Q: In a titration experiment, H2O2(aq) reacts with aqueous MnO4^1- (aq) as represented by the equation…
A: In a titration experiment , H2O2(aq) reacts with aqueous MnO4- as represented by the equation- 5…
Q: For the following reaction, 0.470 moles of magnesium nitride are mixed with 0.102 moles of water.…
A: Mg3N2 + 6 H2O --> 3 Mg(OH)2…
Q: Calculate the following: Number of moles of: AgCl(s) Cl(aq) Pb²+ (aq) [CI] in PbCl2 solution (mol/L)…
A: As per bartleby expert guidelines, I am allowed to answer first three parts of a question. Please…
Q: A student mixed 100.00 mL of Ba(OH):(aq) (unknown molarity) with 500.00 mL of 0.0200 M NaOH(aq) in a…
A: Given that a student mixed 100.00 mL of BaOH2 with 500.00 mL of 0.0200 M NaOH in a conical flask to…
Q: In the following acid-base neutralization, 1.68 g of the solid acid HC6H5O (94.12 g/mol) neutralized…
A: Moles of acid = mass / molar mass = 1.68 / 94.12 = 0.01785 approx Since from the reaction, 1 mole of…
Q: To 2.12L of 0.473M NaOH, you add 3.52L of a second NaOH solution of an unknown concentration. The…
A: Given that : Initial volume = 2.12 L Molarity of initial NaOH = 0.473 M Volume of added NaOH = 3.52…
Q: Complete and balance each of the following molecular equations involving acid-base reactions.…
A:
Q: Complete and balance each of the following molecular equations involving acid-base reactions.…
A: Weak acid reacts with strong base to produce water and sat.
Q: You are preparing standard acid and base solutions for the laboratory, using potassium hydrogen…
A: Here we are using KHP (1.55 g) to standardize NaOH solution. After standardization of NaOH…
Q: The iron in a sample of iron ore can be converted completely to Fe2+ in aqueous solution, and this…
A:
Q: Consider an acid-base neutralization reaction: H2SO4(aq) + 2 NAOH(aq) NazSO4(aq) + 2 H2O(1) --->…
A:
Q: Suppose in an experiment to determine the amount of sodium hypochlorite in bleach, 0.0000552 mol…
A:
Q: For the reaction 3 КОН + Н, РО, → K,PO, + 3 H,0 how many grams of phosphoric acid, H3PO4, are needed…
A: Consider the molar masses of reactant and product species as; Molecular mass of KOH = 56.10 g/mol…
Q: If you are writing an equation of a weak electrolyte dissolving in H20 would it still create cations…
A: An electrolyte will be a strong electrolyte if it is completely dissociated in the presence of…
Q: Aqueous sodium hypochlorite (NaOCl, household bleach) is a strong oxidizing agent that reacts with…
A:
Q: Calculate the molarity of a KNO, solution prepared by adding 10.1103 g KNO, in a 250 mL volumetric…
A:
Q: What are the spectator ions in the following reaction? K2SO4(aq) + BaCl2(aq) --> BaSO4(s) +…
A: The given reaction is,
Q: Given the reaction below, if 128.5 mL of 0.860 M Al(NO3)3 is reacted with 246.5 mL of 1.15 M NaOH…
A: The balanced reaction taking place is given as, => Al(NO3)3 + 3 NaOH -------> Al(OH)3 + 3…
Q: Complete and balance each of the following molecular equations involving acid-base reactions.…
A: Ans The given equation 5. Al(OH)3(aq)+ 3HCl(aq) ---> Reaction of the Al(OH)3(aq) with HCl(aq)…
Q: Which one of the following represents the molecular equation for the reaction of NH, with HI? A) NH,…
A: The molecular equation for a chemical reaction involves the molecular form of the substances used.…
Q: The unbalance equation for the reaction between bromine, Br2(aq), and iodate ion, IO3-(aq), in…
A: The reaction given is, => Br2 + IO3- → Br- + IO4-
Q: he endpoint with 190. ml of 0.4000 Mhydrogen chloride (HCI) solution. balanced chemical equation for…
A: Given reaction is an acid-base reaction in which HCL acid and carbonate is a base because acid gives…
Q: Please Complete the balance and label the raction type Complete and Balance Reaction Type Mg + O2…
A: 2Mg + O2 --> 2MgO This is a type of Combination Reaction.
Q: Following the procedure, a student recorded the initial volume of NaOH in his buret as 1.41 mL. He…
A: Given: Initial volume of NaOH in the burette= 1.41 mL Final volume of NaOH in the burette= 12.91 mL…
Q: An aqueous solution of Pb(NO,), and KI are mixed together. Suppose KI is the limiting reagent.…
A: Limiting Reagent : Limiting reagents are the reactants which is consumed completely during a…
Q: (ii) A student was given 400 cm of aqueous ammonia solution, NH, (aq). The student was asked to…
A: The balanced reaction given is: 2NH3(aq)+H2SO4(aq)→(NH4)2SO4 Conversion unit: 1 L=1 dm31 L=1000 cm3…
Q: a 45 mL of 0.16 M CsOH solution was reacted with 65 mL of 0.0750M H2SO4 solution. -write the…
A: The balanced molecular equation for a reaction between CsOH and H2SO4 is follow…
Q: Write the net ionic equation, including phases, that corresponds to the reaction Pb(CIO,),(aq) +…
A:
Q: A reaction is carried out by mixing together a solution of K,CO, and a solution of Fe(NO,),. K,CO,…
A: Given : K2CO3 + Fe(NO3)2 --> FeCO3 + 2KNO3
Q: Lead(II) nitrate and ammonium iodide react to form lead(II) iodide and ammonium nitrate according to…
A:
Q: A student mixed 100.00 mL of Ba(OH)2(aq) (unknown molarity) with 500.00 mL of 0.0200 M NAOH(aq) in a…
A: Solution -
Q: 25 ml sample of H3PO4, requires 31.15 mL of 0.242N KOH for its titration. What is the normality of…
A:
Q: Reaction 2: Precipitating Cu(OH)2(s) with NaOH(aq) Balanced Formula Equation with states of…
A: as the given question, precipitating reaction, Cu(OH)2 with NaOH to give a product and salt will…
Q: 6 ml of concentrated HCl acid solution was diluted to 180 mL. Then 65 mL sample of the diluted…
A: The volume of the concentrated HCl solution taken = 6 mL The volume of the diluted HCl solution =…
Q: A 1.4350-gram of KHP (actual formula = KHC8H4O4; molar mass = 204.22 g/mol) was dissolved in 50.00…
A: We have given that KHC8H4O4(aq) + NaOH(aq) → NaKC8H4O4(aq) + H2O(l) Mass of KHP = 1.4350g…
Q: What is the correct net ionic equation,
A:
Q: The balanced equation for the neutralization reaction of aqueous H2SO4 with aqueous KOH is shown.…
A:
Q: The Ca in a 200.0-mL sample of natural water was determined by precipitating the cation as CaC,O4…
A: Mass of empty crucible ( A )= 26.6002 g Mass of crucible + CaO ( B) = 26.7134 g Mass of CaO = B…
Q: What volume of 0.479 M lithium hydroxide is required to neutralize 0.627 g of oxalic acid,H2C2O4?…
A:
Q: A 0.879 g sample of a CaCl2·2H2O/K2C2O4·H2O solid salt mixture is dissolved in ~100mL of deionized…
A: Given:Mass of CaCl2.2H2O +mass of K2C2O4·H2O = 0.879 g.Mass of CaC2O4 = 0.284 g.Volume of water =…
Q: What mass of Na2CO3, in grams, is required for the complete reaction of 50.0 mL of 0.125 M HNO3? MW…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A: Given that : We have to balance the following equation under basic conditions : ____CrO42- +…
Q: A titration is performed to determine the amount of sulfuric acid, H2SO4,, in a 6.5mL sample taken…
A:
Q: Complete the following reaction and balance it. Fe(OH)3(aq) + HCl(aq) → a) FeCl3(aq)+ 3…
A: Fe(OH)3 : Iron (III) Hydroxide HCl : Hydrochloric acid FeCl3 : Iron (III) Chloride H2O : Water
Q: When the following equation is balanced properly under basic conditions, what are the coefficients…
A:
Q: A solution of 0.332 M KOH is used to titrate 16.0 mL of a 0.192 M H3PO4 solution. What…
A: No. of moles = Molarity × volume We need to balance ( no. of moles ÷ stoichiometric coefficient) .…
Q: tion is carried out by mixing together a solution of HF and a solution of NaOH. HF (aq) + NaOH (aq)…
A: To write the net ionic equation, first of all write the balanced molecular equation. Then write the…
Q: What is the net ionic equation and ΔHrxn of NaOH(aq) + NH4Cl (aq) -> NaCl(aq) + NH3(aq)+H2O (l)?
A:
Q: A 1.4350-gram of KHP (actual formula = KHC8H4O4; molar mass = 204.22 g/mol) was dissolved in 50.00…
A: Since, equal no. of moles of KHP and NaOH are required according to chemical equation so, we can…
Q: The lead(II)ion can be effectively removed from solution via the addition of phosphate ions. The…
A: Here a solution is made by dissolving 6.93 g of Pb(NO3)2 to 50 ml of water and finally the volume…
Complete the following reactions. (If the reaction doesn’t occur, please write no reaction)
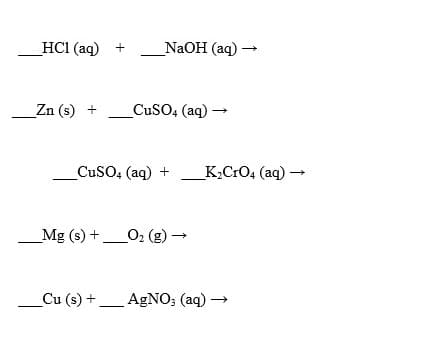
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- Sum of coefficients C7H8 + O2 --> CO2 + H2O after balancing6. One teaspoon of sertraline 20 mg/mL must be mixed into eight fluid ounces of water before consuming. Express the final concentration of sertraline when mixed with water as a ratio strength (w/v). Rounddenominator of ratio strength (w/v) to the nearest whole number.Stock iron(II) solution (200Ug mL-1 Fe) ferrous ammonium sulfate hexahydrate mass= 0.1437g, transfer it to a 100 ml beaker. add 15 ml approx of water and 15m1 'approx of dilute sulphuric acid (2M H2SO.). then transfer FeII to 100 ml flask makeup to the mark with water. calculate the moles of ferrous ammonium sulfate hexahydrate solution in unit ug/mL.
- A) An aqueous solution of ethyl alcohol is made by transfering 4.72 mL of liquid ethyl alcohol to a 200 . mL volumetric flask, and then adding enough water to fill the flask to the mark. What is the volume/volume percentage of ethyl alcohol in the solution? Volume/volume percentage = ________% B) An aqueous solution of acetic acid is made by transfering 17.5 mL of liquid acetic acid to a 200 . mL volumetric flask, and then adding enough water to fill the flask to the mark. What is the volume/volume percentage of acetic acid in the solution? Volume/volume percentage = ________%Magnesium +Acetic Acid --> C2HG5+ O2 ---> Iron (III) carbonate -->A pipet is used to transfer 3.00 mL of a 2.00 M stock solution in flask “S” to a 25.00-mL volumetric flask “A,” which is then diluted with DI H2O to the calibration mark. The solution is thoroughly mixed. Next, 3.00 mL of the solution in volumetric flask “A” is transferred by pipet to a 50.00-mL volumetric flask “B” and then diluted with DI H2O to the calibration mark. Calculate the molarity of the solution in volumetric flask “B.”
- What volume of 95.0% alcohol by weight (density: 0.809g/cm^3) must be used to prepare150 cm^3 of 30.0% alcohol by weight (density: 0.957g/cm^3)A pipet is used to transfer 5.00 mL of a 1.25 M stock solution in flask “S” to a 25.00-mL volumetric flask “B,” which is then diluted with DI H2O to the calibration mark. The solution is thoroughly mixed. Next, 3.00 mL of the solution in volumetric flask “A” is transferred by pipet to a 50.00-mL volumetric flask “B” and then diluted with DI H2O to the calibration mark. Calculate the molarity of the solution in volumetric flask “B.”One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(I) carbonate, in concentrated sulfuric acid The sulfuric acid reacts with the copper(I) carbonate to produce a blue solution of copper(I) sulfate. Scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: Fe(s) + CuSO4(aq) -> Cu(s) + FeSO4(aq) Suppose an industrial quality-control cherist analyzes a sample from a copper processing plant in the following way. He adds powdered iron to a 300. mL copper(I) sulfate sample from the plant until no more copper will precipitate. He then washes, dries, and weighs the precipitate, and finds that it has a mass of 75. mg Calculate the oriqinal concentration of copper(I) sulfate in the sample. Round your answer to 2 significant digits.
- (v/v)% = 10. mL ethanol 150 mL mouthwash × 100% = 6.7 % (v/v) ethanol how do I insert this calculation into my scientific calculator? can you please give me a detailed description I'm trying to watch videos but i don't know how to put into the calculatorA pipet is used to transfer 6.00 mLmL of a 3.75 MM stock solution in flask “S” to a 25.00-mL volumetric flask “A,” which is then diluted with DI H2OH2O to the calibration mark. The solution is thoroughly mixed. Next, 10.00 mLmL of the solution in volumetric flask “A” is transferred by pipet to a 50.00-mL volumetric flask “B” and then diluted with DI H2OH2O to the calibration mark. Calculate the molarity of the solution in volumetric flask “B.”6. A stock solution containing 25% w/v of chlorhexidine is used to prepare an antiseptic such that when the antiseptic is diluted 1 in 25, a 1 in 500 wash solution is obtained which is ready for use by the patient. What volume (ml) of the stock solution is required to prepare 5 litres of the antiseptic to fulfil a bulk manufacturing order?

