Q: Arrange the following substances in order of increasing boiling point. (lowest boiling point first)…
A: the boiling point of given compound are Boiling point of Cl2 = -34.6°C Boiling point of N2 =…
Q: e highest boiling point?
A:
Q: ОН HO.
A: Carboxylic acids have much higher boiling points than hydrocarbons, alcohols, ethers, aldehydes, or…
Q: What is the correct increasing order of boiling points of alcohols having the same number of carbon…
A: Alcohols :- The organic compounds having -OH (hydroxyl) group are called alcohols. Alcohols can be…
Q: Which liquid will have the highest boiling point? CH3Cl CH3OH C3H8 C6H14
A: Boiling point is directly proportional to molar mass .so if molar mass increases then boiling point…
Q: CH3CH2NHCH3 has lower boiling point than CH3CH2CH2NH2 True False
A: Introduction : The amines have boiling points higher than the alkanes but lower than those of…
Q: Predict the relative boiling points for the following molecules, putting the highest at the top of…
A: The given molecules has to be arranged from highest boiling point to lowest boiling point. The…
Q: Pick which of the following pairs will have the higher boiling point. Then, determine why. Pick the…
A: The given molecules are, Top molecule = CH3Cl (Chloromethane) Bottom molecule = CH4 (Methane)
Q: Place the following molecules in order from lowest boiling point to highest boiling point, according…
A:
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: Compounds with strong intermolecular forces have high boiling points. This is because they require…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: Given sets of compounds: CH4 CH3(CH2)2CH3 SiH4 CH4 CF4 CH4 We have to find the…
Q: Arrange the following molecules in order of lowest to highest boiling point.
A: Boiling is affected by various factors like molecular weight, intermolecular force of attraction…
Q: For each pair, which has the highest boiling point? 3. or or
A: The organic compound with higher molecular mass has a higher boiling point than the organic compound…
Q: highest to lowest boiling point.
A: The highest to lowest boiling point can be ranked
Q: Which of the two has a higher boiling point: Propanol or Pentanol?
A: Alcohols are generally soluble in water and they possess "intermolecular H-bonding". Alcohols…
Q: Which one of the following correctly ranks the compounds in order of lowest boiling point highest…
A: We have to arrange the following compounds according to high boiling point We have 3 carbon long…
Q: Place the following molecules in order from lowest boiling point to highest boiling point, according…
A: Boiling point of any molecule depends on 2 things ( in the same priority order as they are…
Q: Arrange each of the following in order of decreasing boiling points. Rank from highest to lowest…
A: Inter molecular forces (IMF) : there are mainly three types of IMF's present between molecules…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: The temperature at which the vapor pressure of a liquid becomes equal to the atmospheric pressure is…
Q: Can someone please explain how to determine boiling points. These need to be ranked in order of…
A: Boiling points are experimentally determined in the lab.
Q: Which of the following would you expect to have the higher boiling point? Explain why.
A: Welcome to bartleby ! Introduction : We have to tell the among given molecules which has higher…
Q: Arrange CH, CH, CHg in order of increasing boiling point temperature: CH4 C3H3 < C2H O CH, < CH4…
A: Boiling point depends upon the molecular mass of alkanes becouse if molecular mass of alkanes…
Q: Which of the following will have a higher boiling point? A. 0.001M C6H12O6 B. 0.001M C12H22O11 C.…
A: Colligative properties are the properties of solutions which having influence on the concentration…
Q: А. HO НО OH OH В. OH COH
A:
Q: 1. Question : Arrange the following compounds in increasing order of their boiling points, CH3CHO,…
A: Since, you have asked multiple questions we will solve the first question for you. If you want any…
Q: PLEASE provide an indepth explaniton as I will be using this to study
A: The boiling point will be high for the compound having strong weak intermolecular forces. If the…
Q: Order the following compounds in order of decreasing boiling point. (Start with the highest boiling…
A:
Q: Which one of the following would have the highest heat of vaporization? A) CH₃OH B) CH₂F₂ C) NF₃ D)…
A: Heat of vaporisation is affected by H- bonding. Heat of vaporisation is directly proportional to H-…
Q: Pick which of the following pairs will have the higher boiling point. Then, determine why. Pick the…
A: The molecule on top is an alcohol having intermolecular H-bonds. In order to boil and form vapour,…
Q: CH3CH2NHCH3 has lower boiling point than CH3CH2CH2NH2 (A) True B) False
A: CH3CH2NHCH3 is a secondary amine. CH3CH2CH2NH2 is a primary amine.
