Q: Consider the problem below: (Equation 1): 2C(graphite) + 2O2(g) à 2CO2(g) ΔH = -787 kJ (Equation ...
A: The given data is as follows: Equation 1: 2C(graphite) + 2O2(g) → 2CO2(g) ∆H1=-787 kJ Equation 2: ...
Q: A certain element forms an FCC unit cell structure with a density of 10.6 g/cm3. Identify the elemen...
A: Density of a unit cell is given by, d=ZMa3×NAWhere, d=density of unit cell M=molar mass a...
Q: Generate the solubility-product expression for a. CuI. b. PbI2. c. BiI3 d. MgNH4PO4
A: The solubility product, Ksp is the mathematical product of the concentrations of its dissolved ions ...
Q: 41. Consider the data showing the initial rate of a reaction products) at several different concentr...
A: Rate of reaction tells about the speed of reaction per unit time. Few chemical reactions are very ve...
Q: Scenario 1 Magnesium is a shiny, flexible metal that can be burned in the presence of air. When it i...
A: Chemical reaction :- When two or more substance reacts with each other to form a new substance havi...
Q: In spectrophotometric analysis, the relationship between the absorbance A and the analyte's concentr...
A: Beer's law states that the absorbance is directly proportional to the concentration of the absorbing...
Q: Write the balanced molecular chemical equation for the reaction in aqueous solution for lead(II) nit...
A: Formula of lead nitrate = Pb(NO3)2 Formula of sodium chloride = NaCl Pb(NO3)2 reacts with NaCl.
Q: Count the significant figures of each quantity. a. 579.420 b. 3.14159265 c. 2 × 1011 d. 50.1 ...
A: Count the significant digit in each of them--
Q: tyIron has an atomic weight of 55.85 g/mol and a density of 7.86 g/cm3. It crystallizes in a cubic u...
A:
Q: Explain how adding aqueous NH3 to the Combined AgCl- Hgz Cl2 frecipi tate brings about the Seferatio...
A: Explain how adding aqueous ammonia to combined AgCl - Hg2Cl2 precipitate brings about seperation of ...
Q: Solve the following problems: The freezing pt. of a solution containing 65g of a compound in 100g be...
A: Given data is as follows: The mass of the unknown compound (solute)= 65 g The mass of benzene (solve...
Q: LO-2 9. Neon (1°Ne) is a noble gas that gives a characteristic reddish glow when a voltage is applie...
A:
Q: c- Calculate the shortest Ni-As, As-As, and Ni-Ni distances. (Use: m(Ni) = 58.69 z/mol; m(As) = 74.9...
A: NiAs form a hcp structure. In this As atom lie on corner of hexagonal while Ni atom in the centre an...
Q: When copper is heated with an excess of sulfur, copper(I) sulfide is formed. In a given experiment, ...
A: Given, When copper is heated with an excess of sulfur, copper(I) sulfide is formed Also, Mass of C...
Q: Calculate the molecular weight, write down the different functional groups and determine the differe...
A: We are discus the molecular weight, functional groups and different non-equivalent hydrogens and ca...
Q: Calculate the mass of diphosphorus pentoxide (P2O5) that contains a million (1.0x1023) oxygen atoms....
A:
Q: Vanadium atom has a radius of 105 pm and crystallizes with a BCC unit cell. Determine the number of ...
A:
Q: See the example in Introduction for set-up of calculations. Calculate the number of (a) moles of N, ...
A: Given: Mass of N2 = 12.7 g.
Q: he best lewis structure will always have a negative formal charge on the most electronegative elemen...
A: Interpretation: We have to define that whether the statement is correct or incorrect.
Q: There are initially 10 mCi of 19Os which undergoes B- decay with a half-life of 15.4 d into 191mir. ...
A: Hii there, As there are multiple sub parts posted. we are answering first-three sub parts . If you n...
Q: Write the balanced NET ionic equation for the reaction when Pb(NO₃)₂ and LiCl are mixed in aqueous s...
A:
Q: when 22.0 g hydrogen combine with oxygen, 196.5 g of water forms. based on the law of conservation m...
A: Given data is as follows: The mass of hydrogen = 22.0 g The mass of water formed = 196.5 g The mass ...
Q: QUESTION 5 Predict the direction of the following reactions in aqueous solution: Write FORWARD (if...
A: Predict the direction of the following reactions in aqueous solution: HF (aq)+ HCOO-(aq) ↔ HCOOH (aq...
Q: Types of Structural Molecular formula Hydrocarbon formula Methane Ethene Propene Butane Pentane
A: Hydrocarbons:
Q: eed structures for molecular ion fragments and i neef fragmentation pattern predictions please
A: Fragmentation pattern very important step for mass spectroscopy. For above molecular ion fragmentati...
Q: OH
A:
Q: A sample of a compound that contains C, H, and O contains 41.38 % C and 3.45 % H. What is the empiri...
A:
Q: Calculate the molecular weight, write down the different functional groups and determine the differe...
A:
Q: If a student weighs 11 g of aluminum and needs twice as many atoms of magnesium as she has of alumin...
A: 1 mole = 6.022×1023 [Avogadro's number ] The mass of one mole of atoms of an element is known as it...
Q: Write the balanced molecular chemical equation for the reaction in aqueous solution for magnesium ch...
A: Write the balanced molecular chemical equation for the reaction in aqueous solution for magnesium ch...
Q: Determine the precision of the graduated cylinders, and get the volume with the right significant fi...
A: The Precision in the Measurements is depend upon the which device is used in the measurements . Mor...
Q: How moles of NazS will be pmduced many with. 2 moles of NaoH Balance it first NabH + HzS -7 Nazs t H...
A:
Q: Sulfur Oxygen Hydrogen a) Draw the Lewis structure of H2SO4 to be consistent with the experimental d...
A: Two S-O bonds are smaller than other two S-O bonds. This indicates that in H2SO4, two S=O bonds and ...
Q: : :O:
A:
Q: - recall the rules for naming ionic and molecular compounds that we learned; do not invent names: - ...
A: Here we have to interpret the above chemical reaction-
Q: Convert 3500cm2 to m2
A: Given Value in unit of Cm2 = 3500 cm2 Value in unit of m2 = ?
Q: Activity J: Write an equation showing the reaction of benzene with Cl2 and FeCl3? How does a substi...
A: Activity J : (1) When benzene is treated with Chlorine,in Presence of Ferric Chloride , then chloro...
Q: Given the following molar conductivities at infinite dilution: sodium propionate (NaPr), 85.92 S cm²...
A: Molar conductivity of NaPr = 85.92 S cm2mol-1 Molar conductivity of HCl =426.04 S cm2mol-1 Molar con...
Q: which of the following isthe correct lewis structure of one phosphorous atom with the appropriate nu...
A: Given : Structure of Compound between Phosphorous To find : Correct Lewis structure
Q: A 32.0 gram sample of chemical X is found to contain 12.0 gram of carbon. The law of Definite Propor...
A: To Solve this problem we need calculate the mass of carbon present in the 65.0 grams of sample . Fo...
Q: 2. The industrial method currently used to produce butanol is the hydration of butene. C,Ha(g) + H,O...
A: Given 4C(s) + 4 H2(g) → C4H8(g) ∆H = -7.1 KJ/mol ...
Q: A sample of solid biphenyl weighing 0.5260 g was ignited in a bomb calorimeter initially at 25°C, pr...
A: The Cp of Solid Biphenyl is 198.39 J/mol/K.Molar mass of Biphenyl = 154 g/mol.Given weight = 0.5260g...
Q: Lewis Structure and Name Silica, dichloromethane, R-carvone, R-limonene, (1R,2R,4R)-dihydrocarveol,...
A: Introduction: Lewis structure represented a proper electronic contribution of the atom in the molecu...
Q: :O:
A:
Q: How many grams of NaOH will be used to prduce 350 grams of water?
A: Given equation is 2 NaOH + H2S -->...
Q: Index of Hydrogen Deficiency (IHD) - What is the IHD for a compound having a molecular formula of C4...
A:
Q: interest are: phenol (pK, = 9.9), and acetic acid (pK, = 4.8). LOH CH3- CH3- он A в D acetate phenol...
A:
Q: Determine the enthalpy of reaction for HCI(g) + NaNO,(s) → HNO_(1) + NaCl(s) 2NACI(s) + H,O(I) → NO(...
A:
Q: . This graph shows a plot of the rate of a reaction versus the con- centration of the reactant. 0.01...
A: It can be seen from the given plot, as the concentration of reactant increases the rate of the react...
Q: n a kinetic experiment a chemist places crystals of iodine in closed reaction vessel. She then intro...
A: Given: Experiment 1: I2 (s) is allowed to react with H2(g). Experiment 2: Iodine is heated to 130℃ a...
Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE.
Write the resonance structure that would result from pushing the electrons as indicated by the curved arrow.
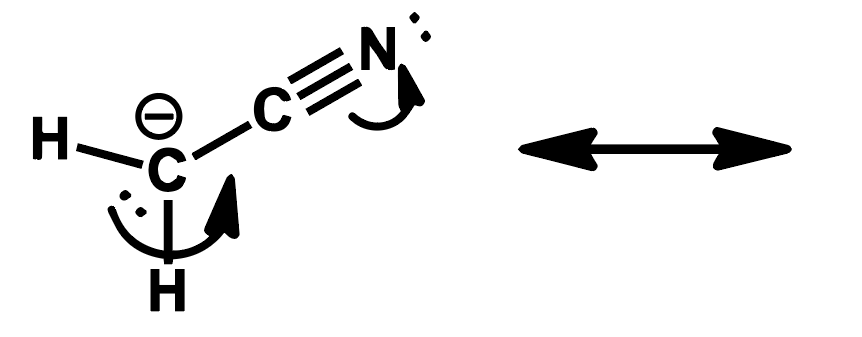
Step by step
Solved in 2 steps with 1 images




