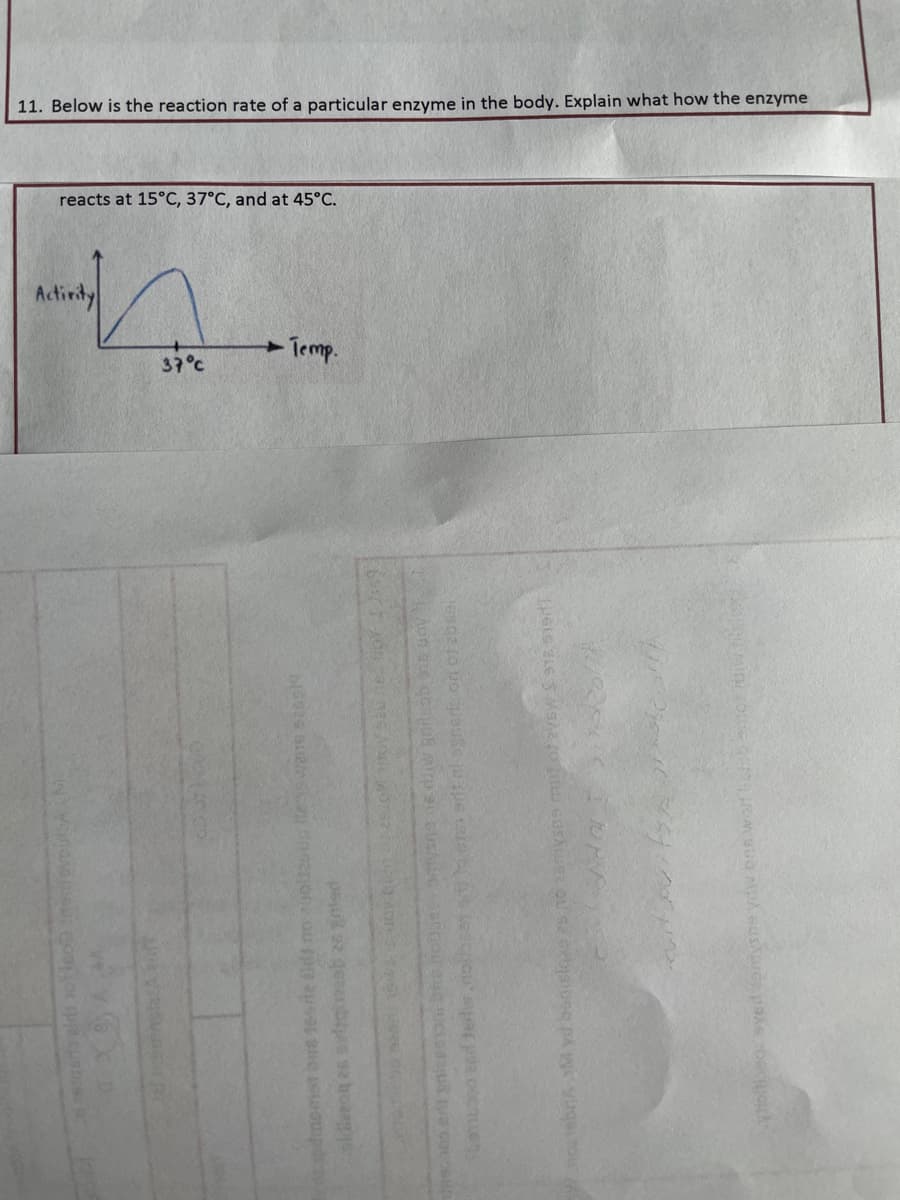
11. Below is the reaction rate of a particular enzyme in the body. Explain what how the enzyme reacts at 15°C, 37°C, and at 45°C. Activity 37°C -Temp.
11. Below is the reaction rate of a particular enzyme in the body. Explain what how the enzyme reacts at 15°C, 37°C, and at 45°C. Activity 37°C -Temp.
Chemistry for Engineering Students
3rd Edition
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter1: Introduction To Chemistry
Section: Chapter Questions
Problem 1.77PAE
Related questions
Question
Transcribed Image Text:sedmomist ons 1
sklizenq es svirginab 25 gated
ADD CSE FU
camboo and Jedw.
on su
bilibeo sved
o era sit nkognerio on of zose,
habn M yd banisique es no sumyse ml of 2vBW $918 519HT
VROUE INAY
37°C
・Temp.
Activity
V
reacts at 15°C, 37°C, and at 45°C.
11. Below is the reaction rate of a particular enzyme in the body. Explain what how the enzyme
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning