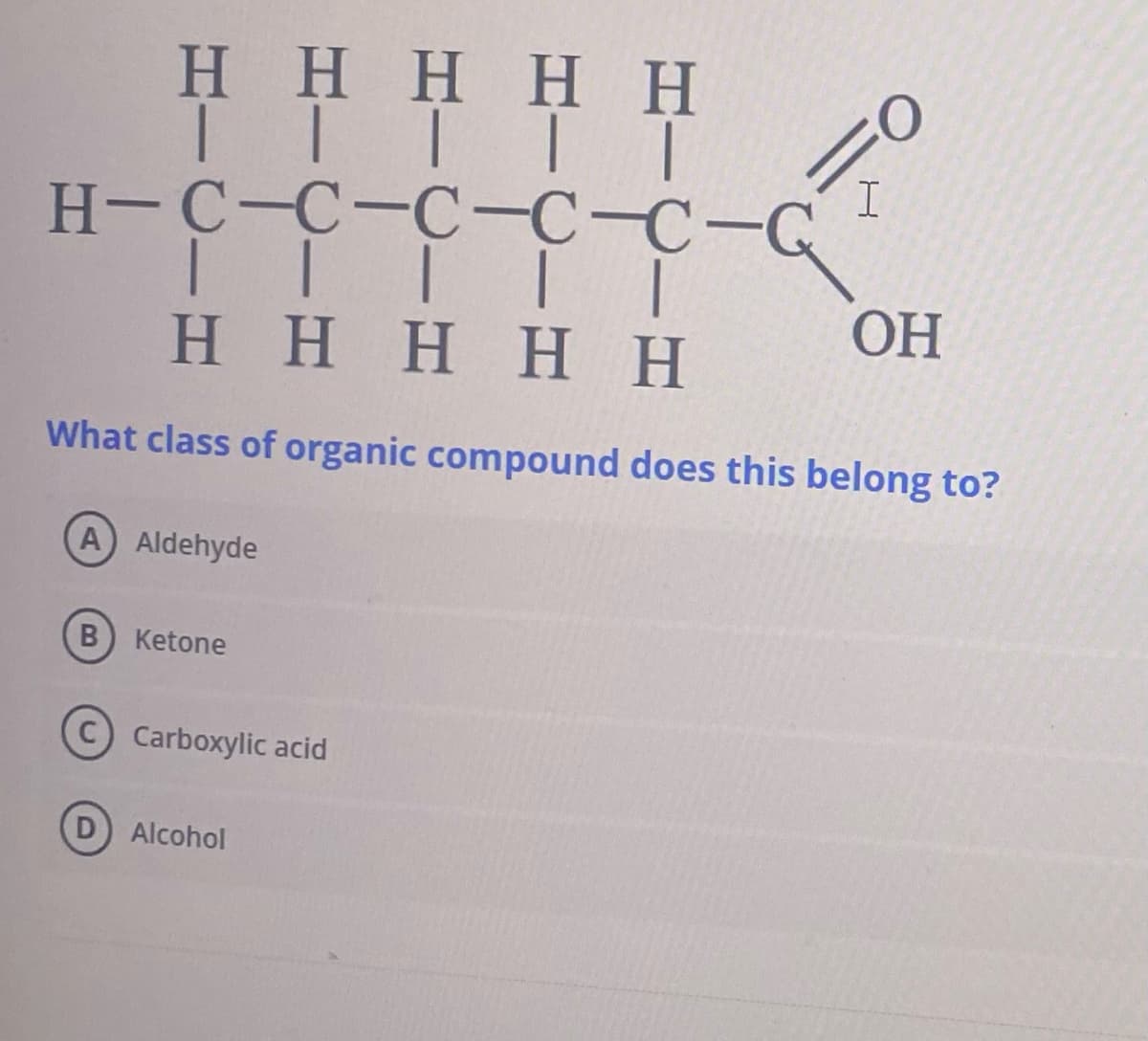
Η Η Η Η Η ΤΙΤΙΤ H-C-C-C-CC-G | | | | | Η Η Η Η Η I ОН
Q: 10. Vitamin B6: coenzyme form, functional groups, mechanism of action, biological role, sources,…
A: Enzymes are biological catalysts that catalyse biochemical reactions. Most enzymes are made up of…
Q: Question 15 of 25 Which of the following is true for the acid-base properties of amino acids? Select…
A: The proteins are made of 20 naturally occurring amino acids. The net charge on the side chain of the…
Q: 1. The role of cofactors in the realization of the catalytic activity of enzymes.
A: Co-factor is the non-protein component that is required for an enzyme activity. Co-factors can be…
Q: . What is the effect of boiling in the test tube with starch solution and saliva?
A: “Since you have asked multiple questions, we will solve the first question for you. If youwant any…
Q: ________________ is the general term for molecules that enzymes modify to produce reaction products.
A: The enzymes are biological catalysts that are mostly protein in nature. The enzymes increases the…
Q: Which carbohydrate test can you use to distinguish a monosaccharide from a disaccharide? hexoses…
A: A carbohydrate is a biomolecule which consists of carbon, hydrogen and oxygen atoms. The hydrogen…
Q: what element is this? after MDA reacts with something Ph CH3 N CH3
A: MDA is malondialdehyde . It is a product of lipid peroxidation in corals. Determination of this MDA…
Q: The side chain of which residue can be hydrogen bond donor: a. Thr b. Ala c. Ile d. Pro e.…
A: There are twenty standard amino acids that differ from each other on the basis of their side-chain…
Q: TABLE 3-1 Properties and Conventions Associated with the Amino Acids pk, values Amino acid…
A: pI refers to the isoelectric point or the pH at which the amino acid carries a positive charge and a…
Q: Since higher concentration colcemid will result in shorter chromosome, you want to change your…
A: It is given that: Molar concentration of the stock solution: 10 ug/ml. Desired dilution…
Q: During anaerobic conditions, lactate travels from the muscle to the liver via the bloodstream. What…
A: The Tricarboxylic acid(TCA) cycle is inactive under anaerobic conditions, thereby the process of…
Q: 7. H 1 H-C -OH H-C-OH 1 H -C 8. H 9. НО I 1 H J=D 0=)) с HIV-H OH HH -C-C-H I OH OH F-DIE-DI - CH…
A: 7. Chemical formula for structure number 7. = C4H8O4 or C4(H2O)4. Yes, this structure is a…
Q: Is the BSA a good stadard to use in the bradford assay?
A: Introduction Proteins are the most abundant macromolecule present in our body. Proteins are made up…
Q: DNA: Whats the structural and functional differences between heterochromatin and euchromatin?
A: DNA, the genetic material is organized as chromosomes inside the cell nucleus. The chromosomes have…
Q: The figure below shows a polynucleotide: a) Label the 5' and the 3'…
A: Polynucleotide are polymer of nucleotides which are formed by the condensation of nucleotide. The…
Q: Nomenclature of vitamins (empirical, biochemical, medical).
A: Vitamins are organic nutrients which can not be synthesized in the body, hence, need to be supplied…
Q: Now use the equations above and the empirical values below to calculate VO₂ (the rate of oxygen…
A:
Q: what does this picture indicate? what is difference bewteen A, B, and C ?
A: INTRODUCTION : Myoglobin - It is an iron and oxygen binding protein found in the skeletal &…
Q: What is the product of the beta oxidation of this fatty acid?