Q: A) Which would have a lower boiling point: O, or F,? Explain. B) Which would have a lower boiling…
A: O2 would have a lower boiling point than F2 , because F2 have stronger London attractions due to…
Q: Which of the following is expected to have the highest boiling point? A) CH₄ B) CH₃Cl C) CH₃OH D)…
A: Boiling point: The temperature at which the vapor pressure of a liquid becomes equal to the…
Q: 4. List the strongest force of attraction for each of the following and circle the structure with…
A: In any organic compound, there can be 3 types of molecular force of attractions. 1) Hydrogen bonding…
Q: Rank the boiling points of the molecules shown below from lowest to highest.
A:
Q: Arrange the following in order of increasing boiling point.
A: The boiling point depends upon the intermolecular forces present between the molecules. So we will…
Q: Put these in order of INCREASING boiling point. OH >
A: Boiling point of any organic compound depends on force exist between the molecule of compound more…
Q: Arrange the following compounds in order of decreasing boiling point. Briefly explain your answer.…
A: Step-1 The given compounds are named as: Ethoxyethane (an ether) 1,3-propan-diol (an alcohol)…
Q: Arrange the following molecules increasing order of boiling points. OH A В D E F
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: H2 CH3 CH3 NH2 CH H2 II III ČH3 CH3
A: Answer :- The order of increasing boiling point of given compound is II < III < I…
Q: Use the numbers 1, 2, and 3 to rank the boiling points of the three compounds melow from highest (1)…
A: As per our guidelines we can only solve first question. Please resubmit the other questions as next…
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: Which compound has the lowest boiling point? A)F2 B) I2 C) Cl2 D) Br2 E) They all have the same…
A: Boiling point increases with increase of mass.
Q: Arrange the following substances in order of increasing boiling point: CH₃CH₂OH, HOCH₂CH₂OH,…
A: The boiling point of a substance is the temperature at which the vapour pressure of the liquid is…
Q: Hexane has a smaller boiling point than nonane. True False
A: Boiling point :- The temperature at which vapour pressure of a liquid becomes equal to atmospheric…
Q: Which of the following has the compounds arranged in order of INCREASING boiling point? Select one:…
A:
Q: Arrange these compounds in order of increasing boiling point (values in °C are -42, 78, 138, and…
A: a) The compound (a) has one hydroxyl group –OH at the one end of the carbon chain; the interaction…
Q: Which of the following sets of compounds di we expect to have the same boiling point? ČI or
A: Boiling point means required energy to break the bond Boiling point increase as the attraction…
Q: Which of the following would you expect to have the highest boiling point? A. CH4 B.…
A: Answer :CH3CH2OH have highest boiling Point Explanation alcohol have High boiling Point as compare…
Q: Which of these substances would exhibit the highest boiling point at 1.00 atm? (A)…
A: The compounds given are CH3CH2CH2CH3, CH3CH2Cl, CH3OH and CH3CH2OH.
Q: For each pair of compounds listed, check the box next to the one with the higher boiling point.…
A: The molecule having higher intermolecular force of attraction is the one having the higher boiling…
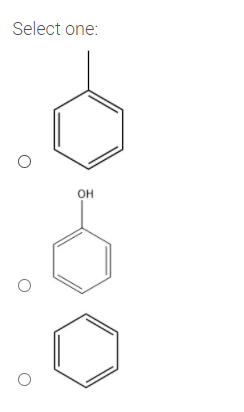
Which molecule below has the highest boiling point?
Step by step
Solved in 2 steps with 1 images

- What is the chemical formula of the following compound? Select one: a. C4H8O b. C3H8O c. C4H9O d. C3H6C3H8OO e. C3H7OH(1) Which of the following choices is the identity ROH? (2) Which of the following choices if the identity of CARBONYL?Please identify the correct ring flipped structure of compound XYZ.
- Rank the following groups in order of decreasing priority. −Cl, −CH3, −SH, −OHPlease help with row 2/question 2 ; the instructions for the specific column in row 2 are above row 1: Follow the instructions in each column. Hint for the last column: draw it with a wedge and again with adash – which gives the correct configuration?circle and name 4 functional groups



