HEMWORK When a calcium chloride solution is mixed with a lead(II) nitrate solution, a precipitate forms. De alanced molecular equation, the complete ionic equation, and the net ionic equation. a. Choose the correctly balanced molecular equation that represents this reaction. CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (8) + PbCl₂ (aq) CaCl₂ (aq) + Pb(NO3)2 (aq) → CaCl₂ (aq) + Pb(NO3)2 (8) CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (aq) + PbCl2 (aq) Ca²+ (aq) + Cl(aq) + Pb²+ (aq) + NO3(aq) → Ca²+ (aq) + NO3(aq) + PbCl2 (8) CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (aq) + PbCl₂ (8) b. Choose the correctly balanced complete ionic equation that represents this reaction. 2+ 02 Ca²+ (aq) + Cl¯ (aq) + 2 Pb²+ (aq) + NO3(aq) → 2 Ca²+ (aq) + NO3(aq) + PbCl₂ (s) CaCl₂ (aq) + Pb(NO3)2 (aq) → CaCl₂ (aq) + Pb(NO3)2 (8) 2+ OCa²+ (aq) + 2 CI (aq) + Pb²+ (aq) + 2NO3(aq) → Ca²+ (aq) + 2NO3(aq) + PbCl₂ (s) OCa²+ (aq) + Cl(aq) + Pb²+ (aq) + NO3(aq) → Ca²+ (aq) + NO3(aq) + PbCl2 (s) c. Choose the correctly balanced net ionic equation that represents this reaction. Submit Pb²+ (aq) +2 Cl(aq) → PbCl2 (8) Ca²+ (aq) + Pb2+ (aq) → Ca²+ (aq) + PbCl₂ (s) Ca²+ (aq) + 2 NO3(aq) → Ca²+ (aq) + 2NO3(aq) Pb²+ (aq) + 2NO3(aq) → PbCl₂ (s) Show Hints
HEMWORK When a calcium chloride solution is mixed with a lead(II) nitrate solution, a precipitate forms. De alanced molecular equation, the complete ionic equation, and the net ionic equation. a. Choose the correctly balanced molecular equation that represents this reaction. CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (8) + PbCl₂ (aq) CaCl₂ (aq) + Pb(NO3)2 (aq) → CaCl₂ (aq) + Pb(NO3)2 (8) CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (aq) + PbCl2 (aq) Ca²+ (aq) + Cl(aq) + Pb²+ (aq) + NO3(aq) → Ca²+ (aq) + NO3(aq) + PbCl2 (8) CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (aq) + PbCl₂ (8) b. Choose the correctly balanced complete ionic equation that represents this reaction. 2+ 02 Ca²+ (aq) + Cl¯ (aq) + 2 Pb²+ (aq) + NO3(aq) → 2 Ca²+ (aq) + NO3(aq) + PbCl₂ (s) CaCl₂ (aq) + Pb(NO3)2 (aq) → CaCl₂ (aq) + Pb(NO3)2 (8) 2+ OCa²+ (aq) + 2 CI (aq) + Pb²+ (aq) + 2NO3(aq) → Ca²+ (aq) + 2NO3(aq) + PbCl₂ (s) OCa²+ (aq) + Cl(aq) + Pb²+ (aq) + NO3(aq) → Ca²+ (aq) + NO3(aq) + PbCl2 (s) c. Choose the correctly balanced net ionic equation that represents this reaction. Submit Pb²+ (aq) +2 Cl(aq) → PbCl2 (8) Ca²+ (aq) + Pb2+ (aq) → Ca²+ (aq) + PbCl₂ (s) Ca²+ (aq) + 2 NO3(aq) → Ca²+ (aq) + 2NO3(aq) Pb²+ (aq) + 2NO3(aq) → PbCl₂ (s) Show Hints
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter4: Stoichiometry: Quantitative Information About Chemical Reactions
Section: Chapter Questions
Problem 74PS: An unknown solid acid is either citric acid or tartaric acid. To determine which acid you have, you...
Related questions
Question
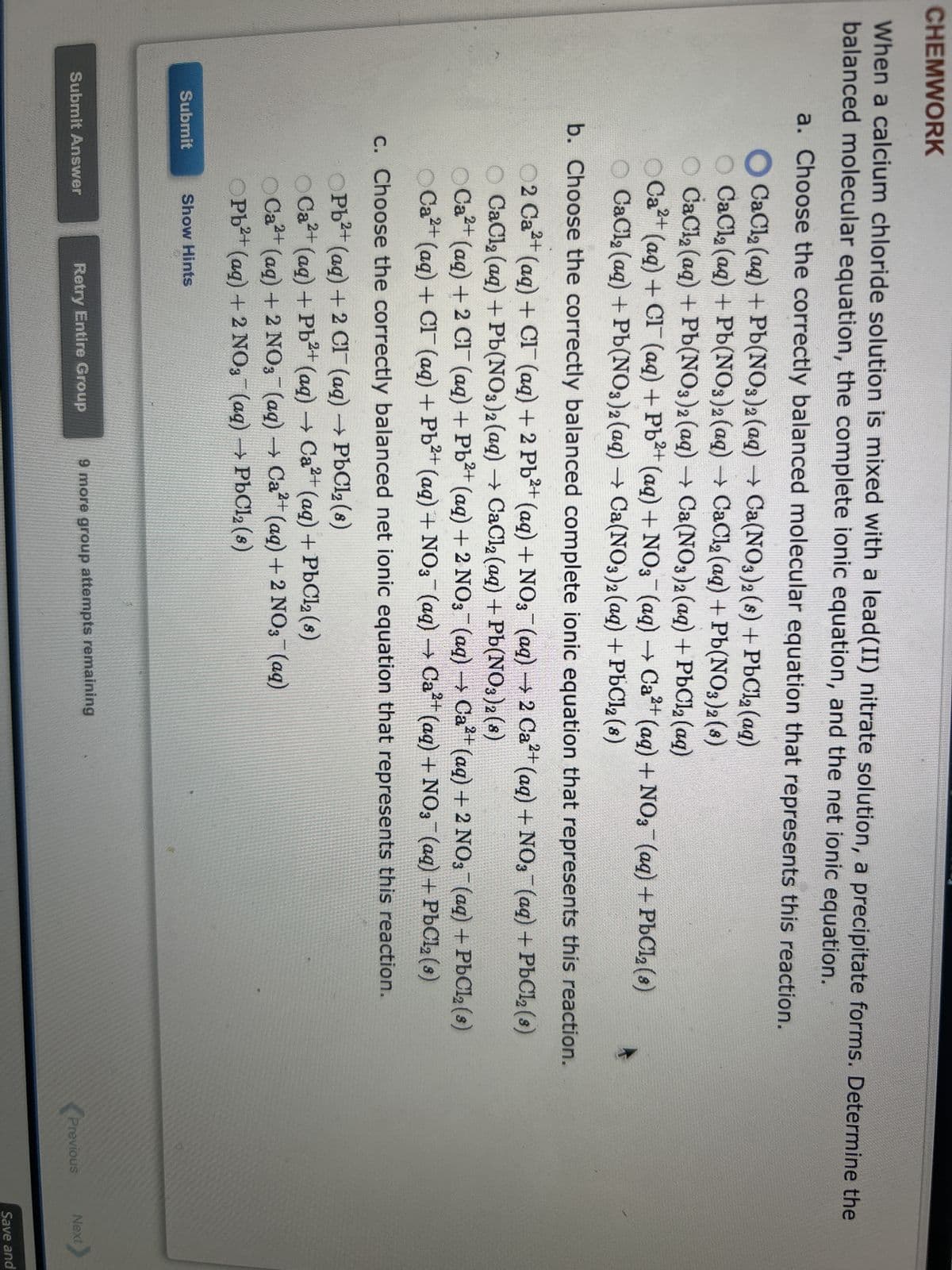
Transcribed Image Text:CHEMWORK
When a calcium chloride solution is mixed with a lead(II) nitrate solution, a precipitate forms. Determine the
balanced molecular equation, the complete ionic equation, and the net ionic equation.
a. Choose the correctly balanced molecular equation that represents this reaction.
CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (8) + PbCl₂ (aq)
O CaCl₂ (aq) + Pb(NO3)2 (aq) → CaCl₂ (aq) + Pb(NO3)2 (8)
O CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (aq) + PbCl₂ (aq)
Ca²+ (aq) + Cl¯¯ (aq) + Pb²+ (aq) + NO3(aq) → Ca²+ (aq) + NO3(aq) + PbCl₂ (8)
CaCl₂ (aq) + Pb(NO3)2 (aq) → Ca(NO3)2 (aq) + PbCl₂ (8)
b. Choose the correctly balanced complete ionic equation that represents this reaction.
2+
2+
02 Ca²+ (aq) + CI (aq) + 2 Pb²+ (aq) + NO3(aq) → 2 Ca²+ (aq) + NO3(aq) + PbCl₂ (s)
O CaCl₂ (aq) + Pb(NO3)2 (aq) → CaCl₂ (aq) + Pb(NO3)2 (8)
2+
2+
OCa²+ (aq) + 2 CI¯ (aq) + Pb²+ (aq) + 2NO3(aq) → Ca²+ (aq) + 2NO3(aq) + PbCl₂ (s)
2+
Ca²+ (aq) + Cl¯ (aq) + Pb²+ (aq) + NO3¯¯ (aq) → Ca²+ (aq) + NO3¯¯ (aq) + PbCl₂ (8)
c. Choose the correctly balanced net ionic equation that represents this reaction.
Submit
2+
OPb²+ (aq) + 2 Cl(aq) → PbCl2 (8)
2+
OCa²+ (aq) + Pb²+ (aq) → Ca²+ (aq) + PbCl₂ (s)
2+
OCa²+ (aq) + 2NO3(aq) → Ca²+ (aq) + 2 NO3(aq)
OPb²+ (aq) + 2NO3(aq) → PbCl₂ (s)
Show Hints
Submit Answer
Retry Entire Group
9 more group attempts remaining
Previous
Next
Save and
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning