Q: DIRECTIONS: Complete the Table . SEE THE EXAMPLE BELOW Chemical Reaction H2O as Yield to acid or…
A:
Q: conjugate acid conjugate base formula Ka formula HCo, 2.2 x 10 HF 6.8 x 10* HS 1.8 x 10
A: Species formed after the removal of H+ from the acid is known as conjugate base of that acid and the…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Čarbonic acid reacts with water to yield bicarbonate ions and hydronium ions: H2CO3 + H2O…
A: Given reaction is H2CO3 + H2O = HCO3- + H3O+ We have to identify the acid ,base and…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: According to Bronsted-Lowry concept Acid is a species which can donate proton and forms its…
Q: conjugate acid conjugate base formula Ка formula HS -7 1.8 x 10 NH3 - 5 1.8 х 10 cio - 7 3.3 x 10
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Conjugate base :- Conjugate base will have one hydrogen less than normal molecular formula.…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Complete the following table --
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Conjugate acid-base pair differs by a proton HA ------> A- + H+ Here, HA is the conjugate acid of…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: In the following equation which is the acid and which is the base CO3² (aq) + H2O (1) → HCO3" (aq) +…
A: This is and acid base reaction which can be explained by bronsted -Lowry acid base theory.
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: + NAOH A OH
A: If the conjugate ion is more stable then the compound will be more acidic.
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: Conjugate acid of any substance is formed when the substance is added with a proton i.e H+ ion. And…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: We know that Ka*Kb = 1.0* 10-14
Q: Complete the table below. Round each of your entries to 2 significant digits. conjugate acid…
A: Conjugate base forms when acid loses proton, on the other hand conjugate acid forms when the base…
Q: Label the acid, base, conjugate acid, and conjugate base in the following reaction: HNO2 + CH3NH2…
A: The concept of conjugate acid-base pair is related to Bronsted-Lowry acid-base theory and according…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: We know that, a conjugate acid is formed when a proton is added to a base, and a conjugate base is…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: formula ka conjugate base formula kb HF 6.8 *10-4 F- 1.470 * 10-11 CH3NH3+ 2.27 * 19-11 CH3NH2…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Given, Ka of HCN = 4.9 x 10-10 Ka of HNO2 = 4.5 x 10-4 Kb of CH3NH2 = 4.4 x 10-4 Conjugate acid…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Relation between Ka and Kb is given by, Ka×Kb=1×10-14
Q: Identify the acid in the below reaction NH3 + H20 – NH4 + OH
A: Acid base reaction concept can be understood by various theories as follows
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Conjugate acid-base pair differs by a proton HA ------> A- + H+ Here, HA is the conjugate acid of…
Q: 3 CH3 O t NH3 >CHz OH + NH
A: Acid is species which donates proton and Base is species which accepts proton.
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: According Bronsted-Lowry acid and base theory, acids are substance which loses protons H+ to form…
Q: Classify the molecular scenes shown according to the type of acid or base each depicts. Scenes (3…
A: Solution:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Complete the table below. Round each of your entries to 2 significant digits. You conjugate acid…
A: In question 1 We are given either conjunction acid or base, we have to give the formula of rest…
Q: Bicarbonate reacts with water to produce carbonate ions and hydronium ions: HCO,- + H20 = CO2?- +…
A:
Q: Circle the phrase in parentheses which will make the statement correct. A solution with a pH of…
A: As you can see from here : pH = 7 is neutral solution pH >7 is basic, As we will go away from the…
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: There are two types of bases/acids. One is strong base/acid which is completely dissociable in into…
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: According to Bronsted-Lowry theory, an acid is a proton donor and a base is a proton acceptor. The…
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: Here, we have to write the formulas of the conjugate acids of CH3NH2 and NH3 and the conjugate base…
Q: Complete the following table: Conjugate Base Compound H2O NH3 Conjugate Acid HS CH;OH HSO4 Н.РО
A:
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: The conjugate acid of methylamine is CH3NH3+. The dissociation constant of this acid can be…
Q: Please place answers in the boxes so I won't be confused. Thank You!!
A: The formation of conjugate acid takes place when any base undergoes the process of protonation (gain…
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A:
Q: Bicarbonate reacts with water to produce carbonate ions and hydronium ions: HCO3-+ H2O = CO2?- +…
A:
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Complete the table of the given above--
Q: The pH of 0.20 M HN03 is O a. 1.58 Ob. 0.70 C. 0.20 Od. 0.63
A: Using formula in these questions: PH = -log[H+] Where as [H+]-------> concentration of H+ Using…
Q: In each equation, label the acids, bases, and conjugate pairs: f. NH3 + H3PO4 == NH4 + + H2PO4 g.…
A:
Q: Complete the table below. Be sure each of your answer entries has the correct number of significant…
A: Bronsted-lowry acid base theory:-According to this theory an acid is a substance whichdonates a…
Q: Complete the table below. Round each of your entries to 2 significant digits. You may assume the…
A: As the strength of the acid or base increases the Ka or Kb value also increases and on decreasing…
Q: What is the pH of the following aqueous solutions: When (H*] = 4.23x10-°M. pH = Type your answer…
A:
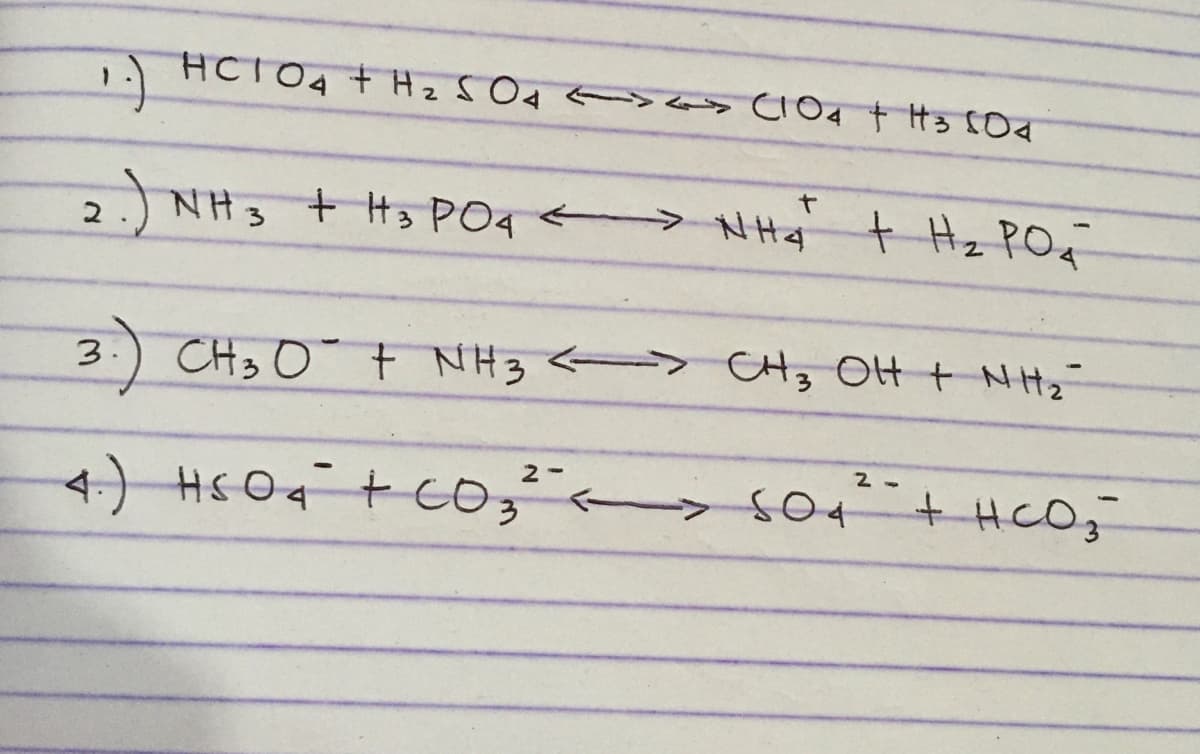
Step by step
Solved in 2 steps with 2 images

- Really hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. When the compounds analyzed by Robrob were passed on to the next test, anotherscientist, Kikoko, needed to determine the molecular weight of the active compound. They created a solution with a concentration of 20.0%w/w from the solid active compound and the solution had a density of 1.40 g/mL. From this solution, 10μL was taken and mixed with 190μL of reagent and water. It was analyzed and showed a concentration of 5x10-4 M compound.a. What is the concentration (in molarity) of the 20%w/w solution? b. Why was the concentration of the solution expressed as %w/w initially? c. What is the molecular weight of the active compound?Please give clear handwritten ansAnswer quetion in letter d, e and f
- Hoping for solutions since I’m having a hard time with this. Pls skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. Kriel just discovered a new protein, Krielase and for it not to unfold and lose its function, he realized that the protein needs to be constantly immersed in a pH = 6.50 solution. He is therefore interested in preparing a 50 mL 0.08 M pH = 4.50 buffer to contain the protein. He listed down the pure acids available to his disposal, and also looked for their acid dissociation constant (Ka) values. Acid Name Ka1 Ka2 Ka3 Nitrous Acid (HNO2) 4.00 x 10-4 N/A N/A Acetic Acid (CH3COOH) 1.76 x 10-5 N/A N/A Ascorbic Acid (HAsc) 7.90 x 10-5 1.60 x 10-12 N/A Phosphoric Acid (H3PO4) 7.52 x 10-3 6.23 x 10-8 4.28 x 10-13 A. Identify which weak acid system is best to use in preparing the buffer? Why? B. Identify the species which will make up the buffer system?…Plz explain, give correct and typed ansAnswer Q 72, 73 & 74 showing clearly all working and detailed explanations



