HCI(9) and HCI(aq) Match the words in the left column to the appropriate blanks in the sentences on the right. Reset fewer particles will have the higher standa occupy a smaller volume entropy at 25 °C. This can best be explained because liquid or aqueous phases have more degrees of freedom associated with movement
HCI(9) and HCI(aq) Match the words in the left column to the appropriate blanks in the sentences on the right. Reset fewer particles will have the higher standa occupy a smaller volume entropy at 25 °C. This can best be explained because liquid or aqueous phases have more degrees of freedom associated with movement
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter8: Electrochemistry And Ionic Solutions
Section: Chapter Questions
Problem 8.76E
Related questions
Question
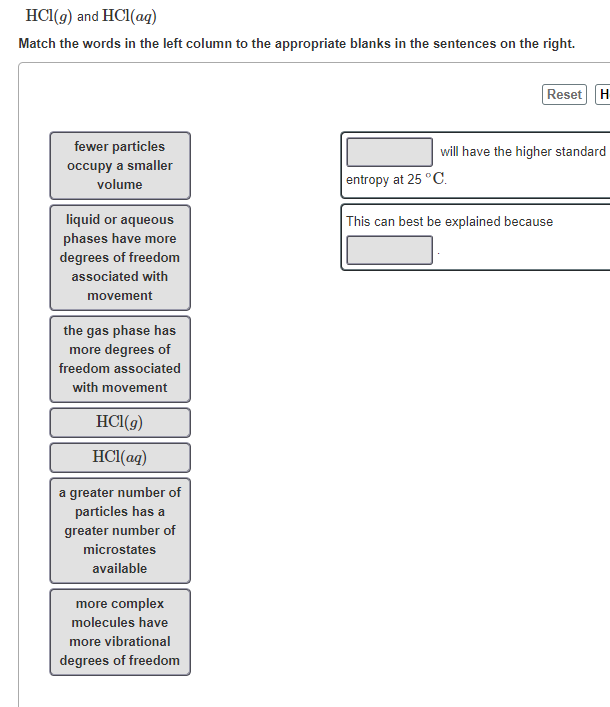
Transcribed Image Text:HCI(9) and HCI(ag)
Match the words in the left column to the appropriate blanks in the sentences on the right.
Reset
fewer particles
will have the higher standard
occupy a smaller
volume
entropy at 25 °C.
This can best be explained because
liquid or aqueous
phases have more
degrees of freedom
associated with
movement
the gas phase has
more degrees of
freedom associated
with movement
HCI(g)
HCI(aq)
a greater number of
particles has a
greater number of
microstates
available
more complex
molecules have
more vibrational
degrees of freedom
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,