Q: When two hydrogen atoms approach each other to form a chemical bond, different interactions occur…
A: Part A) When two hydrogen atoms approach each other to form a chemical bond, different interactions…
Q: the chemical equation. Then write total ionic and net ionic equations for each reaction. For each…
A: In the multi sub-part questions, we solve only first three subparts according to the Bartleby…
Q: Electrophilic aromatic substitution requires the in-situ formation of a given electrophile. Which…
A: Electron defecient species are known as electrophiles. Example : Cl+ , Br+ and NO2+
Q: what is the oxidation of 2-propanol
A: We have to write the oxidation of 2-propanol.
Q: O Macmillan Learning Above what Fe²+ concentration will Fe(OH), precipitate from a buffer solution…
A: pH of buffer solution = 8.67The Ksp of FeOH2 = 4.87 × 10-17
Q: What is the bond order of B₂ and B₂? Which of these species has a longer bond? Species 1 Species 2…
A: We have to find the bond order of B2 and B2+ and, also which has the longer bond.
Q: Predict the product of the oxidation of the given aldehyde. Include all hydrogen atoms.…
A: Oxidation is addition of oxygen or removal of hydrogen
Q: A solution contains 1.33x10-2 M lead nitrate and 1.17x10-2 M nickel(II) acetate. Solid sodium…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Consider the relationship between the energy of electromagnetic radiation and its What will happen…
A:
Q: monochlorination reaction of alkane with the chemical formula C5H12 have 6 isomers, With the…
A: A and B are primary chlorines, and E and F are chiral compounds, with F being created faster than…
Q: Consider the rate law, (Look at the picture) How does the rate change when the concentration of [H+]…
A:
Q: What is the full equation (not the net ionic equation) for the chemical reaction of 0.001 M Fe(NO3)3…
A: 0.001 M Fe(NO3)3 plus 0.0002 M NaSCN diluted up to 10 mL with 0.1M HNO3
Q: a) What is the simplest formula of a compound if a sample of the compound contains 0.273 mol X,…
A: a) Given, moles of X = 0.273 mol moles of Y = 0.819 mol moles of Z = 0.546 mol Simplest formula of a…
Q: One liter of buffer is made by dissolving 103.2 grams of acetic acid, HC₂H3O2, and 103.2 grams of…
A:
Q: At 15 °C, the value of Kw is 4.5 x 10-15. What is the equilibrium concentration of OH- at this…
A:
Q: I can't see the answer/rest of the answer to the first question, can you please rewrite it?
A: Recall the given equation, Fe2O3 + 3 CO → 2 Fe + 3 CO220.0 grams of…
Q: A balloon expands from 0.0100 L to 0.500 L against an external pressure of 10.00 atm. How much work…
A:
Q: Describe how you would prepare 500 mL of a 1.0 M aqueous HCl solution from concentrated HCl…
A: Accordiyto dilution law C1V1 = C2V2 C1 and C2 are the concentrations of stock and diluted…
Q: Select the best description of the molecule shown below. III.. NH₂ Amide Acetal Peptide Carboxylic…
A:
Q: Given the following, what is the complete ionic equation? Na2SO4(aq) + Ba(OH)2(aq) → BaSO4(s) + 2…
A:
Q: 1. Consider the following exothermic reaction CO2(g) + H2O(l) = H+(aq) + HCO3-(aq) Assuming the…
A: Consider the following exothermic reaction CO2(g) + H2O(l) = H+(aq) + HCO3-(aq) Assuming the…
Q: The reaction is 2 SmCl3(aq) + 6 LiCl(aq) + 6 e- → 2 Sm(s) + 6 Li+(aq) + 6 Cl-(aq) Calculate the…
A: As per instruction given in our Q&A guidelines I can answer first question only. This question…
Q: What mass of natural gas (CH4) must you burn to emit 263 kJ of heat? Express the mass in grams to…
A:
Q: How many unique carbons in cnmr
A: To check the equivalent carbon atoms; we must check the environment of that carbon atom. The carbon…
Q: A CH3COCH3 is a base Q Search L B neither an acid nor a base C DELL an acid PARTEA
A: Acids- donate H+ ions Base - donate OH- ions
Q: Label each of the following as polar (P) or non-polar (NP)-just write the letters P or NP below each…
A:
Q: The structure below is a(n) H CH₂OH H OH H OH H OH OH Oa-D-Glucose OB-D-Fructose O a-L-Lactose…
A:
Q: Find the equilibrium constant for the reaction below. At eq, Ecell are both equal 2 Fe³+ +31 = 2…
A: Redox reaction 2Fe3+ + 3I-⇌2Fe2+ + I3- Oxidation half cell reaction (at anode) 3I- →I3- +2e- and…
Q: When 498. mg of a certain molecular compound X are dissolved in 25.0 g of dibenzyl ether…
A: A numerical problem based on properties of liquids. A set of data for depression of freezing point…
Q: You take 0.5 mL of Carb 1 and add it to 2.0 mL of buffer. You then add 7.5 mL Biuret to this sample.…
A: Here 0.5 mL of Carb 1 and add it to 2.0 mL of buffer. then added 7.5 mL Biuret to this sample.
Q: Hydrochloric acid reacts with carbonate ion to form bicarbonate ion and chloride ion. Write and…
A: In balanced chemical equation atoms of each species are equal on both reactant and products side .
Q: If you have the following data about a container of rice, about how many grains of rice are…
A: The mass of rice + container = 786 g. The mass of the container = 332 g. The mass of 1000 Grains of…
Q: Write the rate law for the two elementary reaction equations.…
A:
Q: The reaction shown involves an acid chloride, draw the organic product and select the inorganic…
A: A reaction is shown below We have to draw the major organic product. We also have to give the…
Q: Write the chemical equation for the reaction of N,N-diethyl-1-propanamine with water. Which is the…
A: We have to write the chemical equation for the reaction of N,N-diethyl-1-propanamine with water.
Q: Identify the substrate in the following reaction. Br O DMSO O Br O 2-bromoheptane ONE C- O Nat NaCN…
A:
Q: Consider each elementary step present in the mechanism of an acid-catalyzed hydrolysis of a nitrile.…
A: During reaction mechanism flow of electrons occur from higher electron density to lower.
Q: Derive a pseudo/effective Henry’s constant formula for NH3 dissolved in water. Start from its…
A: The equilibrium equation for this reaction is: NH3 + H2O ⇌ NH3•H2O ⇌ NH4+ + OH-
Q: g+ + Sn2+ oxide reduction equilibria
A:
Q: The amount of product collected after performing a lab experiment is the A. actual yield B.…
A: In question it was asked that what we call product collected after performing lab experiment. On the…
Q: Complete a reaction table (in millimoles) for the net reaction resulting when 41.4 mL of 0.0830 M…
A: A question based on mole concept. A set of data for a balanced equation is given based on which the…
Q: Aqueous iron(II) sulfate (FeSO4) and solid copper are produced by the reaction of solid iron and…
A:
Q: Rank the samples of gas described table below der of increasing average speed of the atoms or…
A:
Q: Calculate the mass of each product formed when 80.18 g of diborane (B₂H,) reacts with excess water:…
A: A question based on mole concept. An unbalanced equation is given using which the mass of products…
Q: onsider the following data on some weak acids and weak bases: name nitrous acid hypochlorous acid…
A: The question is based on the concept of salt hydrolysis. We need to arrange the given salts in…
Q: How many ATOMS of phosphorus are present in 3.91 moles of phosphorus trichloride? ________ atoms of…
A: A mole is the amount (10) of material containing 6.02214 × 1023 particles. 1 mol = 6.02214 × 1023…
Q: 9. Calculate the saturation magnetization for given 2+ cell edge length is 0.839 mm. that each cubic…
A: We will calculate the saturation of magnetisation.
Q: Solid potassium sulfate is slowly added to 150 mL of a 0.455 M barium iodide solution until the…
A: Multiple questions
Q: The ionic compound, CsI, forms a body centered cubic structure (see below) where the anions occupy…
A:
Q: CGHIIND 59811 180 C5H6N2 ||| 150 IL 160 170 C8H803 67032 140 62346 140 130 120 110 100 90 160 150…
A: Step 1: Introduction about DEPT DEPT Distortionless Enhancement in Polarization Transfer DEPT is…
Can you help me part H? Which is the formula of this rule of reaction?
