Q: + II *00 Question 5 Metal Specific Heat (J/g°C) Calcium 0.643 Iron 0.449 Silver 0.235 Gold 0.129 In…
A: Given, For Silver (Ag): mass of Ag = 31.0 g Initial temperature of Ag (Ti) = 200°C Final temperature…
Q: Substance "C" is identified to have a specific heat capacity of 0.452 J/(g.°C). Which substances…
A: As, q = mC∆T So, C = q/m∆T For given value of heat and mass , Specific heat capacity (C) would be…
Q: * 0∞ Consider the following data on some weak acids and weak bases: K. formula formula name 4. -4 -5…
A: As we know, Acidic salt(Strong acid-weak base type) has pH less than 7. Basic salt(Strong…
Q: What is the heat of combustion of gasoline if 4.00g of gasoline are burned in a calorimeter to raise…
A: As gasoline is combusted to heat water , so we would first calculate amount of heat required to heat…
Q: 2. Draw a product that would form when 5-(propylamino)pentanoic acid is treated with acid catalyst.
A: In this reaction amide is formed... In below reaction protonation of O is occur due to…
Q: Write the IUPAC name for the compound below. Be sure to use correct punctuation. CH,CH3…
A: Steps involved: Find the parent chain: Parent chain is the longest carbon chain with alkene Label…
Q: Will a precipitate form when 20.0 mL of 1.8x10-3 M Pb(NO3)2 is added to 30.0 mL of 5.0x10-4 M…
A: Given Data :
Q: True or false and why is false 1 There are 2 major classes of keratins: alpha keratins (found in…
A: 1) True: alpha keratin found in most mammals , claw , hairs nails etc , while beta keratin found in…
Q: Consider the galvanic cell based on the following half-reactions: Zn2+ + 2e → Zn ɛ° = -0.76 V Cd²+ +…
A: Here the standard reduction potential of Cd2+ is higher than standard reduction potential of Zn2+.…
Q: Draw the SN2 mechanism.
A:
Q: Rank the following from the most to the least stable. II II IV а. III > II > IV > I b. I> IV > II >…
A: Stability of alkene is decided by number of alpha hydrogen atom i.e number of hydrogen atoms present…
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of sodium hydroxide…
A: Please find your solution below : Ionic equation is an equation in which the ionic compounds in ghe…
Q: What is the difference between and element and a compound?
A: element is a a pure substance which cannot be decomposed in to a simple substance compounsd is…
Q: Volume (L) 100 80 60 Created by E. Lee for Virtual 40 20 100 200 300 400 500 600 700 800 900…
A:
Q: Given a pressure-composition diagram of b and w what is the portion of the plot describe below? (use…
A: We are given a pressure-composition phase diagram of mixture of compound w and b . In phase diagram…
Q: [Review Topics) [References] Use the References to access important values if needed for this…
A:
Q: What is the function of the warm-water-in in a distillation set-up?
A: Distillation is the process of using heating and cooling to separate and purify the components of a…
Q: Consider a hypothetical diprotic acid (H2A) with pKa1 = 7.301 and pKa2 = 10.00. A 40.0-mL sample of…
A: GIVEN:- Consider a hypothetical diprotic acid (H2A) with pKa1 = 7.301 and pKa2 = 10.00. A40.0-mL…
Q: Be sure to have your electromagnetic spectrum readily available (Chapter 11 in text) A carbon-oxygen…
A:
Q: Phosphorus (P4) and oxygen react to produce diphosphorus pentoxide. If 196.23 g of diphosphorus…
A:
Q: After a radioactive decay that emits gamma radiation, the new resultant isotope has _____ mass vs.…
A: In γ -decay the nucleus changes its state from high to low energy through emission of…
Q: The molecular formula of rimantadine is C12H21N. How many grams of hydrogen are in 36.01 grams of…
A:
Q: For each chemical reaction listed in the first column of the table below, predict the sign (positive…
A:
Q: Calculate the pH of each of the following solutions from the information given.
