Q: For a saturated solution of Zn(OH)2, calculate the activity of Zn and its effective pH in: a) 0.0237...
A:
Q: 12. Recall that the freezing point is depressed by -1.86 °C per mole of solute per kg of water. Calc...
A:
Q: equilibrium
A: In this question we have to explain why the molar solubility for the pure Ca(OH)2 solution differs f...
Q: Measurements show that the enthalpy of a mixture of gaseous reactants decreases by 278. kJ during a ...
A:
Q: a. What is the pH of a 0.20 M solution of sodium acetate? The Ka for acetic acid is 1.8 x 10⁻⁵? b....
A: We have to calculate the pH of the given solution. a. Given that, the concentration of sodium acetat...
Q: Question 7 The cell notation for the Galvanic cell is A(SYA²*/B2(s)/B¯ The Standard Reduction Potent...
A:
Q: owing from question 3, is it possible to separate aspirin from paracetamol using a liquid-liquid act...
A: We have to tell is it possible to separate aspirin from paracetamol.
Q: a. Add arrows to the boxed structure that show the electron movement in the decarboxylation of malon...
A:
Q: 2. Give the mechanism and products for the reaction below. 1. `MgBr 2. + 3. Н, Н.О
A: RMgX i.e. Grignard reagent that is a good source of R- that carry out nucleophilic addition to alkyl...
Q: 100 mL of water was mixed in 100 mL of 100 Proof alcoholic beverage. What is the percentage by volum...
A: percent by volume one of the concentration unit here we are required to find the percentage by volu...
Q: The equilibrium constant Kc for the reaction fructose-1,6-diphosphate = glyceraldehyde-3-phosphate +...
A: Solution -
Q: 3a) What is the AƐ value for the oxidation of cytochrome c by the Cua redox center in Complex IV whe...
A: Answer: Given data: the ratio of cyt C (Fe2+) / cyt C (Fe3+) = 20 and ratio of CuA (Cu2+) / CuA (Cu...
Q: What is the solubility-product expression for La(IO3)3 (s)? a. Ksp = [La3+] [IO3-]1/3 b. Ksp = [La3+...
A:
Q: LIMIT TEST (IMPURITIES - BIOLOGICAL) Instructions: Note the color change of each of the samples by ...
A: A question based on general chemistry that is to be accomplished.
Q: Water Temperature 8 degrees Time (sec) to 60 cm Time (sec) to 40 cm Time (sec) to 20 cm Skittle Colo...
A: A question based on matter that is to be accomplished.
Q: What is the change in molar conformational entropy when the same random coil is stretched from fully...
A: Solution -
Q: 7. A 6.0 grams of NaOH is dissolved in distilled water to make 1.60 - liters of solution. What is th...
A:
Q: Write out the molecular, ionic, and net ionic equations for the neutralization reaction between sulf...
A: KOH(aq) + H2SO4(aq)
Q: 2. The concentration of NH4CI solution (aqueous solution) is 33.5% by mass. If the mass of solution ...
A:
Q: 4. Following from question 3, is it possible to separate aspirin from paracetamol using a liquid-liq...
A: Aspirin (2-acetoxy benzoic acid) and paracetamol (N-(4-hydoxyphenyl)acetamide) can be seprated by th...
Q: If the initial concentration of (CH3)gCBr (aq) is 0.100 M and the half-life of this reaction is 1.45...
A:
Q: A type of solution that contains the maximum amount of solute that is capable of being dissolved is ...
A: A type of solution that contains the maximum amount of solute that is capable of being dissolved is ...
Q: Question 20 Draw the energy level diagrams and electron configurations (long form) for the following...
A: Let us draw the energy level diagrams and then write their electronic configuration.
Q: 1. Ideally, all atoms have the same number of protons and electrons, therefore all atoms are consi...
A: Given, Ideally, all atoms have the same number of protons and electrons, therefore all atoms are con...
Q: The amount of iron in a meteorite is determined by a redox titration using KMnO4 as the titrant. A 2...
A:
Q: The acid ionization constant for Pb(H20)62+(aq) is 6.3×10-7. Calculate the pH of a 0.0307 M solution...
A:
Q: Energy > The image below shows two configurations of a system of 4 particles. Energy levels increase...
A: We know that Entropy S = Kb ln W Where W is the no of arrangement or no of microstate possible . S...
Q: what is the chemical formula for copper(I) chloride and for gold (III) cyanide?
A:
Q: Which of the following characterizations for cyclic ethers is INCORRECT ? They are always aromat...
A:
Q: Which of the following are equivalent to 2,500 ppm Cu2+? (There may be more than one answer) MW...
A: Given data 2500 ppm equal to
Q: How fast is the rate of effusion of CH, compared to that of SO,? 5 2 O O O
A:
Q: Lalisa and Labmi were tasked to design a new and efficient method of producing black inks. To determ...
A: Retardation or retention factor (Rf) value is the ratio of distance traveled by the analyte to that ...
Q: At 3000 K, the partial pressures of CO2, CÓ and O2 are 0.6, 0.4 and 0.2 atm, respectively. Calculate...
A: Temperature = 3000 K Reaction :- 2CO2(g) <--------> 2CO(g) + O2(g)
Q: Consider the reaction N2O4(gas) = 2NO2(gas). At 100 C, the Question equilibrium pressures in a 3.50 ...
A: Dalton's partial pressure law is a gas law that says that the overall pressure exerted by a gas mixt...
Q: Coordination complexes
A: In this question we have to explain that the following statement is true or false, coordination comp...
Q: 4. What mass of NH3 is produced from 12 grams of H2?
A: Here we have to determine the mass of NH3 produced when 12 grams of H2 reacts with excess amount of ...