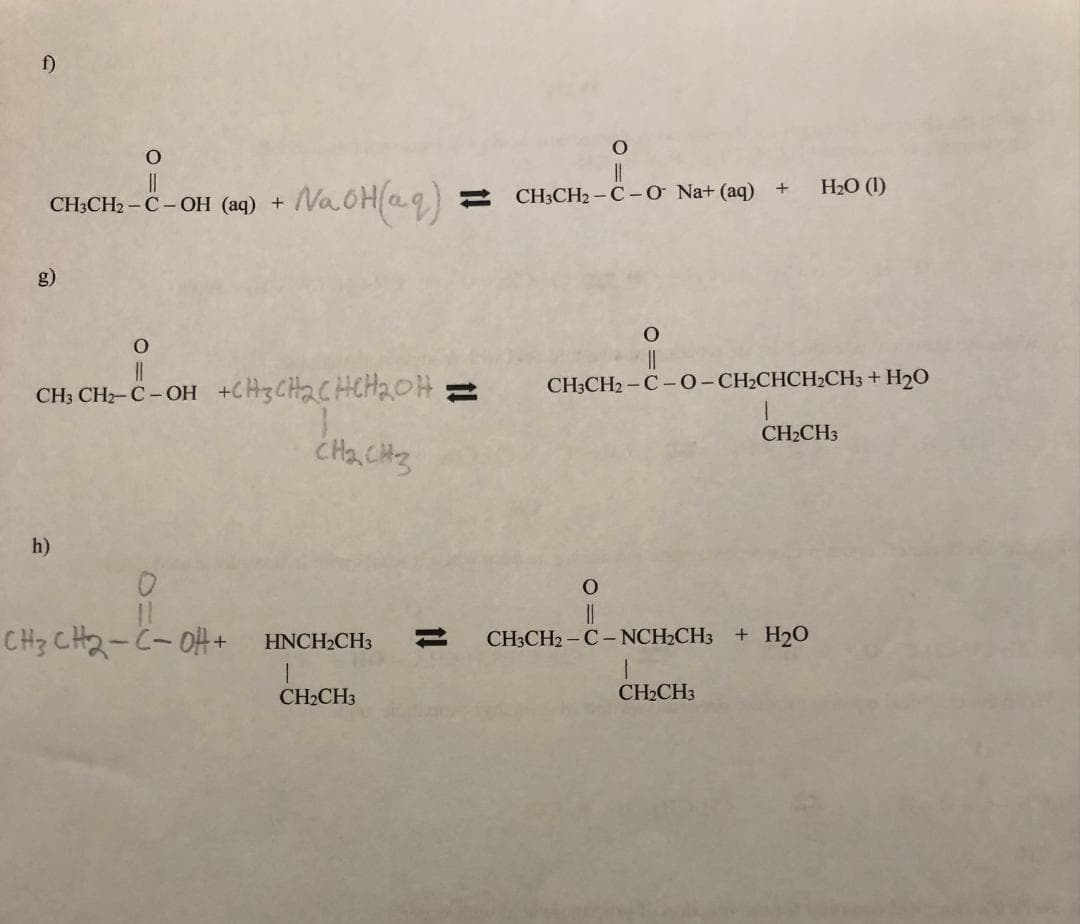
Step by step
Solved in 3 steps with 2 images

- Calculate the ΔG°rxn using the following information. 2 H2S(g) + 3 O2(g) → 2 SO2(g) + 2 H2O(g) ΔG°rxn = ? ΔG°f (kJ/mol) -33.4 -300.1 -228.6 Group of answer choices +990.6 kJ -1124 kJ +562.1 kJ +72.0 kJ -990.6 kJOrgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???Find the deltaH for the reaction below, given the following reactions and subsequent deltaH values: N2H4(l) + H2(g) —> 2NH3 (g) N2H4(l) + CH4O(l) —> CH2O (g) + N2(g) + 3H2(g)—— deltaH= -32.8kj N2(g)+ 3H2(g) —> 2NH3 (g)—deltaH=-57.2 kj CH4O(l) —>CH2O(g) + H2(g) —-deltaH= -65 kj SIG FIGS
- Compound ΔHf (kj/mol) C2H5OH(i) -277.6 H2O(i) -285.8 H2O(g) -241.8 CO2(g) -393.5 CO(g) -110.5 H2O2(i) -187.6 C3H8(g) -103.8 What is the ΔH for the following reaction? C2H5OH(i) + 3O2 (g) --> 2CO2(g) + 3H2O(i)In which species (NO3- or CH3NO2) are the N-Obond(s) longer? In which species (NO3- or CH3NO2) are the N-Obond(s) weaker?Please help me I don't understand this, its a simple mecahnism but im lost.
- Answer Q35, 34 showing detailly all explanationsCalculate ∆Hrxn for the following reaction:N2H4(l) + CH4O(l) → CH2O(g) + N2(g) + 3H2(g)Use the following reactions and given ∆H values.2 NH3(g) → N2H4(l) + H2(g) ΔH = +22.5 kJ/molN2(g) + 3 H 2(g) → 2 NH3(g) ΔH = +57.5 kJ/molCH2O(g) + H2(g) → CH4O(l) ΔH = +81.2 kJ/molCalculate the ∆Hrxn for the reaction below: MgCl2 (aq) + 2 KOH (aq) ---> Mg(OH)2 (s) + 2 KCl (aq) ∆Hfo for MgCl2 (aq) = -797.1 kJ/mol ∆Hfo for KOH (aq) =-424.2 kJ/mol ∆Hfo for Mg(OH)2 (s) = -924.7 kJ/mol ∆Hfo for KCl (aq) = -419.6 kJ/mol ∆Hrxno = ? kJ Don't forget the sign and write your 4 significant figures.

