Heat 8.2 moles of copper from 4 K to 20 K. If Cmol = 0.046 J mol K1, calculate the heat Q required. Include units in your answer Use SI units. Write your answer as a number in normal form.
Heat 8.2 moles of copper from 4 K to 20 K. If Cmol = 0.046 J mol K1, calculate the heat Q required. Include units in your answer Use SI units. Write your answer as a number in normal form.
Chapter3: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 19P: A gas follows on an isothermal curve, where p is the pressure, V is the volume, b is a constant, and...
Related questions
Question
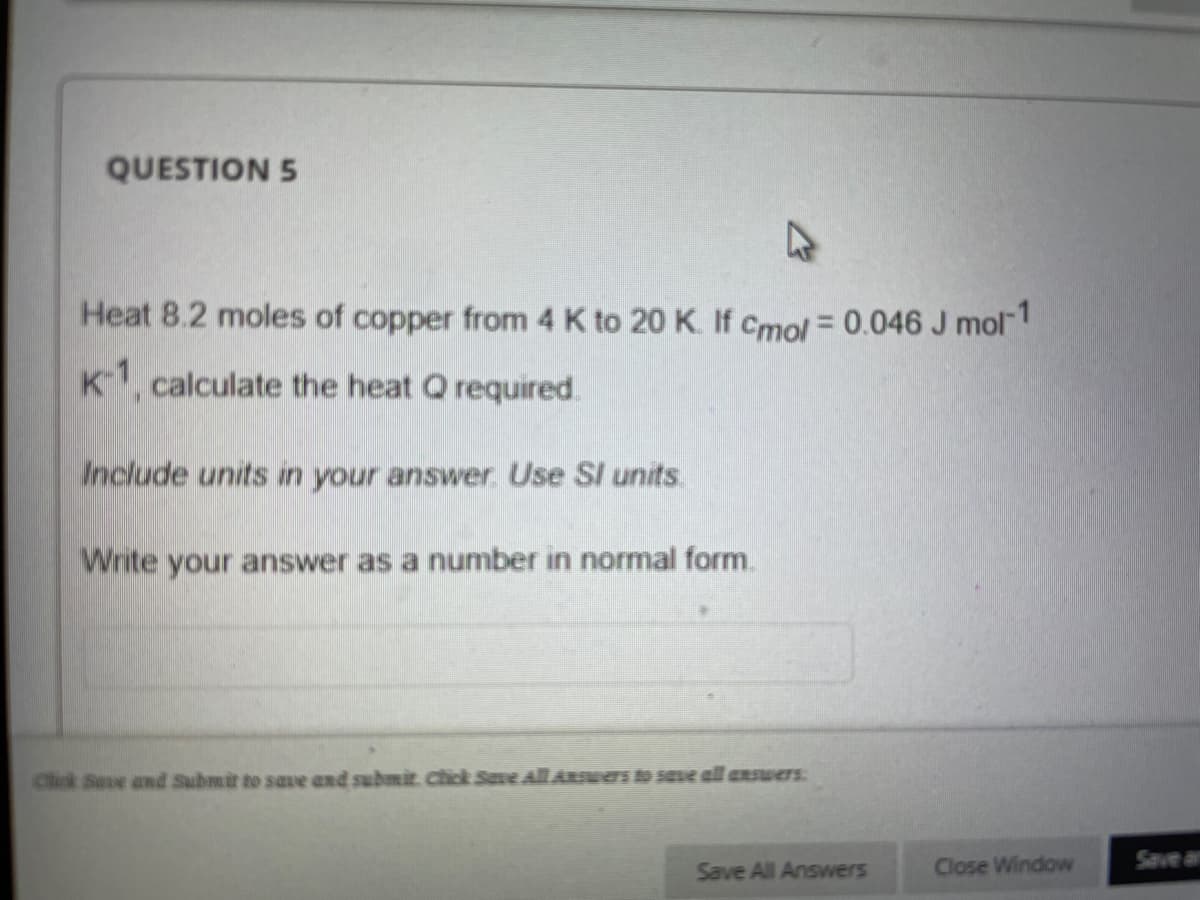
Transcribed Image Text:QUESTION 5
Heat 8.2 moles of copper from 4 K to 20 K. If Cmol = 0.046 J mol
K1, calculate the heat Q required.
Include units in your answer Use SI units
Write your answer as a number in normal form.
Cick Seve and Submir to save and submit. Chick Save All ARswers t save all ansuers:
Save All Answers
Close Window
Save an
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


College Physics
Physics
ISBN:
9781285737027
Author:
Raymond A. Serway, Chris Vuille
Publisher:
Cengage Learning

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

An Introduction to Physical Science
Physics
ISBN:
9781305079137
Author:
James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:
Cengage Learning
