Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.17QAP
Related questions
Question
Help me with this please. Discuss the table.
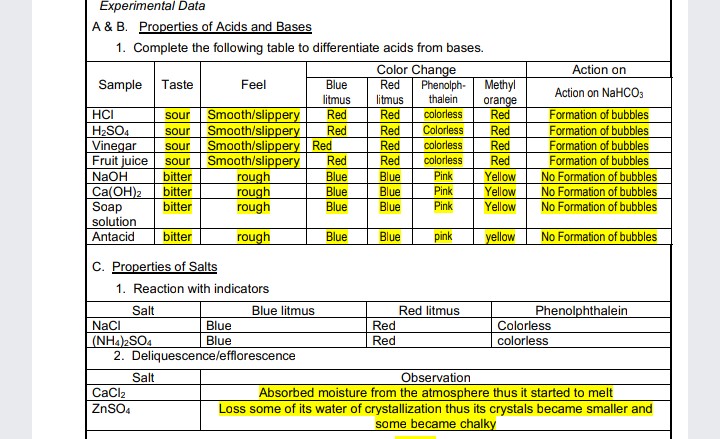
Transcribed Image Text:Experimental Data
A & B. Properties of Acids and Bases
1. Complete the following table to differentiate acids from bases.
Color Change
Action on
Blue
litmus
Phenolph- Methyl
thalein
colorless
Sample Taste
Feel
Red
Action on NaHCOs
litmus
orange
Red
HCI
H2SO4
Vinegar
Fruit juice
Smooth/slippery
Smooth/slippery
Smooth/slippery Red
Smooth/slippery
Formation of bubbles
Formation of bubbles
Formation of bubbles
Formation of bubbles
No Formation of bubbles
No Formation of bubbles
No Formation of bubbles
sour
Red
Red
sour
Red
Red
Colorless
Red
sour
Red
colorless
Red
sour
Red
Red
colorless
Red
NaOH
Blue
Blue
bitter
Pink
rough
rough
rough
Blue
Yellow
Ca(OH)2
Soap
solution
bitter
Blue
Pink
Yellow
bitter
Blue
Blue
Pink
Yellow
bitter
pink
yellow
No Formation of bubbles
Antacid
rough
Blue
Blue
C. Properties of Salts
1. Reaction with indicators
Salt
Blue litmus
Red litmus
Phenolphthalein
NaCI
Blue
Red
Colorless
(NH4)2SO4
2. Deliquescence/lefflorescence
Blue
Red
colorless
Observation
Absorbed moisture from the atmosphere thus it started to melt
Loss some of its water of crystallization thus its crystals became smaller and
some became chalky
Salt
CaCl2
ZnSO4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