Q: -200 2 Fe + 0, →2 F 2C+02-2 CO -300 -400 -500 -600 -700 4/3 Al + 0, → 2/3 Al,0, -1000 -800 -900 -110...
A:
Q: he volume (in
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
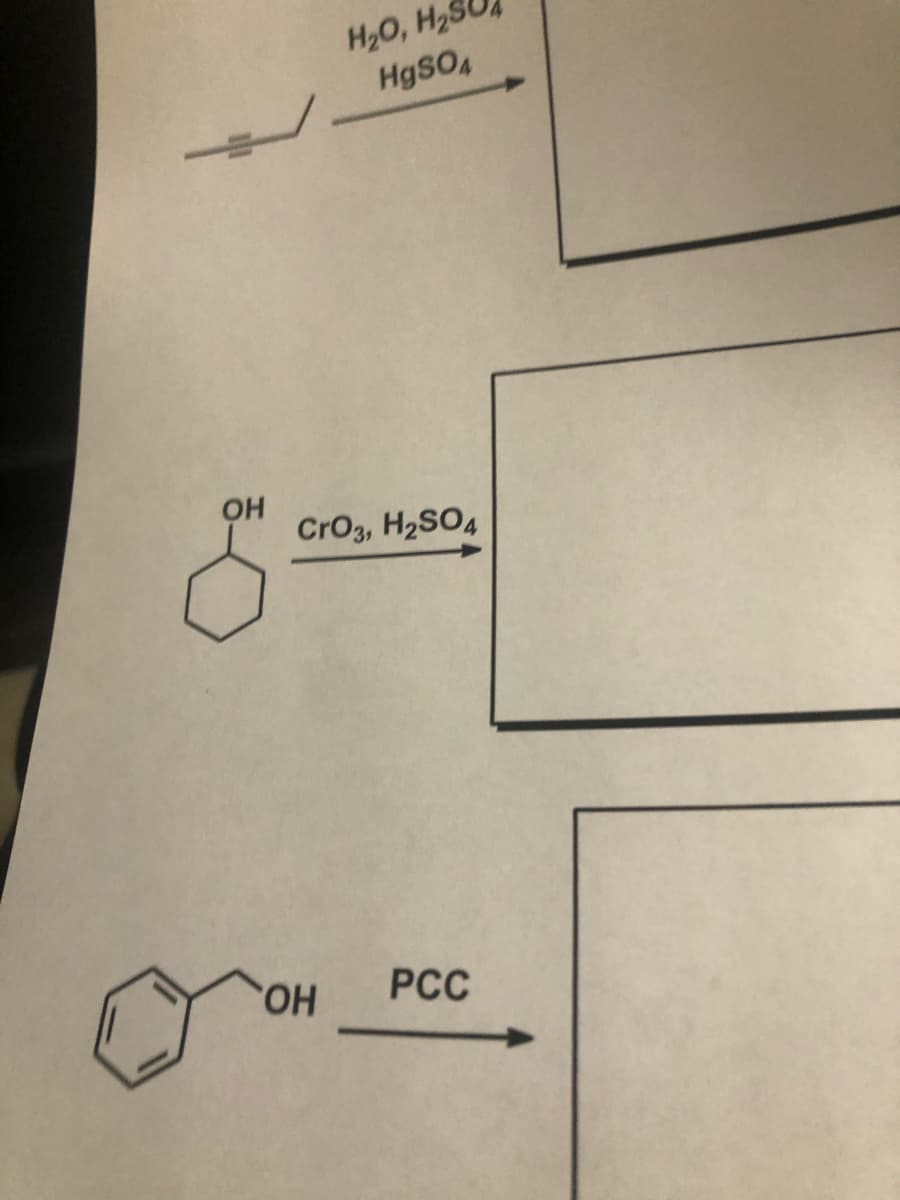
Q: 3. Predict the major product from the following reaction sequence: (Write out complete equations sho...
A:
Q: A chemist determines by measurements that 0.0500 moles of oxygen gas participate in a chemical react...
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: What is the pH of a 0.20 M solution of sodium acetate? The Ka for acetic acid is 1.8 x 10⁻⁵?
A:
Q: KOGRESS CHECK NO, 3 Rank the following sets of substituents in order of priority according to the Ca...
A:
Q: How many isomeric ethers exist that contain C2 and C4 alkyl groups? three five four no corre...
A: Ethers have two hydrocarbon groups bonded to an oxygen.
Q: Assignment #3. The temperature of pure water at equilibrium vapor pressure of 36.2 torr is 45°C. Whe...
A:
Q: Which one has the Highest vapor pressure? Lowest IMF? Why? a. Alcohol b. Sea water c. Water d. Mercu...
A: 1) Highest vapour pressure - Vapor pressure is the pressure caused by the evaporation of liquids. Th...
Q: 40 The ratio of an observed measurement of colligative property for a solute in a solution to the va...
A:
Q: Why are DOC concentrations lower in the deep Pacific than in the deep Atlantic? O Much of the ocean'...
A: The very low DOC are due to microbial decomposition in excess of new carbon introduced by sinking p...
Q: Submit A system expands from a volume of 1.00L to 2.00 L against a constant external pressure of 1.0...
A:
Q: PURITY TEST Instructions: Note the REFRACTIVE INDECES of each of the samples and solve for their % p...
A:
Q: A solution was made by dissolving 3.75 g of a solute in 108.7 g of acetone . The solution boiled at ...
A: Given data,Mass of solute=3.75gMass of acetone=108.7gBoiling point of solution=56.58oCBoiling point ...
Step by step
Solved in 2 steps with 3 images




