Q: What is the molar solubility of stannous sulfide? Ksp = 1.0 x 10-26 2.5 x 10-6 M 1.4 x…
A:
Q: A 0.050 mL drop of 6.0 mol/L hydrochloric acid is added to 1.0 L of a 1.0 mol/L aqueous silver…
A:
Q: Nitroahiline had been observed in experiments on biomass From pine needles and can be used as a…
A:
Q: 1. Methanol (CH3OH), is an excellent fuel and can be made by the reaction of CO and H2. CO + 2H2 ––>…
A: Answer: In a chemical reaction limiting reagent is the reactant that exhaust first in the reaction…
Q: Question 28 The decomposition of hydrogen peroxide, H₂O₂, has a rate constant, k = 3.66 x 10-3 s-1.…
A: This question is the related to the first order Kinetic so in this question given, initial…
Q: Consider the rate constant for ester hydrolysis at different temperatures. What is the activation…
A:
Q: O₂N- HOOC -NH NO₂
A: In the given question we have to write the name of the organic compound. the given compound has…
Q: 1. Calculate the amount of heat that must be absorbed by 10.0 gram of ice at -20C to convert it to…
A:
Q: Question 1 Consider the reaction mechanism for NO2(g) + CO(g) → CO2(g) + NO(g) (1) NO₂(g) + NO₂ (g)…
A:
Q: 1, Br₂, Fe Brz 2. Li 3. Cu I er Cl₂, FeCl Ph₂CuLi
A: In this question, we will write the reaction mechanism for the reaction. You can see below in the…
Q: Question 13 of 26 Using the data in the table, calculate the rate constant of this reaction. A+B C +…
A:
Q: For the decomposition of A into B and C, what is the order of the reaction given the following data?…
A:
Q: Classify the phrases as pertaining to either sphingophospholipids (sphingomyelins),…
A: Sphingophospholipids and Glycerophospholipids are two classification of phospholipids according to…
Q: band gap on insulator i understand the band gap difference between conductor, insulator, and…
A: From the given data we are going to answer why there is a large energy gap in insulators.
Q: The IR spectrum below represents which of the following compounds? Absorbance/% www 4000 3000 500…
A:
Q: ОН о 1. H2NNH2, H+ 2. КОН, Д он
A: The Wolff-Kishner reduction is a reaction used to convert carbonyl functionalities into methylene…
Q: Consider this initial rate data at a certain temperature in the table for the reaction OH(aq) [I]o…
A: The rate law for the reaction can be written as: rate=kOCl-xI-yOH-z ......(1) Here, x is the order…
Q: 2. What is the normal boiling point of the following solutions? (a) 23.5 g NaCl in 120 g H₂O and (b)…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: D= C=O CH3CH₂OH CH₂CH3 tetrahydrogestrinone (THG), a "designer" synthetic steroid HO aldosterone…
A: The ring structure that is common to all steroids is:
Q: If 1000 g of water from Angat River contains 0.0075 g of Ca²+ ion, what is the concentration in ppm…
A: Given: Mass of Ca2+ ion = 0.0075 g Mass of solvent = 1000 g
Q: For the following reaction, 3.64 grams of hydrogen gas are allowed to react with 48.9 grams of…
A: given, mass of hydrogen gas = 3.64 g mass of iodine = 48.9 g we need to calculate mass of…
Q: Rank the substituents shown below in order of Cahn-Ingold-Prelog priorities (1= highest priority…
A:
Q: A sample of gas was contained in a 245 mL flask at a temperature of 23.5 °C and the pressure was…
A:
Q: 1. Consider the following data set. Concentration, mg/L 0.100 0.200 0.300 0.400 0.500 Write capital…
A: We have find out slope, intercept and concentration of an unknown sample with an absorbance reading…
Q: 2. A solution is prepared by mixing 5 ml of ethyl alcohol in 100 ml of water. What is the volume…
A:
Q: An alloy contains 70.8 mol % palladium and 29.2 mol % nickel. Express the composition of this alloy…
A: Molar mass of palladium =106.4 g/mol Molar mass of nickel =58.69 g/mol Let ,palladium is present…
Q: Determine the molar enthalpy of combustion of an unknown fuel if a 2.75 g sample increased the…
A: The molar enthalpy of combustion can be determined by the ratio between the heat gained or lost by…
Q: Below what pH will free phenobarbital, a weak acid, begin to separate from a solution having an…
A:
Q: On March 20, 1997, FDA published a final rule on electronic records and signatures, which FDA will…
A: According to 21 CFR part 11,it states that electronic records and electronic signature are treated…
Q: 2. If you use 5.76mol of sodium fluoride (NaF) and dissolve this into 3.62kg of water, what will be…
A:
Q: A set of data was found to fit the Langmuir isotherm. The equation of the line is y = 9.04 x 10-³x +…
A:
Q: - Propose syntheses of the following compounds from benzene: (a) N,N-Dimethylaniline (b)…
A: Here in the given four conversion, the following points is amost common:- Reduction of -NO2 group…
Q: Experiment No 16: Determine the relative strength of two Acids by studying the hydrolysis of ester…
A: Below Answer is given.
Q: Identify the substance being oxidized and the substance being reduced Ag+Cu-3 → Ag-3+Cu2
A:
Q: 9.18 An intravenous solution to replace potassium loss contains 40. mEq/L each of K* and CI. How…
A: moles of individuals can be calculated from the concentration. Note: As the image contains multiple…
Q: Determine the reaction mechanism for the following reaction and explain CH₂O CH₂ O CH₂ Br CH₂ CH H3C…
A: The reaction of alkyl halide (R-X, R is alkyl and X is halide) with alkoxide (RO-) is known as…
Q: Question 13 What property makes polymeric micelles a desirable drug delivery system? small particle…
A: Polymeric micelle is made of amphiphilic copolymer. It is used as drug delivery carrier.It helps us…
Q: A balloon is sealed with 1.00 mol argon in a laboratory. The laboratory workspace is at sea level…
A: At sea level, atmospheric pressure is equal to 1 atm and room temperature is equal to 298 k. Argon…
Q: The phase diagram for the system nitrobenzene/hexane at 1 atm is shown below. 294 292 290 27317 0…
A: The phase diagram describes the dependency on different variables like pressure, composition,…
Q: Lipoproteins are globular structures that are responsible for transporting lipids through the blood…
A: We have to arrange the given boxes in appropriate column of the table
Q: A patient must drink 4.5oz of a prescribed solution. If only a teaspoon is available, how many…
A: Given, A patient must drink 4.5oz of a prescribed solution. The number of teaspoons should be…
Q: Given the following reaction and instantaneous concentrations, 2?2?(?) ⇌ 2?2(?) + ?2(?) K = 15 [H2]i…
A: The equilibrium constant of a reaction is given by the ratio between the product's concentration to…
Q: A certain local brand of “healthy” chicharon was found to contain 0.4132 g of NaCl after Fajan…
A: Given : Mass of NaCl = 0.4132 gm Molar mass of NaCl = 58.44 g/mol Molar mass of Na+ = 22.99 g/mol…
Q: How to figure out E and Z fornalkenes?
A:
Q: - How would you prepare the following amines, using both Hofmann and Curtius rearrangements on a…
A: The Hofmann and Curtius rearrangement involves the conversion of carboxylic acid to an amine. The…
Q: Given a ternary phase diagram. Where does the two-phase system exists? above the curve O below the…
A: For a given ternary phase diagram, where does the two phase system exist? = below the curve
Q: 4. How much energy (heat) is required to convert 52.0 gram of ice at -10.0℃ to steam at 100 °C?…
A: Given Mass of ice (m)= 52.0 g Heat needs(Q)=?
Q: n 14 ch of the following corresponds to a layer of ions unaffected by fluid motion and is separated…
A: According to Helmholtz this double layer consist of two opposite charged layers at a fixed distance…
Q: Plants undergo photosynthesis in order to provide oxygen into the air, which is vital for the human…
A: Since you have posted multiple questions I'll answer the first question for you. For the rest to get…
Q: A proposed mechanism for the Br-catalyzed aqueous reaction is shown below. Br H+ + HNO₂ + C6H5NH₂…
A:
kindly name the compounds
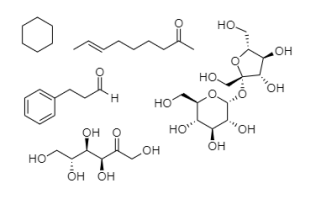
Step by step
Solved in 3 steps with 2 images


