Help Chandler prepare 1.5 L of 0.25 M phosphate buffer with pH 6.15 (K, for H2PO4 = 6.30 x 10-8) from the following reagents: 2.0 M stock buffer solution of the same pH - solid Na2HPO4-6H20 (MM = 250.054 g/mol) and solid NaH2PO4-6H20 (MM = 228.072 g/mol)
Help Chandler prepare 1.5 L of 0.25 M phosphate buffer with pH 6.15 (K, for H2PO4 = 6.30 x 10-8) from the following reagents: 2.0 M stock buffer solution of the same pH - solid Na2HPO4-6H20 (MM = 250.054 g/mol) and solid NaH2PO4-6H20 (MM = 228.072 g/mol)
Chapter6: The Systematic Approach To Equilibria: Solving Many Equations
Section: Chapter Questions
Problem 12P
Related questions
Question
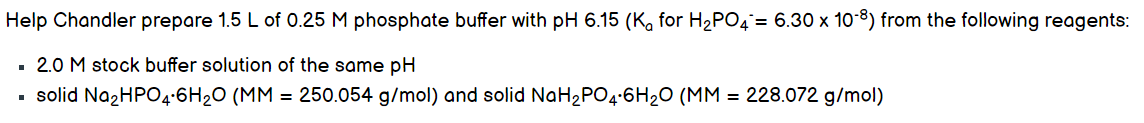
Transcribed Image Text:Help Chandler prepare 1.5 L of 0.25 M phosphate buffer with pH 6.15 (K, for H2PO4= 6.30 x 10-8) from the following reagents:
2.0 M stock buffer solution of the same pH
solid NazHPO4:6H20 (MM = 250.054 g/mol) and solid NaH2PO4:6H20 (MM = 228.072 g/mol)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



