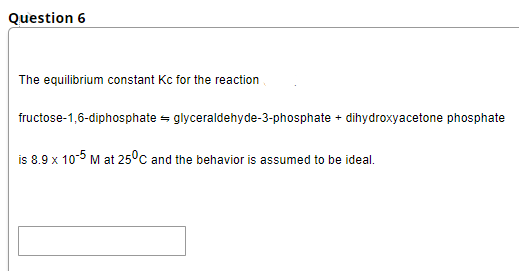
The equilibrium constant Kc for the reaction fructose-1,6-diphosphate = glyceraldehyde-3-phosphate + dihydroxyacetone phosphate is 8.9 x 10-5 M at 25°c and the behavior is assumed to be ideal.
Q: OH
A: According to the rules of IUPAC nomenclature longest carbon chain is numbered at first.Carbon contai...
Q: Which one has the lowest vapor pressure and why? a. Alcohol b. Sea water c. Water d. Mercury e. Dead...
A: Which one has the lowest vapour pressure and what is the behind of it.the answer and explaination is...
Q: e stable or le
A:
Q: Predict the 'H NMR spectrum for the following compound, giving multiplicities and estimates of the c...
A: Here we have to find out the multiplicity ,chemical shift and coupling constants of the respective h...
Q: A chemist measures the amount of fluorine gas produced during an experiment. He finds that 495. g of...
A:
Q: 100 mL of water was mixed in 100 mL of 100 Proof alcoholic beverage. What is the percentage by volum...
A: percent by volume one of the concentration unit here we are required to find the percentage by volu...
Q: Measurements show that the energy of a mixture of gaseous reactants increases by 372. kJ during a ce...
A: The energy change of the given reaction is = 372. kJ The work done on the gas mixture is = -63. kJ C...
Q: Which of the following statements concerning physical properties of ethers is INCORRECT? Ethers are...
A: Ethers are the class of organic compound which have the general formula R-O-R' Where an oxygen atom...
Q: concentration
A:
Q: What is the change in molar conformational entropy when the same random coil is stretched from fully...
A: Solution -
Q: Do read carefully and answer accordingly Chlorophyll a (a plant pigment) has the following molar ex...
A:
Q: The reaction of 5.07g N2 with 0.722g h2 produces 1.27g NH3. What is the percent yield of this reacti...
A: Yield is calculated by ratio of Autual yield to theoretical yield.
Q: Provide the answers with proper number of significant figures in the following operations. pls provi...
A: We know the default base of logarithm is 10. To find the antilog we need to divide the given number...
Q: What is the wavelength (angstroms) of a photon that has an energy of J? 4.38x10-18
A:
Q: The annual production of sulfur dioxide from burning coal and fossil fuels, auto exhaust, and other ...
A: Sulfur reacts with oxygen to form sulfur dioxide. The equation for the balanced chemical reaction is...
Q: a) Identify the type of the followings surfactants. CH3 R-S-o M R-N-CH2CH2-C-o CH3 (ii) (i) b) Which...
A:
Q: End of Chapter Quetions References) This question has multiple parts. Work all the parts to get the ...
A: Mass of KNO3, Potassium Nitrate = 557 g Reaction:- 4KCl + 4HNO3 + O2 -----> 4KNO3 + 2Cl2 + 2H2O
Q: In nomenclature how do you know when you need to add ide, ate, ite, hypo, ous , ic, per, bi, di
A: since you have posted multiple questions, as per guidelines we will solve the first question for you...
Q: Estimate the biomass yield of proponic acid (CH3CH2COOH) from both the stoichiometry and bioenergeti...
A: Solution Given that the biomass yield of proponic acid (CH3CH2COOH...
Q: ᴀ ꜱᴛᴇᴇʟ ᴛʀᴀɪɴ ʀᴀɪʟ ɪꜱ 400 ᴍᴇᴛᴇʀꜱ ʟᴏɴɢ, ɪɴ ᴍᴀʀᴄʜ ɪᴛ ɪꜱ -30 ᴅᴇɢʀᴇᴇ ᴄ ᴀɴᴅ 40 ᴅᴇɢʀᴇᴇ ᴄ ɪɴ ᴊᴜʟʏ. ᴡʜᴀᴛ ɪꜱ ...
A: Detailed solution is provided.
Q: For a saturated solution of Zn(OH)2, calculate the activity of Zn and its effective pH in: a) 0.0237...
A:
Q: A compound containing 5.9265 % H and 94.0735 % O has a molar mass of 34.01468 g/mol. Determine the e...
A: Given that a compound contains 5.9625% H and 94.0735% O
Q: Activity 2 Direction: Complete the table by writing all types of intermolecular forces (IMF) that wi...
A: Intramolecular forces These are the forces that hold atoms together within a molecule. Intermolecul...
Q: consider the reaction 2A + 2B + C -›D + 3E A plot of Ln (pressure in torr) versus 1/ T (in kelvin) p...
A:
Q: How many pounds will 1 cubic foot of cork weigh if one cubic centimeters weighs 0.25 grams?
A: Given, Note: 1 cubic foot = 1 ft3 1 cubic centimetre = 1 cm3
Q: 13)The frequency of electromagnetic radiation required to promote an electron from n = 2 to n = 4 in...
A:
Q: Examine the molecule below. Is this molecule polar? Justify your answer. F .C- Fー
A: Polar covalent bond: The covalent bond is formed by the sharing of electrons between the atoms. If t...
Q: What is the solubility-product expression for La(IO3)3 (s)? a. Ksp = [La3+] [IO3-]1/3 b. Ksp = [La3+...
A:
Q: 1kg of Hydrogen molecules H2contains 33.33 kWh of energy. 1 litter of liquid hydrogen weighs 70.99 g...
A: A question based on energy that is to be accomplished.
Q: Which should be most soluble in water? А) H HH H H C C-C-C–H butane нннн B) 0=C-O-H formic acid C) H...
A:
Q: A system expands from a volume of 1.00 L to 2.00 L against a constant external pressure of 1.00 atm....
A:
Q: What is the application of Sodium Chloride in chromatic separation?
A: Answer - According to the question - NaCl dissociates in Na+ and Cl- in chromatic separation solutio...
Q: What is the frequency of light (s^-1) that has a wavelength of 3.12 x 10^-13cm?
A: Wavelength of light, (λ) = 3.12 × 10-13 cm
Q: degree of unsaturation
A:
Q: Procedure: Using different references or resources (Books, e-books, journals and internet) Ident...
A:
Q: 1. Using the Williamson synthesis show how you would prepare 1-ethoxycyclohexane from 1-cyclohexanol...
A: Given, Using the Williamson synthesis show how you would prepare 1-ethoxycyclohexane from 1-cyclohe...
Q: 18. Which of the indicated bonds in each pair of compounds is shorter? CH3 a. CH3-C=CH or C=CH2 c. C...
A: With increasing in effective nuclear charge of one atom on the other bonded atom, i.e. with increasi...
Q: For each pair of molecules below, choose the one you would predict to be more stable and briefly exp...
A:
Q: Which of the following are extrinsic properties? being made of paper O being to the left of a flagpo...
A: An extrinsic property is not essential or inherent to the subject that is being characterized.
Q: Lalisa and Labmi were tasked to design a new and efficient method of producing black inks. To determ...
A: Retardation or retention factor (Rf) value is the ratio of distance traveled by the analyte to that ...
Q: What solvent would you use to differentiate compound A from compound B? Compound A Compound B CH3CCH...
A: The small aldehydes and ketones are freely soluble in water but solubility falls with chain length. ...
Q: 1kg of Hydrogen molecules H2contains 33.33 kWh of energy. 1 litter of liquid hydrogen weighs 70.99 g...
A:
Q: he molar mass of nicotine is 162.1 g/mol. It contains 74.0 % carbon, 8.7 % ydrogen, and 17.3 % nitro...
A: Given that compound contains 74% Carbon 8.7% Hydrogen and 17.3% Nitrogen.
Q: 3. Fill in the boxes for the 2 products below AI H
A:
Q: 6 A sample of lead glass is prepared by melting together 20.0g silica , SiO2 and 80.0g of lead (II) ...
A: we are required to find the mole fraction of silica in the glass which is prepared by mixing 20 g of...
Q: (Q97) What is the value of the equilibrium constant (Keg) for the following hypothetical reaction: 2...
A:
Q: 5. For each of the following pairs of substances, identify the conjugate bases for each one and indi...
A: conjugate base is the substance formed when an acid loses a hydrogen ion. Considered a base because ...
Q: 1. An air mixture contains 0.00015 mol CO in 10 mol of air. What is the CO concentration in ppmv? As...
A: Given number of moles of CO = 1.5 × 10-4 molesTotal moles of the mixture = 10 mole
Q: What CrO42- concentration is required to initiate precipitation of Ag2CrO4 from a solution that is 0...
A: Solubility can be defined as maximum amount of solute that can be dissolved in solvent so as to obta...
Q: chromate/dichromate system is prepared (Refer to the picture) Predict the shift in direction of t...
A: Le chateliers principle - When stress is applied to a system under equilibrium such as change in co...