A:
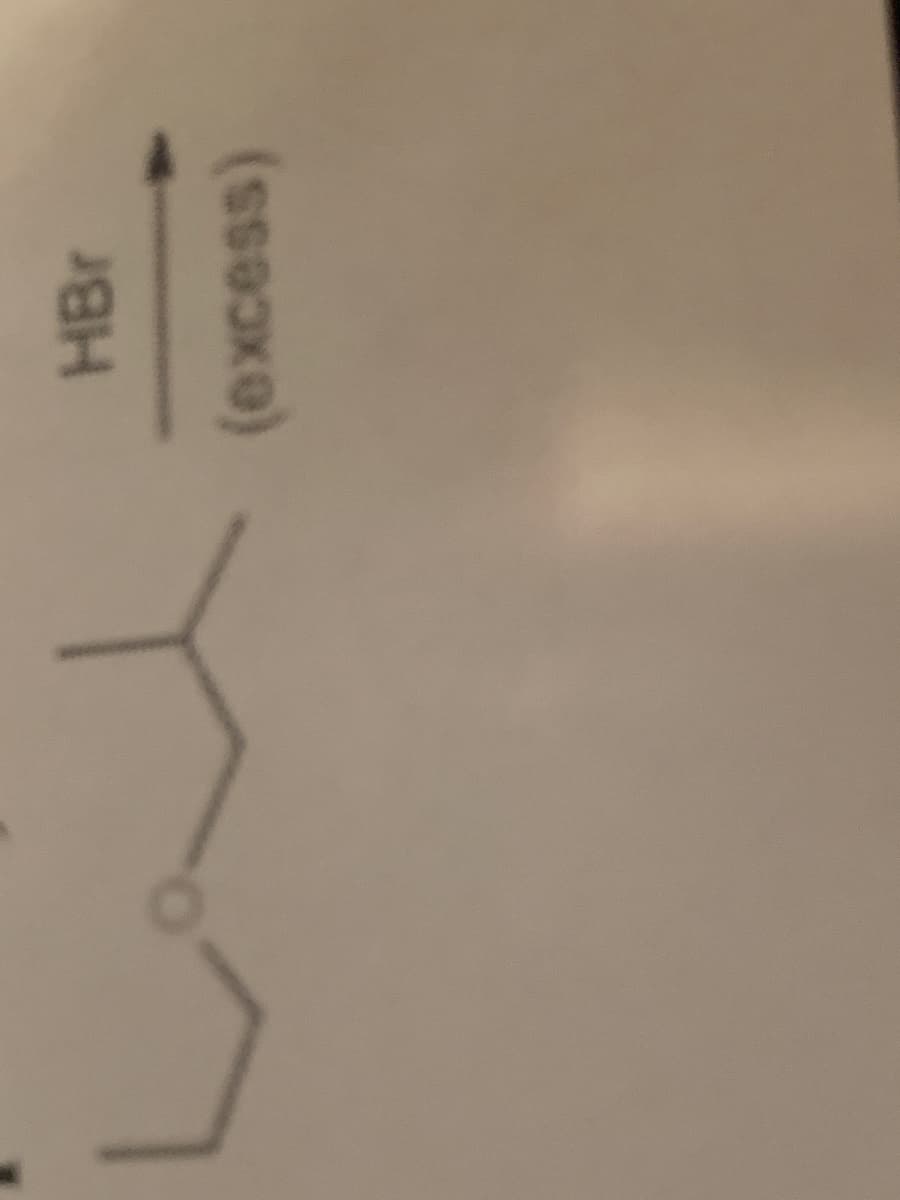
Q: 2. Write the equation for the reaction between propylamine and HBr.
A: Note : Since you have posted multiple questions, we are entitled to answer the first only. Please…
Q: There are two steps in the extraction of copper metal from chalcocite, a copper ore. In the first…
A:
Q: Which of the following compounds has the smallest Keq for the addition of water. CF3
A:
Q: O STATES OF MATTER Understanding how solubility varies with temperature and. Predict V will be…
A:
Q: Use the given standard entropy values to calculate the standard entropy change to convert hydrogen…
A: 1.) To calculate entropy change we would use given standard entropy values of all species. Note…
Q: Which of the following compounds has the smallest Keq for the addition of water. CF3
A:
Q: Solubility of AgCl is 0.0000123 M. What is the solubility product, Ksp for AgCl?
A: Given: Molar solubility of AgCl = 0.0000123 M
Q: Write the wave function of the orbital occupied by the last electron in the 47Ag atom
A:
Q: The C=O bond the C=C bond in alkenes. O A. as polar as B. is less polar than O C. nonpolar like O D.…
A:
Q: Label each as either an element or compound. Ni NO2 HI
A: 1. Ni - This is an element of d-block with atomic number 28 in periodic table. 2. NO2 -This is a…
Q: Freon-12 (CCI2 F2) is used as a refrigerant in air conditioners and as a propellant in aerosol cans.…
A:
Q: For the the molecule represented below: A Но. C OH ОН В Rank the labeled acidic protons (H atoms)…
A: The proton which can easily be removed is most acidic and vive versa.
Q: A certain radioactive nuclide has a half life of 8.00 hour(s). Calculate the rate constant for this…
A:
Q: Would an aqueous solution of 0.25M sodium formate be acidic, basic or neutral? Write the equation…
A: Given, An aqueous solution of 0.25M sodium formate is:
Q: जाNe काट nmss of चकnc m दुn १ ४०c ०4 चंकाट \S ०.388১/8'C .चnc i5 उरcतामी० केटक्ी headed ৪৪. 3C Jo…
A:
Q: The names and chemical formulae of some chemical compounds are written in the first two columns of…
A: we need find the major species in water when the given compounds are dissolved in water
Q: How many bonds could each of the following chelating ligands form with a metal ion?
A: Given; structure of molecules
Q: The solubility of PbBr2 is 0.00101 M. What is the solubility product, Ksp for PbBr2. Report answer…
A: Given : Solubility of PbBr2 is 0.00101 M
Q: Draw the structural formula for each of the following. formaldehyde (methanal)
A: Structural formula:
Q: Nitric acid is often manufactured from the atmospheric gases nitrogen and oxygen, plus hydrogen…
A: NH3 is formed from Haber process ..
Q: Answer the questions in the table below about the shape of the hydrogen cyanide (HCN) molecule. How…
A: The molecule given is hydrogen cyanide (HCN).
Q: Calcium sulfide and water react to produce calcium hydroxide and hydrosulfuric acid. If 59.44 g of…
A:
Q: X represents a nonmetal atom with an atomic mass of 107.91 amu. Calculate the number of grams of…
A:
Q: Number of atoms of Carbon in 7 moles of C4H10 are: O 42.15x10^23 O 24.09x10^23 O 168.62x10^23 O…
A: Number of atoms of carbon in 7 moles of C4H10 - - > Carbon atoms = 4 Total number of carbon…
Q: What is the comrect product for the following reaction? H2N. .CI H2N. H2N. II H2N. IV Select one:…
A:
Q: Draw the Lewis structure for the selenium dioxide (Seo,) molecule. Be sure to incude all resonance…
A: Lewis structure of SeO2 is
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