A: The given fatty acid is caproic acid. Beta-oxidation is the oxidation of fatty acid on the…
Q: Question 1 Which of the following conditions would fail to produce a triacylglycerol? Group of…
A: Triacylglycerol : A triglyceride or triacylglycerol (TG) molecule is made up of a glycerol as…
Q: 384 Structure and Function of Hemoglobin Q4.4- In addition to O2, another diatomic molecule that…
A: Carbon monoxide is a colorless, odorless, tasteless and stable oxide of carbon. Its molecular…
Q: An egg placed in boiling water becomes hardboiled in 3 minutes. What changes have occurred in the…
A: All proteins have three levels of conformations known as primary, secondary, and tertiary.…
Q: What is the physical or structural difference between heterochromatin (also called "heterochromatic…
A: The term "chromatin" describes the DNA and protein combination that makes up the chromosomes present…
Q: Which of the following substances can deliver electrons to the ETC to help pump out H+ across the…
A: Electron transport chain is a chain of electron carriers that transfer electrons to molecular…
Q: Using an arrow, draw the site of cleavage for the following peptides that are reacted by: Pepsin…
A: Site specific proteases are enzymes that cleaves polypeptide chains only at specific points. Trypsin…
Q: What charged groups are present in leucine at a pH = 7? OA) 1× NH3¹ B) 1 x COO OC) 1× NH3¹ and 1 ×…
A: Amino acids have a central carbon atom known as α-carbon atom. Each amino acid has an amino group,…
Q: The β-Sheet is formed mainly by a. hydrophobic interaction b. hydrogen bonding between the…
A: Alpha-helix and beta-sheets are types of secondary structures that are mainly found in protein…
Q: Calculate ΔGinward. Is energy required for transport to happen? The internal pH of a lysosome= 4.0…
A: The amount of change in free energy i.e. change in Gibbs free energy for the transport of an ion…
Q: Identify three enzymes and the scientists who worked on them. What are the importance of these…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: 11. Use the data below to answer the following question: How much more energy is stored in a gram of…
A: Glycogen and fat are two molecules used by human body to store energy. The inside and even outside…
Q: Which of the following statements is CORRECT? A) Hexokinase IV is allosterically inhibited by…
A: Enzyme plays an important role in all the metabolic activities in our body. They themselves remain…
Q: 1. What two enzymes in glycolysis are regulated by high ATP concentrations?
A: INTRODUCTION : Glycolysis : It is a metabolic pathway in which glucose is breaken down into pyruvate…
Q: H ОН CH2OH D H ОН H 0 البلاد - ОН H CH2OH H H ОН H 0 ОН Which of the following statements correctly…
A: The sugars often contain alcohol and carbonyl functional groups and the intramolecular hemiacetal…
Q: Lipids Function in Cell Signaling Q5.2: The lipids 7-dehydrocholesterol and arachidonate are…
A: Cell signalling is the process in which the cell responds to certain molecules called signalling…
Q: Which of the following concerning hemoglobin is true? a) BPG decreases at high altitudes and causes…
A: 1 Hemoglobin is a globular protein, ie it is roughly spherical. It is an oxygen-binding protein that…
Q: Describe the specific multi-enzyme example discussed in class of how enzyme activity can be altered…
A: Enzymes are usually comprised of proteins. It work as a catalyst for several biochemical reactions.…
Q: How does mottling on tablets appear during drying process? Give your insights on how mottling can be…
A: Introduction During tablet processing different types of defect may arise. An tablet should be free…
Q: In glycatysis, conversion of a molecule, of glucose, into 2 Molecules of Pyryvate, results in a net…
A: Glycolysis is the process of conversion of one molecule of glucose to two molecules of pyruvate. A…
Q: is the statement true or false?