Step by step
Solved in 2 steps with 1 images

- Which of the following best describes the structure below A. Dextrorotatory B. Ketose C. Hexose D. Levarotatory E. Pentose F. Aldose G. Terosefor the equilibrium PH3BCI3(s)=PH3(g)+BCI3(g) Kp=0.052 at 60degreesCelsius calaculate KcWhich of the following combination in each pair is likely to produce more Maillard browning when heated at 95 °C for 4 hours? Explain the chemical basis for your choice. Sucrose + glycine, pH 7.0 vs. glucose + glycine, pH 7.0 Maltose with a dextrose equivalency (DE) of 20 + glycine, pH 9.0 vs. maltose + glycine, pH 9.0 Lactose + glycine, pH 8.0 vs. lactose + glycine, pH 4.0
- Write down the reactions: D-Glucose + [Ag(NH3)2]+ →Which of the following combination in each pair is likely to produce more Maillard browning when heated at 95 °C for 4 hours? Explain the chemical basis for your choice. A.Sucrose + glycine, pH 7.0 vs. glucose + glycine, pH 7.0 B.Maltose with a dextrose equivalency (DE) of 20 + glycine, pH 9.0 vs. maltose + glycine, pH 9.0 C.Lactose + glycine, pH 8.0 vs. lactose + glycine, pH 4.0 Please answer very soon will give rating surely All questions answers neededOne step in glycolysis is the conversion of fructose-6-phosphate to fructose-1,6-bisphosphate. The direct reaction of fructose-6-phosphate with inorganic phosphate fructose-6-phosphate + Pi ⇌ fructose-1,6-bisphosphate + H₂O is very unfavorable, with ∆G'°= 16.3 kJ/mol and K'eq = 1.39×10⁻³. However, in glycolysis the reaction is coupled to ATP hydrolysis: fructose-6-phosphate + ATP ⇌ fructose-1,6-phosphate + ADP and the reaction if favorable. What is the equilibrium [fructose-1,6-bisphosphate]/[fructose-6-phosphate] ratio in the coupled reaction when [ATP] = [ADP]? For ATP hydrolysis ∆G'°= -30.5 kJ/mol at 25°C.
- The carbonyl group in d-galactose may be isomerized from C1 to C2 by brief treatmentwith dilute base (by the enediol rearrangement, Section 23-7). The product is the C4epimer of fructose. Draw the furanose structure of the product.In one of the steps in this pathway fructose 1,6-biphosphate (F1,6BP) is converted to gylceraldehyde-3-phosphate (G-3-p) and dihydroxyacetone phosphate (DHAP). This reaction is catalyzed by the enzyme aldolase. For this reaction at 25 celcuius and Ph7 we have: Keq= 10-4M and Delta G = +5456 cal/mol Calculate the following: The concentration of F1, 6BP, DHAP and G-3-P at equilibrium when the initial F1,6BP is (A) 1M, (b) 10-2 M, (c) 2 X 10*4 M and (d) 10-5 M. I been trying to solve this problem but I dont even know where to begin with. Atleast help me with problem A, I would be able to guide myself through.Homework on: Carbohydrates V-11. Write Haworth projection formulas for such substances: 1.1 α-D-glucopyranose;1.2 β-D-fructofuranose;1.3 lactose.1.4 In each formula show the hemi-acetal hydroxyl.2. Write: why saccharose is a non-reducing disaccharide. 3. Write the equations of reactions:3.1 glucose +Ag2O(NH3)→;3.2 glucose +H2→;3.3 α-D-glucopyranose +CH3OH(HCl)→;3.4 β-D-glucopyranose +(CH3CO)2O→;3.5 maltose hydrolysis;3.6 glucaric acid formation from glucose.4. Describe the signs of reactions:4.1 silver mirror reaction for glucose;4.2 Selivanov's reactions for the fructose;4.3 glucose reactions with Cu(OH)2 without heating.
- Regarding 4-O- (α-D-psychofuranosyl) -β-D-allopyranose. Please indicate the RIGHT alternative: (a) The disaccharide reacts with CH3OH in an acid medium to form a glycoside that cannot be oxidized with HNO3. (b) It is a reducing disaccharide only in basic medium. (c) In the structure there is only one glycosidic bond that is of the type β 1-O-4 ' (d) The hydrolysis products of this disaccharide do not show mutarrotation. (e) The disaccharide structure contains two six-membered rings.The disaccharide lactulose consists of a d-galactopyranose subunit and a d-fructofuranose subunit joined by a β-1,4-glycosidic linkage. After treatmentof lactulose with 1. excess CH3I/Ag2O, 2. HCl/H2O, the d-galactopyranose subunit was found to have one nonmethylated OH group, whereas thed-fructofuranose subunit had two. Draw the structure of α-lactulose.Write the chemical equations of the following reactions(a) Glucose in heated with conc. H2SO4.(b) Sodium nitrate is heated with conc. H2SO4.

