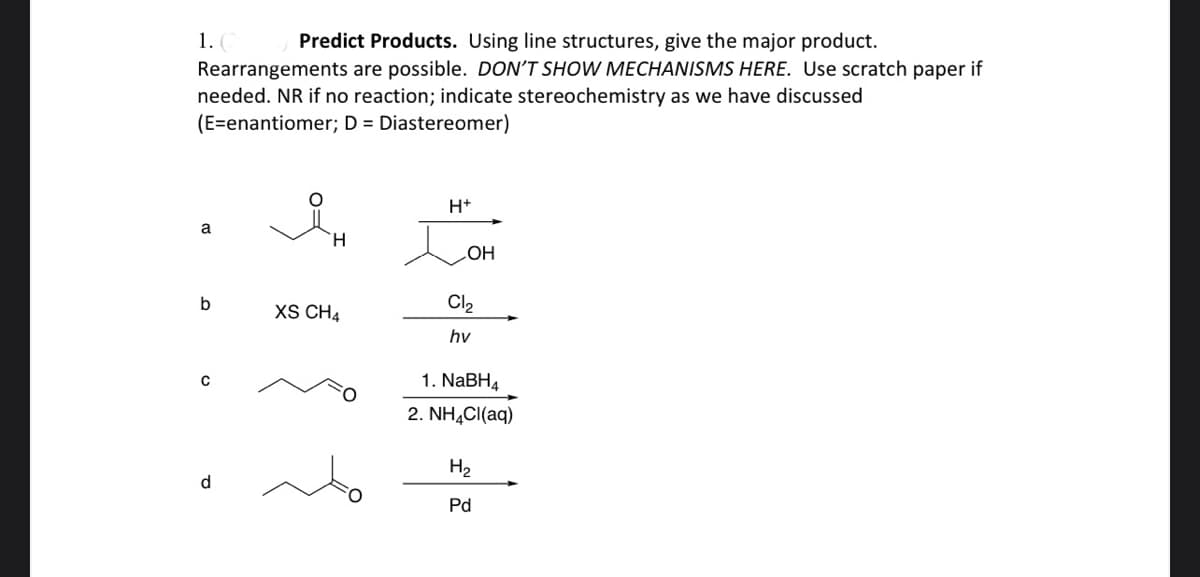
Q: Balance the following equations:
A: balancing of reaction is done to follow the conservation of mass.
Q: ain a8 me is worth significantly more than words.. (X) ()
A: Boiling point of amines depend upon intermolecular forces present in amines.
Q: 4.21 Write ionic and net ionic equations for the followingreactions:
A: When a chemical reaction occurs, it can be written in different forms of the chemical equation.…
Q: a CH3CH2C =CH CH3 CHg ĊCH3 1. ВнЗ (A 2. Н202 (в) кMпо4 or оз 1. NaNH2, NH3 2. СНзон D H2SO4, H20,…
A: HYDRATION OF ALKYNE With the expansion of water, alkynes can be hydrated to frame enols that…
Q: KMNO4 H20 Br NaOH H20 AICI3 CS2 1. LIAIH4 2. H* NAOH HO.
A: The final products are given below -
Q: OH NH₂ å 1. bl NH₂ 2. H₂O 1.1 eq 2. H₂O/ HCI (excess) -NH₂
A: In the last two question, 2c) and 2d) we will draw the organic major product from the starting…
Q: 35 Find Paotow =7 Protow 2. 99 Tc' Find electn = W Elecct now 3. 16,2- Find neutns =8 nethons
A: No. of protons is equal to the atomic no. Mass no. is equal to the no. of protons and no. of…
Q: Me Br2, hv Me Me
A:
Q: Draw the structures of compounds A–H in the following synthesis scheme.
A:
Q: 5) 6) O2(g) Al(s) + Ba(CIO3)2(aq) + ->> Al₂O3(s) Na3PO4(aq) Ba3(PO4)2(s) + NaClO3(aq)
A: A chemical reaction is defined as a method where reactants react with each other under reaction…
Q: H3O+ 1. NaCN b. CH;CH,CH,CH2B 2. CH3CH2CH2MgBr 3. H3O* С. 1. SOCI2 "СООН 2. CH3CH,NH2 3. LIAIH4 4.…
A: All reagents have a particular function. Using it we can provide the following answers-
Q: Identify compounds A-M in the following reaction sequence.
A: Given,
Q: c) NaOH (aq) + H2O(1) → Na*(aq) + OH(aq) d) HCIO3 (aq) + H2OM → H*(aq) + CI03 (aq)
A:
Q: ACHOP-S> C-F > F-F SWAAA C-O > C-F > F-F CO C-F > P-S > Na-F EAPS>C-0> C-F > Na-F KaA CF>C-0 > P-S >…
A: Given that : We have to arrange the following bonds according to the decreasing bond polarity : P-S,…
Q: с. 1. 2 equiv PhLi THE EtO. 2. H30* d. NABH4 МеОН
A:
Q: 10): Me Et 11) Me Cl2 Me... HOSHH
A: As per bartleby guidelines we are supposed to answer only one question. So I have answered the first…
Q: Cl2 n NH,CI ACI В -C+n HCI EtMgCl 6 NaOEt F+ G D+E
A: In the given reaction, phosphorus trichloride is the main group halide AClx. Main group element:…
Q: Draw the structures of compounds A–I in the following synthesis scheme.
A: In the first reaction, at higher temperatures, 1,3-butadiene gives 1,4 or we can say, Michael…
Q: NaCIO (aq) + H2SO4 (aq) + 2 KI (aq) → NaCl (aq) + H2O (1) + K2SO4 (aq) + I2 (aq)
A: Write the balanced molecular equation Split electrolytes to their corresponding ions to achieve…
Q: 1) NaBH4 2) acid (а) 1) LİAI[OC(CH3)3]3H 2) acid CI (b) 1) LIAIH4 2) acid (c) OCH3 1) LIAIH4 2)…
A: Given reactions:
Q: (A) Cl2, H2O (B) H2O, H2SO G (4) HCl, H2SO4 (D) H2O, Hoot 애
A:
Q: A. Br2 aq. HBr H;CCO,H В. H3C' a.) LDA b.) PhSeBr c.) H2O2
A:
Q: Draw the products of each reduction reaction.
A: Hello. Since the question contains multiple sub-parts, the first three sub-parts will be only…
Q: d. HCl(aq) + KOH(aq) H2O(1) + KCI(aq) - e. Zn(s) + CuSO«(aq)ZnSO4(aq) + Cu(s) | f. NaH,PO4(aq) +…
A:
Q: _Pb(NO3)2 (aq) _KI (aq) _Pbl2 (s) + _KNO; (aq) b.
A: In this question, we want to Balanced this given Equation because coefficient is not given. How we…
Q: H3O*, + Liz(CH3),CuCN THE me OH ? H OH
A: Q1. Gilman reagents. Q2. Oxidative cleavage of 1,2-diols.
Q: Write balanced net ionic equations for the following reactions:
A: Molecular equation ⇒ 2AgNO3aq+ZnCl2aq→2AgCls+ZnNO32aqTotal ionic…
Q: 4. Zn(s) + Ni2(aq) Zn*(aq) + Nics) 5. Pb(s) + Sn*2(aq) Pb*2(aq) + Sn(s) 6. Al(s) / Al*3(aq) //…
A: Since you have posted multiple questions, we will solve first one for you. To get remaining parts…
Q: Li, NH3 ELOH CH3CI AICI3 Br2 hv но. NaOH(aq) O2, 500°C MnO2 sOCl, но CHCI3
A:
Q: Draw the products of each reaction, and indicate the stereochemistry at all stereogenic centers.
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: c) 2CO2(8) + O2(8) → 2CO2(8) d) 302(8) e) 2NH3(8) + CO2(g) → NH2CONH2(aq) + H2O1) → 203(8)
A:
Q: H2SO4 Но CI NH2 1) A D O, H3O* NAOH 2) РСС, 1) ЗCН31 CH2Cl2, A 2) Ag20, H-0, Д C E B
A: The question is based on the concept of organic reactions. we have to identify the product formed in…
Q: HO 'N' N. ČH3 ČH3 О 1) (СНЗСH2)2NH, HCІ (сat.), NaBHЗCN О 1)) LAH;B ) нЗо+; 2) (СHЗСН2)2NH, HCІ…
A: The details solution for this reaction is provided below in attach image.
Q: (a) Iz(s) + 5 Cu²* (aq) + 6 H2O(1) (b) Hg²*(aq) + 21 (aq) (c) H2SO3(aq) + 2 Mn(s) + 4 H*(aq) → S(s)…
A: I2(s) + 5Cu+2 (aq) + 6H2O → 2IO3-(aq) + 5 Cu (s) + 12 H+ E cell of the reaction = Ecathode…
Q: AgNO3 (aq) → ? a) CaBr2(aq) b) LizSO4(aq) ZnCl2{aq) c) HCl(ag) KOH(aq)
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both…
Q: Zn(s) +. ZnCl (aq) + H2(g) 3. HCl(aq) Cu(s) + HC(aq) CuCly(aq) +. H2g) - Mg(s) + HCI(aq) MgCl2(aq) +…
A:
Q: 1. LAH CO2ET 2. aq H*
A:
Q: OH KMnO4 H3C- -CH3 H,SO4, H2O C. OH H,PO, D.
A: The reactions given are,
Q: 24. cont. NACN, (CH,),NCHO 30°C 1) NABH4 H 2) НC\ag) iBr KOH, CH;CH,OH f) 78°C Itt 1) Hg(O,CCF3)2,…
A:
Q: e) Ba(OH)2(a9) + H2OM → Ba (aa) + 20H(aq) f) Be(OH)2(a9) + H2OM → Be* (20) + 20H(29)
A: Answer:- This question is answered by using the simple concept of nature of reaction which depends…
Q: AgNO3(aq) + K₂SO4(aq) 2AJ H3PO4 (aq) C2H6 (g) + 1504 NaOH (aq) + O2 (g) KNO3(aq) + Ag2SO4(s))…
A: Count the number of atoms of each element present on both sides of the reaction and then balance…
Q: (a) Iz(s) + 5 Cu²* (aq) + 6 H;O(I) –→ 2 103"(aq) + 5 Cu(s) + 12 H*(aq) (b) Hg²*(aq) + 21 (aq) →…
A: I2(s) + 5Cu+2 (aq) + 6H2O → 2IO3-(aq) + 5 Cu (s) + 12 H+ E cell of the reaction = Ecathode…
Q: Write net ionic equations for the following reactions:
A: A net ionic equation is a chemical equation that only consists of chemical species which is…
Q: 9) 10) Al(s) + F2(g) + ― HCl(aq) NaBr(aq) → AICI3(aq) + NaF (aq) + H₂(g) Br₂(1)
A: Balanced equation means number of each atom in both side are present in equal number.
Q: . ) HCN(aq)+ H2O(l)⇌H3O+(aq)+CN−(aq)HCN(aq)+ H2O(l)⇌H3O+(aq)+CN−(aq) Express your answers as…
A:
Q: 3 но. OH Он 19 18 /14 13 17 15 16 B 12 11 OH Oll HN. HOll. 20 NH2 10 6 7 H2N 8 ÕH H2N NH2 5. 9. A.
A: According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority.2) If first atom is…
Q: OH OH H2N CHa CH3 CH2OH но но. OH H3C CH: NH H HN. NH NHCH3 HOOC CONH2 но HO, 32.7.2
A:
Q: Identify each of the following reactions as a precipitation, neutralization, or redox reaction:
A: (a) Redox reaction. Oxidation number of Mg is changed from 0 to +2. (b) Neutralization reaction.…
Step by step
Solved in 2 steps with 1 images

- Could you please show me how to draw in the electron arrows to account for the following mechanistic step. Solve it asapThe synthesis above can be performed with some combination of the reagents listed below. Give the necessary reagents in the correct order. Be sure to consider stereochemistry and regiochemistry.A. NaOHB. mCPBAC. OsO4, tBuOOHD. NaHE. H2, LindlarF. CH3CH2CH2BrG. CH3CH2ClH. NaNH2I. H3O+J. Na, NH3(l)Assign the stereochemical configuration (E or Z) for the alkene below. Show your work, indicating clearly which groups are assigned high priority (e.g., through assigning the groups numbers, circling only the high priority groups, or labeling groups as high or low).
- Your task is to synthesize trans-1, 2-cyclohexanediol, beginning with cyclohexene. What reagent(s) will accomplish this task in good yield? Group of answer choices cold aqueous KMnO4 PCC, CH2C12, 0 oC excess dilute aqueous acid (H2O /H +1) MCPBA followed by dilute aqueous acid (H2O / H+1) Please answer fast i give you upvote.pls explain in detail and find the major products for each oneThe compound 3,4-dimethyl-hexan-3-ol of 3R, 4S configuration is treated with a concentrated HBr solution at room temperature. A mixture of two stereoisomers is obtained.If the reaction mixture above is heated, the appearance of several other compounds is observed. 1) Draw the different compounds obtained using the wedge-flywheel representation. 2) What is the majority product? Explain 3) Propose a modification of the experimental conditions in order to obtain the exclusive formation of these compounds obtained after heating
- Based on the more stable conformation (conformation A), need help providing two mechanisms for the E2 elimination of the protonated version of cis-1 using a general base (B:) to give alkenes 2 and 3. Clearly show which hydrogens meet the stereochemical requirements for E2-elimination Using Zaitsev’s rule, indicate which is favoured. Pictured is also a reaction mechanism to show what the alkenes 2 and 3 are. Thank you :)Predict the mechanimsm and reactants to synthesize the picture below. Write legibly and ill give you a thumbs up.Draw mechanism arrows and then predict the major products and stereochemistry. Also include the labels of thermodynamic and kinetic 1-methylcyclohexene with BH3 followed later with –OH, H2O2, and H2O.
- predict the products: XCL2+ E3PO4 --->Cyclohexanone + (CH₃)₂NH and continue with catalyze by H+. Predict the product of this reactionOChem HELP... Draw the conformational structures (chair or boat conformations) for the MAJOR product formed when 1-tert-butylcyclohexene reacts with each of the following reagents. Also, indicate if the product obtained is racemic form. a) Br2, CCl4 b) Br2, H2O c) OsO4, then aqueous NaHSO3 d) ICl e) mCPBA, then H3O+, H20 f) O3, then Me2S (conformational structure not required) g) BH3:THF, then H2O2, HO- h) D2, Pt i) Hg(OAc)2 in THF-H2O, then NaBH4, HO- j) BD3 :THF, then CH3CO2T

