Q: 10 A 3.40-g sample of a mixture of naphthalene (C, H.) and pyrene (CH) is dissolved in 35.0 g…
A: Given: The mass of the mixture of naphthalene and pyrene = 3.40 g The mass of benzene (solvent) =…
Q: Convert 5.0 g of H2O into molecules of O2 need to react in the following equation: 2H2 + O2 -> 2H2O…
A:
Q: solution is made using 197.5 g of water (MM=18.02 g/mol) and 120.0 g of ethanol(MM=46.07 g/mol).…
A: Given that: Mass of water= 197.5g Molar mass of water = 18.02g/mol Moles = given mass ÷ molar mass
Q: 1. What is referred to as the change in the reactant or a product with time? A.) Chemical reaction…
A:
Q: Carbon-14 has a half-life of 5730 years, how old is a dress that was found to have a specific…
A:
Q: Describe how temperature change, both positive and negative, is a determinant of heat. 2.) If a…
A: these are multiple question . I solve first one only other part upload seperately. Thankyou
Q: Practice Exercise For the reaction N₂(g) + 3H₂(g) — 2NH3(g) Kp is 4.3 X 104 at 375°C. Calculate K,…
A:
Q: ミッションX Cl b. / Hol プ れた。 ↓
A: Here we have to synthesize the following given compounds from the given starting materials by…
Q: Determine the value of K at 25 °C for the reaction 2H20 + 2CI2= 4H* + 4CI' + O2 E° (Volts) Half-Cell…
A:
Q: 5.) Tell whether each of the following elements is an inner transition metal, noble gas, alkali…
A: Identify the elements and give its period and group number ---
Q: 3. For the reaction: 2 NO2 (g) + O2 (g) 2 2 NO3 (g) at 923 °C. Kc is 42.5. If 0.0500 mole of NO2…
A:
Q: How many electrons can be described by the quantum numbers n = 2, l = 2, ml = 2? a. 14 b. 2 c. 6 d.…
A:
Q: carbonate ion, CO32−,CO32−, draw all of the resonance structures. Identify which orbitals overlap
A:
Q: CI 1 mole HCI q ? CI Via 1,2-addition Via 1,4-addition The starting material should be a hydrocarbon
A:
Q: (Q63) Into what two neutrally charged species does carbonic acid dissociate? Bicarbonate and water O…
A:
Q: Find the number of octet electron of the following compound PCI3 CHCI CO
A: Octet rule: Atoms can gain a full valence shell by either sharing electrons (covalent bonding) or by…
Q: Select the correct major product(s). If a rearrangement occurs, assume it occurs to form the major…
A:
Q: Identify reagents that can be used to achieve the following transformation): OH он The…
A: Detail mechanistic pathway is given below to carry out the conversion
Q: Among the gases listed, which would have the highest rate of effusion? NH3 SO₂ NO₂ CH₁
A:
Q: Draw an expanded structural formula for 3 of the compounds below. How many different resonance peaks…
A: The no. of signals in NMR is depend upon the environment of proton in the chemical strucutre. Same…
Q: lonic bonds are the interactions between a cation and an anion. Metals tend to form cations giving a…
A: Metal donate the electron and nonmetal accept the electron. Tables are given here.
Q: Draw the products of the following Diels-Alder reactions, and indicate the stereochemistry at all…
A: We have to predict the product and indicate the stereochemistry also.
Q: The combined gas law relates which of these? pressure and volume only volume and temperature only…
A:
Q: NH2 1.? CI COOH 2.? ? Ph3P-CHPH Ph Me HO Ме H. N. 2 moles of ? OEt N. NH2 F. F. 1. ? EtO 2. ? EtO F…
A: Detail mechanistic pathway is shown below to find out the reagent for the given conversion
Q: (CH3),Có onformation of
A: According to CIP sequence rule, if higher priority groups are same side of double bond then it is Z…
Q: Which of the following is a strong acid? O acetic acid hydrochloric acid hydrofluoric acid O…
A: The given acids are Acetic acid, hydrochloric acid, hydrofluoric acid and phosphoric acid.
Q: Use the observation in the first column to answer the question in the second column. observation…
A: Vapor pressure is the pressure exerted by the vapor above the liquid on that liquid. Boling point is…
Q: B C EtO. H 2. H3O* & Heat Ninhydrin NH3 COO NH3 COO Aq. HCI & Heat NH3 + CO₂ + purple compound ?+ ?…
A:
Q: (ksp) The soubiliy of caicium phosphare, CA3(POe, IS 7.1 x 10-7 mole Ines What are the molar…
A: Given, [Ca3(PO4)2] = 7.1×10-7 M Calculate concentration of its ions ?
