HC CH reaction with HC CH lysosome .CH CH HC CH3 HC CH3 H2 HC C CH HC CH CH3 CH3 OH OH inside the cell, propranolol sometimes encounters lysosomes, which alter its NH group as shown above (right). reacting with the lysosomes, propranolol becomes trapped inside the cell and can cause psoriasis or other inflammatory side effects. Use IMFS to explain why the altered propranolol gets trapped. T
HC CH reaction with HC CH lysosome .CH CH HC CH3 HC CH3 H2 HC C CH HC CH CH3 CH3 OH OH inside the cell, propranolol sometimes encounters lysosomes, which alter its NH group as shown above (right). reacting with the lysosomes, propranolol becomes trapped inside the cell and can cause psoriasis or other inflammatory side effects. Use IMFS to explain why the altered propranolol gets trapped. T
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter31: Completing The Protein Life Cycle: Folding, Processing, And Degradation
Section: Chapter Questions
Problem 9P
Related questions
Question
100%
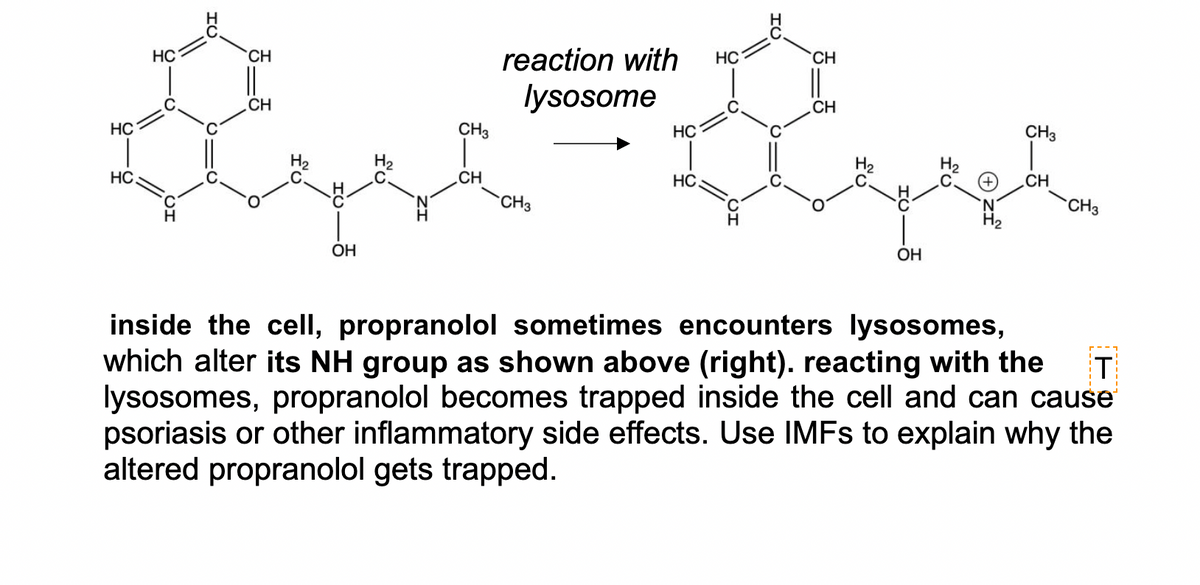
Transcribed Image Text:HC
CH
reaction with
HC
CH
lysosome
CH
CH
HC
CH3
HC
CH3
HC
CH
HC.
CH
CH3
CH3
ОН
OH
inside the cell, propranolol sometimes encounters lysosomes,
which alter its NH group as shown above (right). reacting with the
lysosomes, propranolol becomes trapped inside the cell and can cause
psoriasis or other inflammatory side effects. Use IMFS to explain why the
altered propranolol gets trapped.
T
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biology (MindTap Course List)
Biology
ISBN:
9781337392938
Author:
Eldra Solomon, Charles Martin, Diana W. Martin, Linda R. Berg
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning