Q: Referring to the molecule below, which atom is the most acidic H bonded to? * 15 13 но 14 12 11 16…
A: An acid is a compound which dissociate to give hydrogen ion on dissolving in water. Higher the…
Q: pH + pOH = 1.0 x 10-14 is this true or false?
A: pH + pOH = 1.0 * 10-14 This statement is false
Q: 2. I. Name the following acids based on their dry and aqueous forms. Chemical Formula Dry Aqueous…
A: Strong acid is an acid which dissociates completely in solution to give the hydrogen ions whereas a…
Q: Consider only the major contributing Lewis structures. How many total resonance structures does the…
A: Resonance :- The phenomenon of representation of a molecule or ion with more than one Lewis…
Q: Label each substance "acid" or "base", write the products and their charges, and indicate the…
A: Acid- The species which is able to donate H+ ion. Base- The species which is able accept H+ ion.
Q: e acid
A: Acid ionization constant Ka is given by the constant of ions divided by the concentration of…
Q: Consider the following data on some weak acids and weak bases: acid base Ka name formula name…
A: The salt is an ionic compound formed when an acid reacts with a base. The salt solution can be…
Q: Use the table below to answer the following questions: Table 1 - List of Weak Acids Acid Formula Ka…
A: According to Bronsted Lowry concept bases accept protonH+ and acids donate protonH+. Recall the…
Q: write the chemical formula of the base: Cl-(aq)+HSO4-(aq) HCI(aq)+S042-(aq)
A: The given reaction is shown below.
Q: Arrange the compounds in each set in order of increasing base strength. consult Table 4.1 for pKa…
A: Acids: These are substance that donates proton. Bases: These are substances that accepts proton.…
Q: Which of the following is not an acid
A: Given group of answer choices HBr , HCl , HI , HNO3 are acid because they gives H+ ions in water.
Q: your e Text. C6H5NH2 Write the formula for the conjugate acid of each of the following bases.…
A: Write conjugate acid of C6H5NH2 ?
Q: Choose the STRONGEST base from the list below NH2 NH2 `N' NO2 II II IV
A:
Q: Question 3 Using your PHET simulator #1, which of the following species is most prevalent in the…
A:
Q: Acid Base Answer Bank НСООН HCOO- CH,COOCH, HNO3
A: Given compounds- HCOOH, HCOO-, -CH2COOCH3,HNO3
Q: H2PO4 is an amphiprotic ion since it has both a conjugate acid and a conjugate base. Identify both…
A:
Q: Show your explanation and solution: The pH of 0.1M hydrazine (N2H4) is 10.47, and the pH of 0.1M…
A: Given: The pH of 0.1M hydrazine= 10.47 The pH of 0.1M ammonia=11.12 To find: Which compound is a…
Q: Arrange the compounds in each set in order of increasing base strength. consult Table 4.1 for pKa…
A: Ethoxide is an base, which accepts proton and becomes ethanol (conjugate acid). Ammonium ion is an…
Q: 12. For the reaction shown below, draw the conjugate acid and conjugate base in the appropriate…
A: Acid after loosing hydrogen ion it becomes conjugate base . Base after gaining hydrogen ion it…
Q: An Acid contains more O [H] > [OH] OIOH] > [H] )Contains no (H) OH-OH
A: Acid The substance which lose the Hydrogen Atoms are called as Acid . The substance which gain the…
Q: Fill in the table below and answer the questions using these formulas. pH = -log [H*] pOH = -log…
A: # The ionic product of water in we used to calculate the hydrogen ion concentration or hydroxide Ion…
Q: Which acid is named correctly? A. HNO3 nitric acid B. HNO3 nitrate acid C. H2SO3 sulfate acid D.…
A: we have to say the correct name of the acid.
Q: label the acid,base, conjugate acid, and conjucate base pairs please answer number 2
A: A pair of substances which differ by H+ ion are called conjugate-acid base pairs HClO4 +H2SO4-…
Q: Which of the following is a polypeotic acid? a)perchloric acid b)nitric acid c)acetic acid d)…
A: Any acid which can donate or release more than one proton when dissolved in solvent is called as…
Q: For the system here: HOBR +OH-1 ------H2O +OBr -1 Bronsted would classified the base species as OBr…
A:
Q: conjugate base of HPO2-4
A: Conjugate base is obtained by loss of H+
Q: Conjugate Base Formula Kb of Conjugate Base Acid Name Acid Formula Ка pka Acetic acid HC,H,O, 1.8 x…
A: Pka = -Log Ka Conjugate Base structure is Deprotonated form of acid
Q: Show your work for the question above: "12. What is the pH of a solution in which [OH] = 0.000005…
A: Given in the question , [OH-1] = 0.000005 m pH = 14 + log[OH-1]
Q: Arrange the compounds in each set in order of increasing base strength. consult Table 4.1 for pKa…
A: Higher the acidity, higher is the Ka and lower is the pKa.
Q: A solution was prepared with 3.2 x 10-11 M [H+]. Is this solution acidic or basic? Group of answer…
A:
Q: complete a net ionic equation for each proton-transfer reaction using curved arrows to show the flow…
A: The net ionic equation for each proton-transfer-reaction using curved arrows to show the flow of…
Q: Classify each reactant and product in the reaction as an acid or base according to the Brønsted…
A:
Q: [ Select ] taste bitter, V [ Select ] taste sour. Bases Acids
A: Acids are substances which have the ability to donate hydrogen ions whereas bases are substances…
Q: Try the following problem then post a readable scan or photo of your work in Blackboard under the…
A: Solution: The conjugate base for given compound are provided below:
Q: QUESTION 4 The conjugate base to HSO4 is SO, O True False TION
A: We have to predict whether given statement is false or true.
Q: Kotone one· 2.
A: IUPAC nomenclature
Q: Least acidic Most acidic Answer Bank O,N но OH -OH O,N- OH
A: The order of acidic character from least acidic to most acidic is shown below for all the four…
Q: Ph Ph OH Ph b 1.03 2. NaBH4 (excess) Ph LDA in THF allyl bromide
A: The reagents (a), (b) and product (c), (d) can be predicted by analysing the product, reagent and…
Q: Interactive 3D display mode H.C CH, CH Include the appropriate base in your response. Draw the m…
A: We can findout starting material of this compound by doing retrosynthetic analysis of this compound.…
Q: Indicate whether each of the following statements is true or false. For each statement that is…
A: The above statement is correct but its reasoning is false. H2S is less acidic than H2Te because on…
Q: complete a net ionic equation for each proton-transfer reaction using curved arrows to show the flow…
A:
Q: Q2. Complete the following table and show your work below. (H;O'] [OH] pH acidic, basic, or neutral?…