A: The pentose phosphate pathway carries out the complete oxidation of glucose by completely oxidizing…
Q: Discuss the role (direct or indirect) of carbohydrates on cancer and Suggest an appropriate…
A: Carbohydrates, lipids, and proteins are the biomolecules that are required in large amounts by the…
Q: 10. CH OH HO -0-3 OH C I II. Cд H_Os 12. С 8 Hile O_ 13. СА Н 14. C Hio Os 15. C_H_D- 16. C_H 1 20 -…
A: Carbohydrates are polyhydroxy organic compounds with a general formula of (CH2O)x. Carbohydrates can…
Q: Based on the structure of sucrose below, is it classified as a reducing sugar? 6CH OH ОН ОН 3…
A: Chemically, carbohydrates are polyhydroxy aldehydes/ketones. They have the general formula :…
Q: Conversion of disaccharides to monosaccharides are associated with: O stomach O intestinal mucosa…
A: Chemically carbohydrates are polyhydroxy aldehydes or ketones. They have the general formula :…
Q: What is one technique or property of a protein that you could use to monitor the fractions so you…
A: In column chromatography, there is a stationary phase and the mobile phase. The stationary phase…
Q: In order to study protein structures and functions, many protein techniques have been developed for…
A: Proteins are composed of twenty standard amino acids attached together via peptide bonds. These…
Q: Question 21 of 25 What are Gram-negative bacteria? Select the correct response(s): They have a cell…
A:
Q: 5. Oxaloacetate, glycerol, and lactate can be used in gluconeogenesis. T F
A: Sometimes organisms need a method to synthesize glucose from non-carbohydrates precursors. The…
Q: 6. What term is used to describe reactions that generate metabolic intermediates of a pathway? A.…
A: The reactions that generate metabolic intermediates of a pathway is termed as - Answer- B.…
Q: Need help, please.
A: Malate dehydrogenase is an enzyme that catalyzes the final reaction of the TCA cycle. In the first…
Q: Show the Hydrolysis of a dipeptide containing the amino acids threonine and methionine.
A: Amino acids are joined together in a linear manner by peptide linkage, known as the primary…
Step by step
Solved in 2 steps

- A monosaccharide designated as an aldehyde sugar contains (a) a terminal carboxyl group (b) an internal carboxyl group (c) a terminal carbonyl group (d) an internal carbonyl group (e) a terminal carboxyl group and an internal carbonyl groupThe following chemical reactants produce the ester ethyl ethanoate (C4H8O2): C2H6O + CH3COOH What type of reaction occurs to make ethyl ethanoate? condensation hydrolysis combustion acid-base reactionThiamin, or Vitamin B1, contains a positively chargedfive-membered ring called a thiazolium ring. Explainwhy the thiazolium ring is aromatic.
- Which of the following substituents are listed in correct order of priority from highest to lowest (line indicates bond to alkyl C)? A. I–, H–, Cl–, Br– B. H–, CH3 –, CH3-CH2–, Br– C. Br–, I–, F–, OH– D. Br–,CH3-CH2–, CH3 –, H– E. H–, OH–, CH3–, Br–The molecular formula for glucose is C6H12O6. What wouldbe the molecular formula for a polymer made by linking tenglucose molecules together by dehydration reactions?(A) C60H120O60(B) C60H102O51(C) C60H100O50(D) C60H111O51Which of the following compounds would be most soluble in carbon tetrachloride, CCl4? a. NH3 d. CH3OHb. H2O e. C6H6c. HCl
- Draw the condensed structural formula for the fatty acid whose numerical shorthand designation is 18:2 (△ 9,12)When blood levels of glucose are higher than normal,glucose molecules react with protein side chains such as thatof lysine residues to produced glycated side chains. Explainwhy this process is dangerous. What is the name of the firstfunctional group that is formed during glycation?In the Equation: HCO3- + HCl --------> Conjugate Acid + Conjugate Base a ) Which reactant is the Acid? b) Which reactant is the Base? c) What Conjugate Acid will form in this reaction? d) What Conjugate Base will form in this reaction?
- Sphingolipids can contain of all of the following except A. glycerol. B. sphingosine. C. phosphocholine. D. a mono- or oligosaccharide.Draw an aspartic acid and label: a. The acarbon, aamino, and acarboxyl groups (you can use the chart at the end of the exam for the R group). b. At pH=11, what would be the overall charge on aspartic acid in solution? You can use the short-hand rule...A carboxyl group is symbolizeda. —OH.b. —NH2.c. —CH3.d. —CH2OH.e. —COOH.







