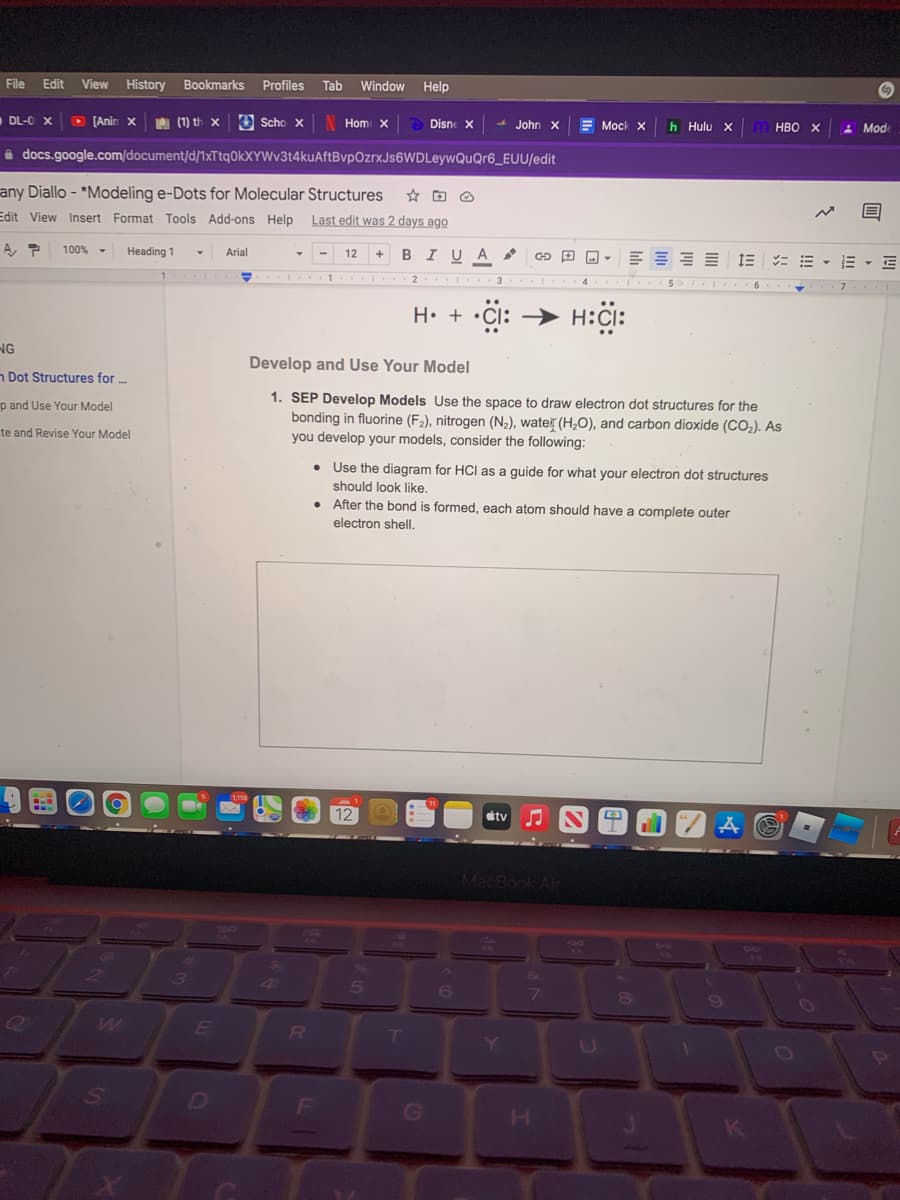
H• + •Cl: Develop and Use Your Model 1. SEP Develop Models Use the space to draw electron dot structures for the bonding in fluorine (F2), nitrogen (N,), water (H,O), and carbon dioxide (CO,). As you develop your models, consider the following: • Use the diagram for HCl as a guide for what your electron dot structures should look like. • After the bond is formed, each atom should have a complete outer electron shell.
H• + •Cl: Develop and Use Your Model 1. SEP Develop Models Use the space to draw electron dot structures for the bonding in fluorine (F2), nitrogen (N,), water (H,O), and carbon dioxide (CO,). As you develop your models, consider the following: • Use the diagram for HCl as a guide for what your electron dot structures should look like. • After the bond is formed, each atom should have a complete outer electron shell.
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
hello please help as much as possible
Transcribed Image Text:File
Edit
View
History
Bookmarks
Profiles
Tab
Window
Help
DL-O X
D [Anir X
A (1) th x O Scho x
Disne X
- John X
Hom x
E Mock X
h Hulu
HBO
- Mod
i docs.google.com/document/d/1xTtq0kXYWv3t4kuAftBvpOzrxJs6WDLeywQuQr6_EUU/edit
any Diallo - *Modeling e-Dots for Molecular Structures
Edit View Insert Format Tools Add-ons Help
Last edit was 2 days ago
A P
100% -
Heading 1
в IUA
Arial
12
= E - E - E
2
3
5
4
H• + •Cl: → H:CI:
NG
Develop and Use Your Model
n Dot Structures for .
1. SEP Develop Models Use the space to draw electron dot structures for the
bonding in fluorine (F2), nitrogen (N2), wate (H,O), and carbon dioxide (CO2). As
you develop your models, consider the following:
p and Use Your Model
te and Revise Your Model
• Use the diagram for HCI as a guide for what your electron dot structures
should look like.
• After the bond is formed, each atom should have a complete outer
electron shell.
12
tv
MacBook Air
3.
5.
6.
8O
W
Y
U
F
G
Transcribed Image Text:View
History Bookmarks
Profiles
Tab
Window
Help
• [Anin X
A (1) th x e Scho x
N Hom X
> Disne X
- John x
E Mock x
h Hulu X
м нво х
: Mode X
E Oum x
ogle.com/document/d/1xTtq0kXYWv3t4kuAftBvpOzrxJs6WDLeywQuQr6_EUU/edit
- *Modeling e-Dots for Molecular Structures
Insert Format Tools Add-ons Help Last edit was 2 days ago
TURI
12 + BI UA GD
= E - E - E E X
100% -
Heading 1
Arial
2 3
4 SI I 6
Copyright © Savvas Leaming Company LLC. All Rights Reserved.
Savvas is not responsible for any modifications made by end users to the content posted in its original format
ructures for .
e Your Model
Levise Your Model
NAME
DATE
CLASS
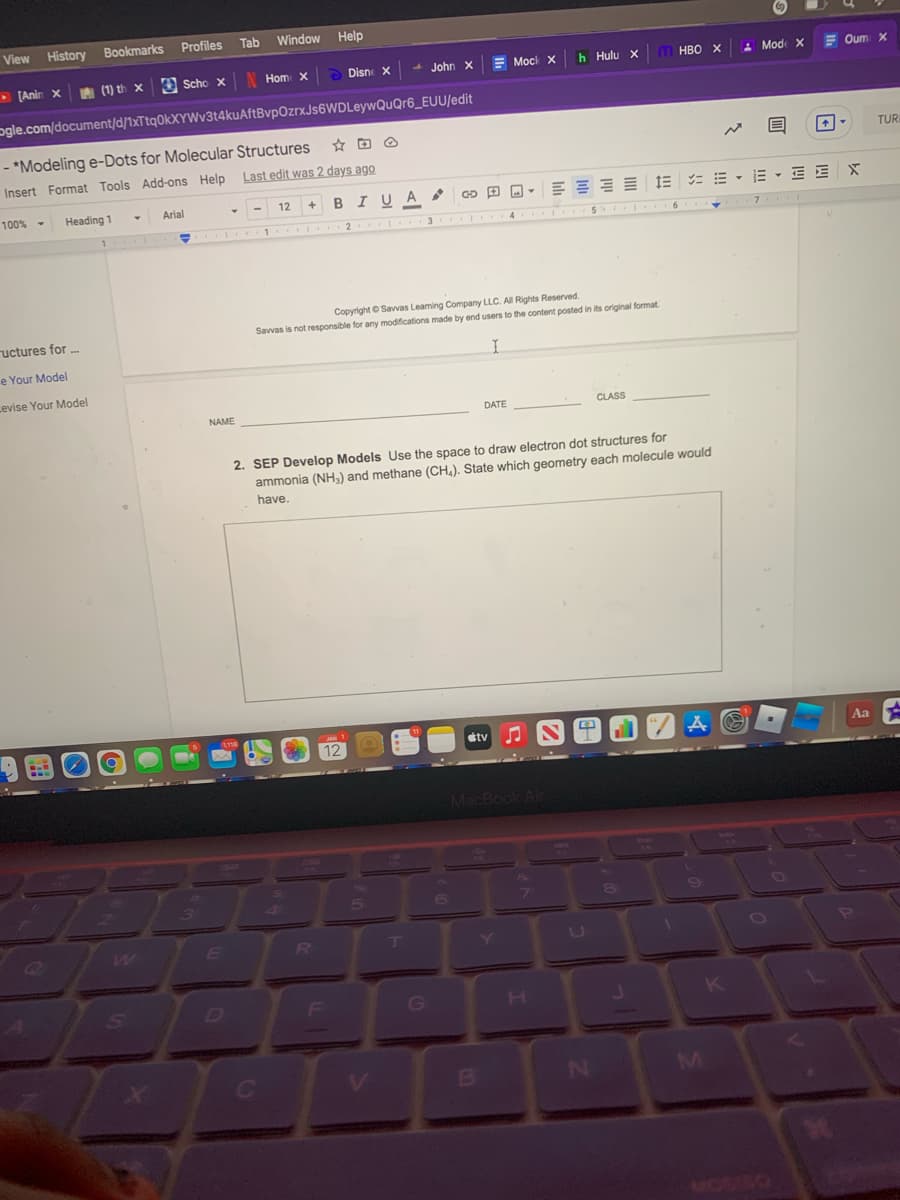
2. SEP Develop Models Use the space to draw electron dot structures for
ammonia (NH,) and methane (CH,). State which geometry each molecule would
have.
JAN
etv
Aa
12
MacBook Alr
3)
4s
5.
RU
LU
D
H.
N
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning
