 M HCl? The resulting solution was diluted to 100 mL. Round your calculated value for pH to two figures to the right of the decimal point.Calculate the pH, pOH, and fraction of solute protonated or deprotonated in the following aqueous solutions: (a) 0.150 M CH3CH(OH)COOH(aq) (lactic acid), (b) 2.4 x 10-4 M CH3CH(OH)COOH(aq), (c) 0.25 M C6H5SO3H(aq) (benzenesulfonic acid). The appropriate values for pKa or pKb are given inthe table.
- Calculate the pH of a 0.100 M of NaCH3COO solution. Ka of CH3COOH = 1.8 x 10-5 Ka = Kw/KbA solution of volume 0.500 L contains 1.68 g NH3 (Kb = 1.8 x 10^-5) and 4.05 g (NH4)2SO4. If 0.88 g NaOH is added to the solution, what will be the resulting ph?Calculate the mass (in g) of solid sodium hydroxide (molar mass = 39.997 g/mol) required to prepare: 5 litres of a solution with pH 12.4. 85 mL of a 2.60 M solution. 250 mL of 50 % (w/w) solution. The density of 50 % (w/w) NaOH is 1.515 g/mL.
- The hardness of water (hardness count) is usually expressed in parts per million (by mass) of CaCO3. What is the molar concentration of Ca2+ ions in a water sample with a hardness count of 175mg CaCO3il? How many milliliters of concentrated sulfuric acid, 94.0% (w/w), specific gravity of 1.831 are required to prepare 1 liter of a 0.100 M solution? The solubility-product constant for Ce(IO3)3 is 3.2x1010. What is the Ce3+ concentration in a solution prepared by mixing 50.0 ml of 0.0250 M Ce3.. with 50.00 ml of water?H2. Titrations are performed by a scientist in the laboratory as follows: a solution of deuterated hydrochloric acid contains 1.08 g DCl in 750 ml of deionised, distilled water. An average amount of 14.95 ml of the DCl(aq) solution is titrated with 25.0 ml of Ca(OH)2 to produce an endpoint (as determined by an appropriate indicator). What is the molarity of the Ca(OH)2 solution? You must include all of your calculations Please give typed answerLemon juice contains citric acid which is tribasic(H3Y). 25cm^3 of lemon juice was diluted to 250cm^3. 25cm^3 of the solution was neutralized by 15cm^3 of 0.1M sodium hydroxide. a) Calculate; i) concentration of the diluted solution of lemon juice ii) concentration of the original lemon juice. iii) percentage by mass of citric acid in lemon juice.
- The pH for 0.050 M CH3NH2 (Kb for CH3NH2 = 4.0 X 10^-4 )?A solution containing only 4.0 μM of Ca(OH)2 and the compound Hydrocyanic acid. This compound in solution is partially dissociated in the solution and in equilibrium with a specific concentration 68.50 ppmv in the headspace of the container. The total pressure of the headspace is 1.5 atm and the temperature is 25 °C. What is the resulting pH? Make sure to show your work.The dissociation occurs in following manner:Ca(OH)2 ↔ Ca2+ + 2 OH− (complete dissociation)HCN ↔ CN− + H+(complete dissociation)You are given a 27 % (w/v) solution of psicose. Molar mass: psicose (C6H12O6) = 180.156 g/mol. You dissolve 6.31 g of potassium formate in 250 mL of 0.25 M formic acid (HCOOH). This solution was then made up to a total volume of 500 mL with distilled water. Potassium formate (KHCO2): molar mass = 84.12 g/mol Calculate the pH of this solution given the pKa of formic acid = 3.77.

