Q: DMF Br ELONA ELOH Нeat 1. LIAIH, 2. H.0
A:
Q: CHA ÖH Pocls, 4 CHe CHs 'N- CHg QCH] CHs - HOl eblora eaffeime eaffeine
A: In most keto-enol tautomerisms, the equilibrium lies by far toward the keto form, indicating that th...
Q: The [OH–] = 1.3 x 10–6 M for a 0.025 M solution of a weak base. Calculate the value of Kb for this w...
A: Kb value for given weak base is 5.2×10^-5.
Q: Arrange the compounds in order of increasing solubility in water (least soluble first).
A:
Q: Which among these islare desirable properties for the type of precipitate required in gravimetric an...
A:
Q: A binary mixture of gases A and B contained 0.515 g of A (100.0 g/mol) at a total pressure of 1825 t...
A: We know that, PV = nRT Given, pressure P = 1825 torr = Volume V = 362.2 ml = 0.3622 L Temperature ...
Q: The degree of dissociation of acids is often expressed in terms of pKa. pka then is Directly measure...
A: We know pKa = -log(Ka) = log(1/Ka) where Ka is acid dissociation constant thus reciprocal log of di...
Q: Explain how the following are soluble in water: a. ethylamine b. diethylamine c. aniline
A:
Q: When aqueous solutions of potassium acetate and hydroiodic acid are mixed, an aqueous solution of po...
A:
Q: 25ºC, AgCl (Ksp = 1.6 x 10-10) is less soluble as compared to PbCl2(Ksp = 2.4 x 10-4) True or false...
A: We know that, solubility of salt depends on Ksp value of salt. Ksp value is directly proportional t...
Q: Potassium metal and chlorine gas combine to form potassium chloride. 5. Aluminum and hydrochloric a...
A:
Q: A piece of unknown metal with a mass of 18.01 g is heated in boiling water to 98.9°C and then droppe...
A:
Q: Can someone explain how to get the following: h(1)-0(2)-c(3) bond angle? also, determine the hybri...
A: Given:
Q: 1. Copy and Answer-Hand written on a yellow pad or bond paper. Balance the redox reaction, determine...
A:
Q: Do you think the indicated pH consistently conforms with the indicated [H,O*] and [OH] concentration...
A: We know that, pH + pOH = Pkw = 14 And pH = -log[H3O+] pOH = -log[OH-] Given, pH = 5 Th...
Q: Silane, SiH4, burns according to the reaction, SiH4 + 2O2 → SiO2 + 2H2O, with ΔH° = –1429 kJ. How mu...
A:
Q: Which of the following compounds would display 4 different signals (two doublets, a singlet, and a t...
A: The NMR spectrum provides the following details about the structure of the molecules: The number of...
Q: Determine t
A: Concept: Katayama Equatiom γ Mρl-ρg23=k' Tc-Y
Q: The following changes will occur when NaF is added to HF = H30*+F" Equilibrium will shift from left ...
A:
Q: BASIC QUESTIONS) CHOICES: TRUE FALSE In collecting gas over water, the height of displaced water i...
A: 1. True 2. True. 3. True 4. False Equilibrium vapour pressure is met when the rate of vapo...
Q: Calculate the volume, in dm³, occupied by 26.4 g of carbon dioxide at r.t.p. 24 a (A, values: C = 12...
A:
Q: After equilibrium is reached in the reaction of 6.30 g H2 with 150. g I2, analysis shows that the 1....
A: An equilibrium constant Keq is ratio of the concentration of product to reactant at equilibrium
Q: 8. (E)-1,2-bis(2,4,6-trinitrophenyl)ethene 9. 3-nitro-5-(trichloromethyl)phenyl benzoate 10. 4-ethyl...
A:
Q: How many liters of H. gas would be produced by the complete reaction of 2.93 g of Al solid at STP ac...
A:
Q: Please show the complete solution (Formula, Given and Conversion of Units) A heat exchanger was ins...
A: Given, rate of exchanger is 0.50 kg/s Molecular weight is 32 and k=1.32 Gas is cooled from 150° to...
Q: Silane, SiH4, burns according to the reaction, SiH4 + 2O2 → SiO2 + 2H2O, with ΔH° = –1429 kJ. How mu...
A: In the question, negative sign of ∆H° means energy is released during the burning of SiH4 You can s...
Q: Decide which element probably forms a compound with hydrogen that has a chemical formula most and le...
A:
Q: What are the advantages of using the KBr in IR spectroscopic sample preparation?
A: This spectroscopy contains infrared range of electromagnetic spectrum and it's wavelength is greater...
Q: Which of the following acid/ conjugate base pairs would function best as a buffer at physiological p...
A: 1- First we should know about physiological pH it's value is 7.40 it's maintain the blood pH because...
Q: The complex below that can show the symptoms of bond isomerism is .... A. [Co(NH3)4Cl2]+ B. Ni(H2O)6...
A:
Q: A buffer consisting of lactic acid and sodium lactate has the pH = 4.30. Given that the concentratio...
A: Consider the given information is as follows; Concentration of lactic acid = 0.12 M C...
Q: A gas cylinder has an internal volume of 2,600L. If the sun heats the gas cylinder from a temperatur...
A:
Q: 12. Consider the following polymers, which will have the lowest T? H CH3 H +CH2-C (CH,-C +CH2-C CH3 ...
A: Ans. 12. Option B polymer has lowest Tg value (-75°c) 13. Option C is polar polymer. 14. A- polypr...
Q: (Ь) (а)
A: Name each alkene using the cis and trans system:
Q: Vinyl alcohol H. Он
A:
Q: What is the role of stationary phase and mobile phase in Chromatography? Please shortly write at you...
A: Concept is based on Chromatography.
Q: Write a summary for preparation of p-Nitroaniline experiment pertaining to three step synthesis, ace...
A:
Q: How is the geometry of a molecule defined and why is the study of molecular geometry important?
A: The geometry of the molecule and it's important is given below.
Q: QUESTION 11 The following are used as primary standards for NaOH EXCEPT O sulfamic acid sodium carbo...
A:
Q: Which is the stronger acid, aspirin or salicylic acid?
A: Acid:- Acid are those species which donates H+ ion . Or Those species ...
Q: QUESTION 27 Which of the following statements describe a colloidal precipitate? O All statements are...
A: A colloid is a mixture in which one substance of microscopically dispersed insoluble particles are s...
Q: the IUPAC name
A:
Q: Cot Na - 3. You are given 250.0 mL of 0.100 M CH;CH;COOH (propionic acid, K = 1.35 x 10). YouCH3CH w...
A: pH of the solution is equal to the negative logarithm of the hydrogen ion concentration. Given data:...
Q: Starting from benzene, show how to prepare the following compound. Provide all necessary reagents an...
A:
Q: chain theory, the complex compound K2[CrCO(CN)5] is an electrolyte by releasing A. 2 K+ ions B. 1 mo...
A: We have to tell which option is correct from given choices of options by telling how given compound ...
Q: Based from the given critical temperature values in the table, rank the following compounds accordin...
A: The critical temperature signifies the force of attraction between the molecules. The higher the cri...
Q: QUESTION 18 Molarity of HCI if titration of 25.00 mL of 0.04010 M Ba(OH)2 required 19.92 mL of the a...
A:
Q: What product would be formed, if the Starting Material SM reacts with hydrochloric acid? why the Sta...
A: The Given starting material will react with both NaOH and HCl because of presence
Q: Try to name the following compounds using the E/Z convention: What is the name of this molecule?
A: Given, molecule
Q: - Give the common names of the following compounds 1. 2. 3. 4. 5.
A: For common names of ethers, just naming both alkyl groups before "ether". For common names of thio e...
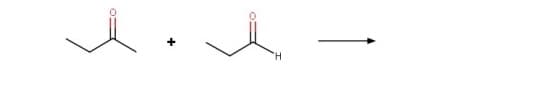
Step by step
Solved in 4 steps with 4 images