Q: Which of the following statement(s) is(are) true? Select all that apply: The fingerprint region of…
A: The question asks different aspects about infrared spectroscopy. The energy associated with the…
Q: но H2SO4 HNO3 Il HO NO III o-NO, NO но 0-so, IV V NO, VI VII NO so. What is the product of this…
A: Given reaction: Here phenol is treated with nitric acid in presence of sulfuric acid. The products…
Q: For a particular redox reaction, NO₂ is oxidized to NO3 and Fe³+ is reduced to Fe²+. Complete and…
A: Oxidation is loss of electrons. Reduction is gain of electrons
Q: A coffee cup calorimeter with a heat capacity of 28.9 J/°C is set up to analyze an unknown metal…
A: we need to determine the identity of the metal according to the given data
Q: = The decomposition of hypothetical molecule XY shown below has an activation energy Ea kj 100.- and…
A: Arrhenius equation says that k=Aexp(-Ea/RT) where, k=rate constant, A=pre-exponential…
Q: Consider the reaction: CO(g) + Cl₂(g)COCI₂(g) Using standard absolute entropies at 298K, calculate…
A:
Q: Consider the reactions: A→2B ΔH1 2B→3C ΔH2 What is ΔH for the reaction A→3C ?
A: According to the Hess’s Law Irrespective of number of steps the total enthalpy change of a reaction…
Q: The standard cell potential for the galvanic cell below is 3.63 V. What is the cell potential for…
A:
Q: What is the frequency of light having a wavelength of 4.50 x 10 -6 cm?
A:
Q: Question # 3: Organic acid anhydrides are very important structural motifs in organic chemistry.…
A: Detail synthetic scheme is shown below
Q: 3. Label the orbitals below with their respective subshell name and how many nodes there are. Hint:…
A: The shape of s-subshell is spherical, the shape of p-subshell is dumbbell shape, the shape of…
Q: В. Propose a mechanism to account for the following reaction. Hint: Start by removing the most…
A:
Q: How many formula units of sodium chloride are present in a 3.500 mol sample
A: We have to predict the number of formula units.
Q: Compound 7 Compound 8 CH, CH, CH,CH, CH, CH,CH
A:
Q: Which electron transition produces light of the highest frequency in the hydrogen atom? a. 3s --->…
A: To find Which electron transition produces light of the highest frequency in the hydrogen atom?
Q: A student burns a small snack food item and finds that it releases 60,900 J of energy. How many…
A:
Q: Which of the following is a strong acid? O acetic acid Ohydrochloric acid Ohydrofluoric acid O…
A: More stable the conjugate base of an acid, more stronger is the acid. Strong acid has a lower pKa…
Q: Describe the following equations in terms of 4) Grams : Ca3(PO4)2 + 3SiO2 + 5C --> 3CaSiO3 + 5CO =…
A: Answer: In given questions, we have to convert the chemical equations into word equations in terms…
Q: CHEMICAL REACTION: (in acidic solution) Separate into ½ reactions H3ASO4 (aq) Ox + Zn (s) ->> AsH3…
A: The given chemical reaction is H3AsO4aq + Zns → AsH3g + Zn2+aq. We have to balance the given redox…
Q: There is no P(0) branch in rotational spectroscopy. True False
A: P branch arises in rotational spectroscopy.
Q: Question 24 Cual es la secuencia de reacciones necesaria para llevar a cabo la siguiente…
A: Detail mechanistic pathway is given below
Step by step
Solved in 2 steps with 2 images

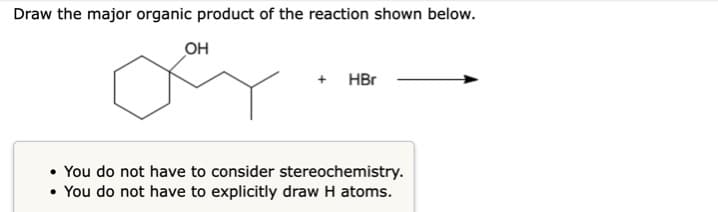
- Draw the major organic product of the reaction shown below.You do not have to consider stereochemistry.You do not have to explicitly draw H atoms.draw the major organic product generated in the reaction below. Pay attention to Regio and stereochemical detailFor the dehydrohalogenation (E2) reaction shown, draw the major organic product, including stereochemistry.
- Give the major organic product or missing starting material for the following reactions. Please do b or a explain as well.In the box to the left of each reaction below, write the mechanism by which it occurs (could be SN1, SN2, or E1, or even 2 of them). Then draw the product(s).A student proposes the following reaction mechanism for the reaction in Model 6. Which step inthis mechanism is least favorable? Explain your reasoning.
- Draw the major organic product of the reaction shown below.You do not have to consider stereochemistry.You do not have to explicitly draw H atoms.In cases where there is more than one answer, just draw one.Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. Predict the product of the reaction below.Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. What is the product of the reaction below?
- Draw the major organic product of the reaction shown below.For the following dehydrohalogenation (E2) reaction, draw the Zaitsev product(s) showing the stereochemistry clearly. If there is more than one organic product, both products can be drawn in the same box.what is the final and major product of the reaction shown below?

