A student performed three KHP titrations in Part 1 in order to standardize their NaOH solution. They reported the following measurements: Mass of KHP + Vial Mass of 'Empty' Vial Final Burette Reading Initial Burette Reading Determination #1 16.7538 g 16.2649 g 21.67 mL 0.40 mL Determination #2 14.1709 g 13.5560 g 26.98 mL 0.31 mL Determination #3 15.6625 g 15.0833 g 26.04 mL 0.93 mL How many moles of KHC8H4O4 did they use in their second titration? Report your answer to the correct number of significant figures and only report the numerical value (no units).
A student performed three KHP titrations in Part 1 in order to standardize their NaOH solution. They reported the following measurements: Mass of KHP + Vial Mass of 'Empty' Vial Final Burette Reading Initial Burette Reading Determination #1 16.7538 g 16.2649 g 21.67 mL 0.40 mL Determination #2 14.1709 g 13.5560 g 26.98 mL 0.31 mL Determination #3 15.6625 g 15.0833 g 26.04 mL 0.93 mL How many moles of KHC8H4O4 did they use in their second titration? Report your answer to the correct number of significant figures and only report the numerical value (no units).
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter20: Molecular Spectroscopy And Photochemistry
Section: Chapter Questions
Problem 67CP
Related questions
Question
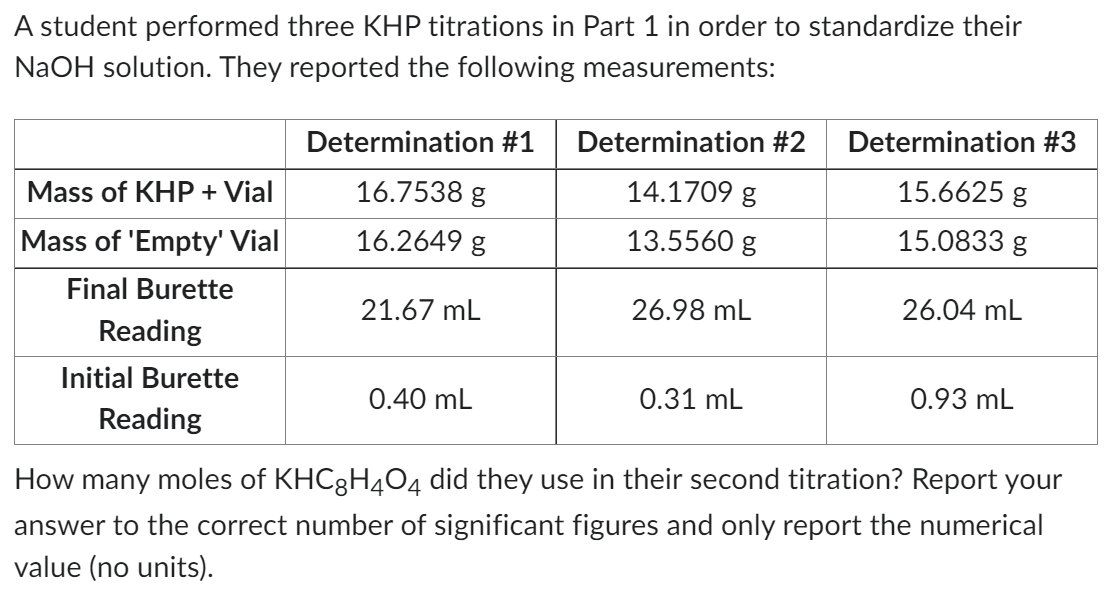
Transcribed Image Text:A student performed three KHP titrations in Part 1 in order to standardize their
NaOH solution. They reported the following measurements:
Mass of KHP + Vial
Mass of 'Empty' Vial
Final Burette
Reading
Initial Burette
Reading
Determination #1
16.7538 g
16.2649 g
21.67 mL
0.40 mL
Determination #2
14.1709 g
13.5560 g
26.98 mL
0.31 mL
Determination #3
15.6625 g
15.0833 g
26.04 mL
0.93 mL
How many moles of KHC8H4O4 did they use in their second titration? Report your
answer to the correct number of significant figures and only report the numerical
value (no units).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
hi, can you reexplain the
0.6149 g * 1 mol KHP204.22 g= 0.003011 mol KHP
Moles of KHP(KHC8H4O4) used in second titration = 0.003011 mol (Answer)
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax
