Here is a graph of the pressure of nitrogen dioxide (NO,) in a reaction vessel during a certain chemical reaction. Use this graph to answer the questions in the table below. 30- 25- 20- 18.707 atm 15- 10- 5- 10 15 20 25 30 seconds O created Is NO, being created or destroyed by the chemical O destroyed reaction? O neither created nor destroyed If NO, is being created or destroyed, what is the rate at which it is being created or destroyed 9 seconds after the reaction starts? Round your answer to 2 significant digits. Also be sure your answer has the correct unit symbol. If NO, is being created or destroyed, what is the average rate at which it is being created or destroyed during the first 9 seconds of the reaction? Round your answer to 2 significant digits. Also be sure your answer has the correct unit symbol.
Here is a graph of the pressure of nitrogen dioxide (NO,) in a reaction vessel during a certain chemical reaction. Use this graph to answer the questions in the table below. 30- 25- 20- 18.707 atm 15- 10- 5- 10 15 20 25 30 seconds O created Is NO, being created or destroyed by the chemical O destroyed reaction? O neither created nor destroyed If NO, is being created or destroyed, what is the rate at which it is being created or destroyed 9 seconds after the reaction starts? Round your answer to 2 significant digits. Also be sure your answer has the correct unit symbol. If NO, is being created or destroyed, what is the average rate at which it is being created or destroyed during the first 9 seconds of the reaction? Round your answer to 2 significant digits. Also be sure your answer has the correct unit symbol.
Chapter8: Reaction Rates And Equilibrium
Section: Chapter Questions
Problem 8.18E
Related questions
Question
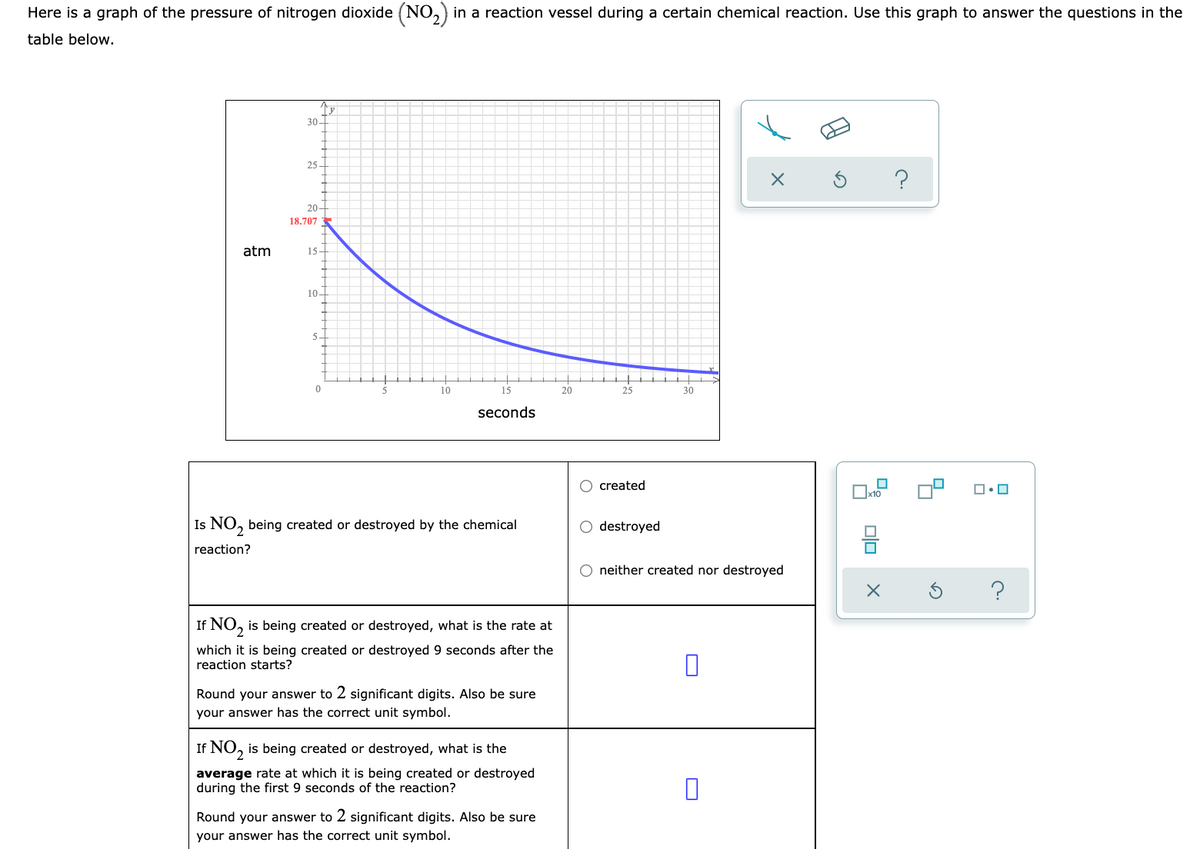
Transcribed Image Text:Here is a graph of the pressure of nitrogen dioxide (NO,) in a reaction vessel during a certain chemical reaction. Use this graph to answer the questions in the
table below.
30-
25 -
20-
18.707
atm
15-
10-
10
15
20
25
30
seconds
O created
x10
Is NO, being created or destroyed by the chemical
destroyed
reaction?
neither created nor destroyed
If NO, is being created or destroyed, what is the rate at
which it is being created or destroyed 9 seconds after the
reaction starts?
Round your answer to 2 significant digits. Also be sure
your answer has the correct unit symbol.
If NO, is being created or destroyed, what is the
average rate at which it is being created or destroyed
during the first 9 seconds of the reaction?
Round your answer to 2 significant digits. Also be sure
your answer has the correct unit symbol.
Expert Solution
Step 1

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
