Q: The enthalpy change of a reaction Hrxn equals heat q, if the reaction is performed ... at constant…
A: Introduction The enthalpy of reaction, or change in enthalpy (denoted ΔH), is a measurement of the…
Q: How many alkyl groups would be equatorial in the most stable conformation of the cyclohexane below?…
A: we have to determine the number of alkyl groups at the equatorial position of stable conformation of…
Q: What is the vapor pressure of a pure sample of water at 37∘C, if a 0.50Msolution of BaCl2 has a…
A: BaCl2 is a non-volatile solute. Its presence in the solution causes the lowering of vapor pressure…
Q: What is the energy change when the temperature of 11.2 grams of solid diamond is decreased from 39.3…
A: Formula used :Q = m * c * ΔT where: Q is the heat transfer (in J)m is the mass of the solid diamond…
Q: 1. When treated under SN1 condtions, the two chloridees below give the same pair of alcohols in the…
A:
Q: Q2: For the following molecules, determine the number of IR active C-O stretching vibration: OFI a)…
A: In group theory, a representation is a set of matrices corresponding to symmetry operations such…
Q: 1.1 For the following reaction carried out at constant temperature and pressure, what is true about…
A: Given : gaseous reaction
Q: Standardization of Sodium Hydroxide (NaOH) Solution Mass of KHP used (g) Initial buret reading (mL)…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Vapor pressure (mm Hg) 900 800 700 600 500 400 300 200 100 0. Carbon disulfide 10 20 30 °C Methano…
A: Answer: variation of vapor pressure with temperature is given in the mentioned diagrams and we have…
Q: What is the identity of the uknown metal?
A: Compare calculated the density of unknown metal with the given densities of metals. given: density…
Q: The reaction A→ B has a standard state free energy of -15 kJ/mol and a cellular free energy of 0…
A: Introduction Spontaneous reaction in chemistry is a process in which a chemical reaction proceeds…
Q: 2) Consider the following reaction and the data below: 2NO +2H₂ → N₂ + 2H₂O [H₂], M 0.100 0.185…
A: a) we have to find the order of each reactant
Q: 4) Use data for AG°f to calculate equilibrium constants for each reaction at 25C: J ·R = (31 Mal R…
A:
Q: A mass spectrum of an unknown compound shows an [M]+ peak with an [M+2]* peak of similar height.…
A: Given that The mass spectrum of an unknown compound shows an [M]+ peak with an [M+2] peak of…
Q: 2 3 3 17 S Total unique 'H Chemical Shifts for each compound shown above. 4 7 3
A:
Q: If a buffer solution is 0.210 M in a weak acid (K₁ = 3.0 x 10-5) and 0.480 M in its conjugate base,…
A: For buffer solution, Henderson equation is pH = pKa + log [salt][acid]
Q: Calculate the mass percent of solute in the following solutions: 1.) 6.50 grams of NaCl dissolved in…
A: It is based on the concept of mass percentage of solute . Here we are require to mass percent of…
Q: 8. Below are 4 molecules and 4 NMR spectra. Match the molecule to its respective NMR spectrum. 3 2…
A: The H on the carbon atom adjacent to electronegative atom O will be highly deshielded. Their…
Q: The freezing point of pure benzene (CoHo) is 5.49 °C. The freezing point of a solution made using…
A: A decrease in the freezing point is a colligative property. The decrease in the freezing point is…
Q: 0.What will be the transport number of ions in an aqua solution of 0.050 M HCI using the moving…
A: Transport number is a concept in electrochemistry that refers to the fraction of the total current…
Q: What is the multiplicity expected in the hydrogen NMR spectrum for the hydrogen atoms marked by a…
A: Given : structure of molecule Tip : multiplicity in NMR = n+1 n = adjacent hydrogen
Q: Determine whether each of the amino acids is polar, nonpolar, positively charged, or negatively…
A:
Q: What differentiates the Group 3A Cations from the Group 3B cations How do we prevent the…
A: Qualitative analysis is a branch of analytical chemistry that is concerned with the identification…
Q: Under standard biochemical conditions, which ONE of the following relationships must be TRUE by…
A: Under the given set of conditions, a chemical reaction will be spontaneous if the change in the…
Q: Determine the concentration of an HBr solution if a 50.00 mL aliquot of the solution yields 0.5745 g…
A: Given , mass of AgBr added = 0.5745 g Ksp(AgBr) = 5.0×10-13
Q: What would be the approximate H-C-H bond angle in CH3OH? (In other words, what is the approximate…
A:
Q: 5. In the lowest energy conformation of the compound below, how many alkyl substituents are axial…
A:
Q: Nitroglycerin, a very unstable compound (molecular structure shown below), is a liquid at room…
A: Answer: In this question by using the stoichiometric correlation we have to find out the total moles…
Q: A volume of 500.0 mL of 0.130 M NaOH is added to 605 mL of 0.200 M weak acid (Ka = 3.87 × 10¯). What…
A: Given -> Volume of NaOH= V1 = 500 ml Molarity of NaOH = M1 = 0.130 M Volume of weak acid = V2 =…
Q: The experimentally measured freezing point of a 1.20 m aqueous solution of MgCl2 is -6.03°C. The…
A:
Q: Oxidation and reduction reactions are symbiotic (one can't exist without the other). Cite evidence…
A: Solution - This is true that oxidation and reduction occurs simultaneously. When an atom looses…
Q: The net electric force acting on each of the three objects below is sketched as a green arrow. For…
A:
Q: When a solution is made... both the enthalpy (AHsoln) and the entropy (AS soln) of mixing are always…
A: The enthalpy of solution, also known as the heat of solution, is the amount of heat absorbed or…
Q: Please provide an equation for the following nuclear process: 231 Pa decays via ß-emission. 91
A: This question is related to nuclear chemistry. There are mainly three types of radioactive decay-…
Q: A student prepares a reaction in which hexane is converted to 1,3-hexadiene. In this reaction, what…
A: Please find your solution below : Oxidation is a type of chemical reaction in which the loss of…
Q: Provide a name or a Structure
A: Given: name and structure of molecule Tip : identify parent group
Q: 5. Based on your knowledge of thermodynamics and kinetics, clearly provide the predicted outcome of…
A: This problem is based on general organic chemistry reaction mechanism and is a part of hydrocarbon .…
Q: 4. The mass of the average boysenberry is 0.28 ounces. The average Tesla weighs 4900 lbs. If one…
A: Given: mass of boysenberry = 0.28 ounces = 0.28 x 28.3495 g = 7.938 g mass of Tesla = 4900 lbs =…
Q: 4)√ for the units) for the reaction 2A + 2B + 2C products. Show all work (shortcuts are allowed, but…
A:
Q: 1. pH=7.31 PCO2-51 HCO3=27 What is the imbalance and is there compensation (if so, what type)? 2.…
A:
Q: Which equation is used to determine the amount of time required for the initial concentration rate…
A:
Q: A flask contains a mixture of two gases: NO2 and Ne. If the flask contains 0.518 mol of NO2and 16.7…
A:
Q: 14. How is the reactivity of isolated C=C double bonds different from the double bonds in benzene?…
A: We know in presence of light free radical mechanism will proceed and allylic free radical is more…
Q: For the following reaction, 15.5 grams of phosphorus (P4) are allowed to react with 41.6 grams of…
A:
Q: The following Lewis diagram represents the valence electron configuration of a main-group element.…
A: Given : main group element which has 1 valence electron.
Q: Coffee is an acidic beverage with a typical pH = 4.85, while milk has a pH = 6.80. Assuming no…
A: Answer: To solve this question we have to calculate the number of moles of H+ ions in both coffee…
Q: 3. Mechanisms and Rate Laws: What is the rate law for the overall reaction based on the proposed…
A: Answer: In the chemical reaction that takes place in more than one step, slowest step of the…
Q: 2. Identify any missing formal charges on the following structures. H-C- -0-CH₂-CH₂ ww :0: H₂C-S-CH₂…
A: formal charge = number of valence electrons - bonding electrons2 - number of non bonding valence…
Q: 3) Predict the products of the following reactions: a) Y b) c) Br Mg Ether Mg Ether (N) H₂O 1. 2x…
A: Alkyl magnesium bromide is known as a Grignard reagent. The Grignard reagent is a very useful…
Q: The following thermochemical equation is for the reaction of NH4NO3 (s) to form N₂O(g) and NH4NO3…
A:
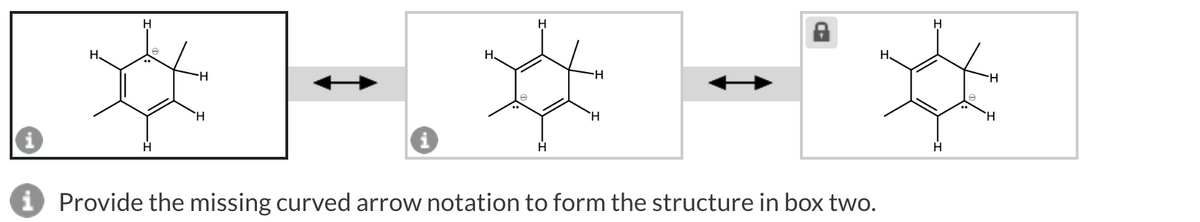
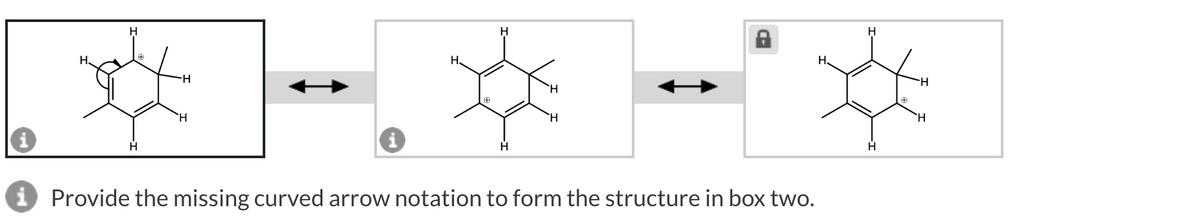
Electrons move from electron rich center towards electron deficient center. Here arrow notation is from electron rich to electron deficient center occurs.
Step by step
Solved in 2 steps with 1 images

- draw all the molecule separately in chemdraw like in step 1 draw one compound , in step 2 draw another , for the last compound draw structure and arrow and written part below and above itWhats the name of step and 1 and drar mechanksm arrows even showing electrons on oxygen atomsWhich line (A or B) corresponds to the energy change for an ortho/para substitution?
- please answer quickly and correctly please round to 3 sig figsThe molecule in the accompanying figure is ____, and has ____. A. unsaturated; no carbon-carbon double bonds B. saturated; a carbon-carbon double bond C. unsaturated; a carbon-carbon double bond D. saturated; no carbon-carbon double bondsPlease write the missing reagent(s) over the arrow after analyzing the functional group(s) in reactants and products and please answer questions A through E
- Complete the reaction below. Circle and identify all the functional groups and linkages in reactants and products. Also identify the type of biochemical reactionCan you help draw the curved arrow for these structures?Draw the orbital interaction that illustrates the hyperconjugation that takes place in propyne.