A: It is an equilibrium constant which depends only on temperature.
Q: Consider the structures below: How many is/are Lewis base? 0 HC1 BC13 CH₂OH O a. 5 O b.0 O c. 1 O…
A: Here we have to determine the number of Lewis base from the following given compounds.
Q: K, for benzoic acid, C6H; COOH, is 6.30 × 10-5. K, for phenol (a weak acid), C,H;OH, is 1.00 ×…
A: From the above data, Ka for acid is more = So, forming acid is the strongest acid. compared to…
Q: Arrange the compounds in each set in order of increasing base strength. consult Table 4.1 for pKa…
A: Base strength of a species is its ability to accept H+ from another species. The greater the ability…
Q: Acid A: Ka = 6.43 x 10- Acid B: Ka = 4 27 × 10-6 Acid C. Ka = 3.85 × 10-5 Acid D: Ka = 2.37 × 10-4
A: * Ka represent dissociation constant * Greater the dissociation constant, greater is ka value…
Q: Arrange the following in order of DECREASING acidity. Type letter codes only of your answer in…
A: +M effecting groups on benzene ring decreases the acidity of benzoic acid and - M effecting groups…
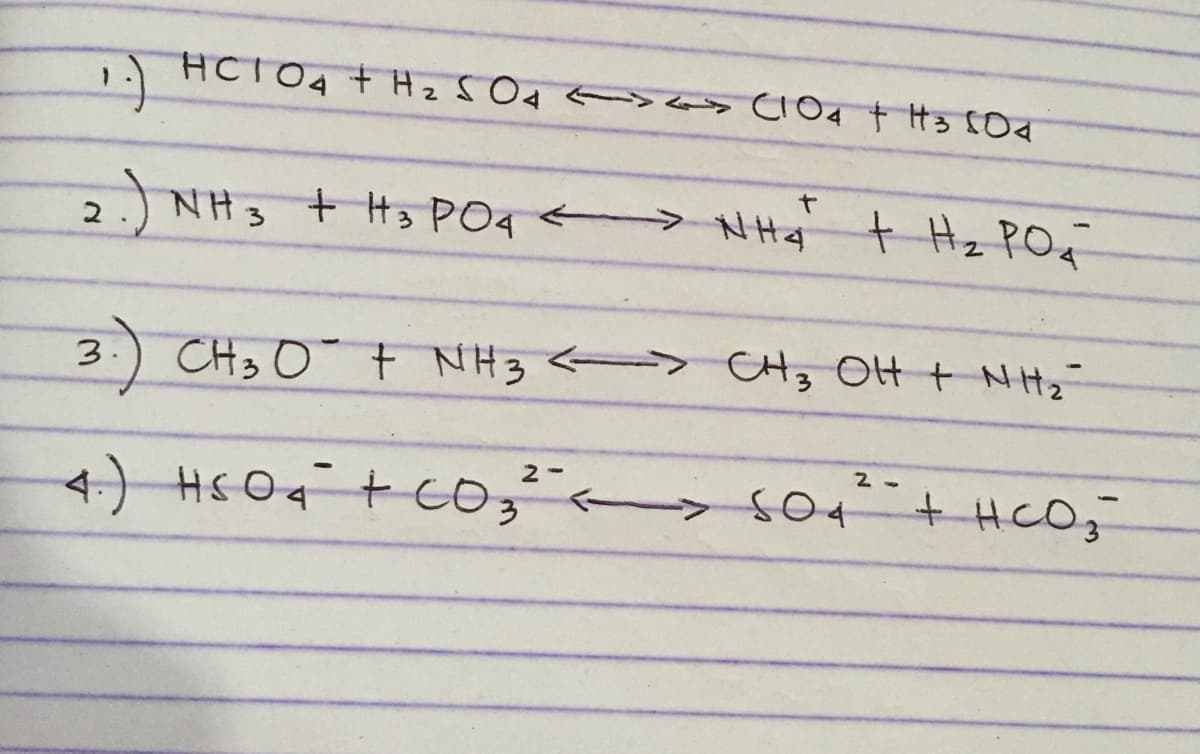
Step by step
Solved in 2 steps with 1 images

- Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. When the compounds analyzed by Robrob were passed on to the next test, anotherscientist, Kikoko, needed to determine the molecular weight of the active compound. They created a solution with a concentration of 20.0%w/w from the solid active compound and the solution had a density of 1.40 g/mL. From this solution, 10μL was taken and mixed with 190μL of reagent and water. It was analyzed and showed a concentration of 5x10-4 M compound.a. What is the concentration (in molarity) of the 20%w/w solution? b. Why was the concentration of the solution expressed as %w/w initially? c. What is the molecular weight of the active compound?Please give clear handwritten ansAnswer quetion in letter d, e and f
- Hoping for solutions since I’m having a hard time with this. Pls skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. Kriel just discovered a new protein, Krielase and for it not to unfold and lose its function, he realized that the protein needs to be constantly immersed in a pH = 6.50 solution. He is therefore interested in preparing a 50 mL 0.08 M pH = 4.50 buffer to contain the protein. He listed down the pure acids available to his disposal, and also looked for their acid dissociation constant (Ka) values. Acid Name Ka1 Ka2 Ka3 Nitrous Acid (HNO2) 4.00 x 10-4 N/A N/A Acetic Acid (CH3COOH) 1.76 x 10-5 N/A N/A Ascorbic Acid (HAsc) 7.90 x 10-5 1.60 x 10-12 N/A Phosphoric Acid (H3PO4) 7.52 x 10-3 6.23 x 10-8 4.28 x 10-13 A. Identify which weak acid system is best to use in preparing the buffer? Why? B. Identify the species which will make up the buffer system?…Plz explain, give correct and typed ansAnswer Q 72, 73 & 74 showing clearly all working and detailed explanations



