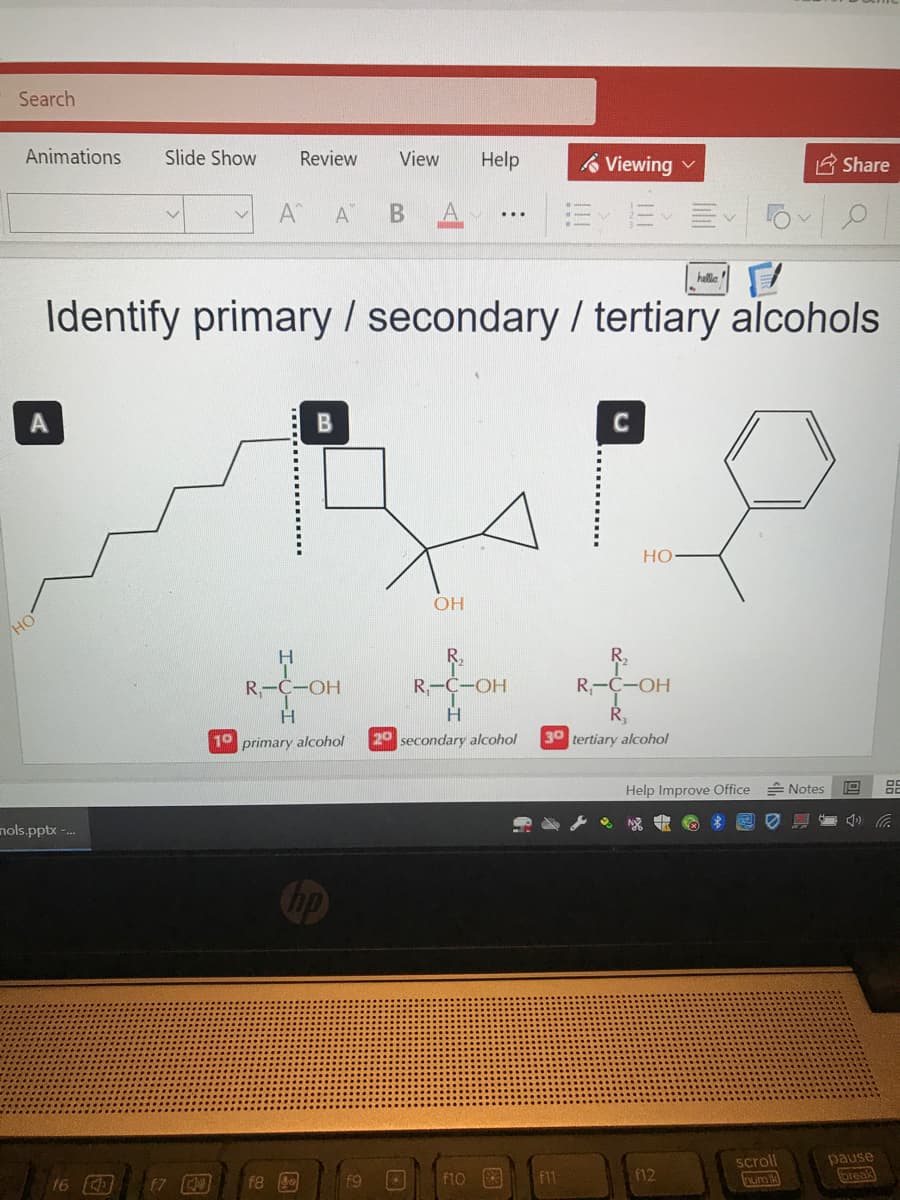
hllia entify primary /secondary / tertiary alcohols B C Но- OH R. R, R,-C-OH Ř, 30 tertiary alcohol R,-C-OH R-C-OH H. 10 primary alcohol 20 secondary alcohol Help Improve Office Notes
Q: a. Alcohol CH3-CH=C-CH3 180°C ČH3 b. Alcohol H,SO4 CH3-CH=CH2 180°C c. Alcohol H2SO4…
A:
Q: Which one of these molecules is the least soluble in water? CH3-CH2-CH2-CH2-CH2 OH CH3-CH-CH3…
A: The given compounds are alcohols. The alcohols are soluble in water as they form hydrogen bonding…
Q: Rank the following structures (A, B, C, D) from the (1) lowest to the (4) highest solubility in…
A:
Q: Question 1 Not yet answered Marked out of 2.00 P Flag question Correctly classify the following as a…
A: primary alcohol - primary alcohol is alcohol that is connected to primry carbon which is attached to…
Q: H PhH₂CH₂C PhH₂CH₂C H 14 .... CH3 4 CH3 mcpba mcpba ? ? HT/H₂O H™/H₂O ? ?
A: Organic reactions are those in which organic reactant react to form organic products.
Q: H2SO4 a. Alcohol CH3-CH=C-CH3 180°C ČH3 b. Alcohol H,SO4 → CH3-CH=CH2 180°C c. Alcohol H2SO4 →…
A: The species alkene is generated through the alcohol dehydration in the sight of the sulphuric acid…
Q: An unknown sample of alcohol is tested with Lucas reagent (ZnCl2 + HCI). An insoluble cloudy layer…
A:
Q: O Propauoic aad CHyCH2 COOH + H2D CH3 CH2 CO0 + Hg OT lag) [c] [c-ca] (x = degree of dissociation)…
A: 1) Dissociation of propanoic acid propanoic acid is a weak monoprotic acid, that dissociates weakly.…
Q: 3 2 PPM OH CI CH2-CH3 CH3-CH2-CH2-CH-OH CH3-CH-CH2-CH3 CH3-C CH2-CH3 CH3 CI
A:
Q: ethanol (C2H5OH) are present in 50.0 g-sample?
A:
Q: 2. Calculate the missing data in the following table. Compound AHvap (kJ/mol) ASvap [J/(mol·K)]…
A: Multiple question given here All solve with suitable explanation below step by step.
Q: (a) NaCN (d) NEt H2SO4, 0°C dropwise EINH3 + (b) MEOH (exs.) (e) TSOH, toluene heat 1) Ph3P Br (c)…
A:
Q: Identify primary / secondary / tertiary alcohols B C C,H5 H2 H2 H2 H,C-C-c-CHH,C-C- -c-CH3 H2…
A: Alcohols are those compounds which contains at least one hydroxyl (OH-) group attached with Carbon…
Q: Evaporation and Intermolecular Attractive Forces Complete the following data table. AT (°C) 10.47…
A:
Q: Classify the compounds as either more soluble in water or more soluble in nonpolar organic solvent.…
A:
Q: H3C CH,OH H,SO, CH3 H3C- H3C- H3C- H3C- OH H3C- OH H;C- OH OH 0-CH3 H3C-O -CH3 H3C-O -CH3 H3C-O A C
A:
Q: OM HBr+ 10.0 mL 1.0 M NaOH O M HBr + 10.0 mL 1.0 M CH3NH3*CH (pKb CH3NH2 1 O M HBr + 10.0 mL 1.0 M…
A: As we know, Acidic solution has pH less than 7. Basic solution has pH greater than 7 . While…
Q: OH он H3C*
A:
Q: Make the following changes on the molecule in the drawing area below: 1. First, make sure this is…
A: The solution is given below -
Q: 1.NANH:/liquid NH2/-78°C 1.NANH:/liquid NH:/-78°C A( C3H16) B(C19H36) H-CEC-H 2.1- Bromo-5-methyl…
A:
Q: Identify the compound with molecular formula C6H10O that gives the following DEPT 13C NMR spectrum:
A: From the NMR a) 1CH3 proton show peak around 28 ppm only 1 methyl group is present b) CH2 proton…
Q: HHH H-C-C-C-0- TABLE 3.8 Families of Organic Compounds HHH Name General Formula Family Example Name…
A: Name of the organic compounds
Q: Alcohols with two or more - OH groups have higher boiling point. I. Alcohols with more than two -OH…
A: The alcohols are the organic compound having atleast one OH group attach to carbon atom. For more…
Q: Which is likely to be more soluble in benzene C,H,? a) Circle one: NH, or CH,CH,CH,CH,CH,CH,Br b)…
A: Answer) The solubility of compound depends on the nature of bond present in the compound . Principle…
Q: For which of the following reactions should you use a trap to collect noxious gases?
A: Several organic reactions release harmful gases which are sometimes corrosive and sometimes very…
Q: Classify these alcohols as primary (1°), secondary (2°), or tertiary (3⁰). 1° 2° Answer Bank CH₂CH3…
A: If a carbon atom has an -OH group surrounded by one carbon, it is a primary alcohol. If a carbon…
Q: 11. Construct a model of 1-butanol: CH-CH-CH-CH:OH. See page 10 of the instruction booklet for an…
A: The given molecule is 1-butanol, CH3-CH2-CH2-CH2-OH, which has a total of 4 C atoms.
Q: ÇOOCH3 CH3. (CH)2СHNH2, CH;OH, A 1. КОН, Н2О 2. H*, H,O CH;CH, COOCH3 1. ether, 20 °C 2. H", Н,О…
A: There are some reactions which are shown above . we have to draw the product of the above reactions…
Q: 22. Name the following common substances and identify if they are organic compounds. a b. H H-N-H H…
A: The answer to the following question is-
Q: Make the following changes on the molecule in the drawing area below: 1. First, make sure this is…
A: The solution is given below -
Q: A :0-H :0: :0: H-ö: CH-CH2-CH2 CH-CH2-CH2 H3C H3C
A:
Q: For each of the three horizontal series below, rank each molecule from highest (1) to lowest (4)…
A: Answer Series 1: Generally boiling point of alcohols are increase with increasing molecular mass, In…
Q: Which of the following substances is suitable for Dumas method to determine its molar mass? a.…
A: Correct option is (d) propane
Q: Part 2 What is he molar mass of acetone? Pournd your ansuel 10 four significant figures. Part 3 what…
A: 2) Given: Acetone i.e. CH3COCH3.
Q: WRITE YOUR ANSWERS ON THE FOLLOWING NUMBERS Sample Answer 1. BEL B. D. K HCOOCH.CH 1.oil of A ethyl…
A: The answer of questions 3 and 4 is given below.
Q: Which alcohol burned with the most yellow flame? O methanol ethanol propanol O they were the same
A: On the basis of no of carbon and burning efficiency we figure out this problem very easily.
Q: G. Nomenclature of Alcohols: Critical Thinking Question (CTQ), alcohols (use IUPAC rules indicated…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: positive and negative impact of alcohol chemistry in socitey
A: Positive impact of alcohol in the society : Alcohol is used for formation of many products which are…
Q: Hello I am having trouble with my homework. Can you help and explain?? It is due tonight. My…
A:
Q: Phenol Water Phenol mole %phenol ar) T °C mol mol fraction gr gr 10 4 35 10 46 10 6 50 10 8 58 10 12…
A: Topic -- Finding the value 1. Moles of phenol 2. Moles of water 3. Mole fraction of phenol…
Q: H H CH3 H;C CH3 H;C CH3 CH3
A: Protonation of alkene Carbocation rearrangement (formation of more stable carbocation intermediate)…
Q: 14.3 Matching Questions Select the correct name for the following. 75) A) Propan-3-one CH3CH2C…
A:
Q: Rank the following compounds according to increasing solubility in water. 1 CH-CH-CH-CH; II…
A: Organic compounds are mostly made up of carbon, hydrogen, oxygen, nitrogen atoms. Organic compounds…
Q: 1. Hg COAC)2 , M20 HO a) 2. NaBHly, NAOM major product b) 1) O3 2) (CH3)2S C10H18
A:
Q: Identify the least soluble in water? A. PEG B. Acetone C. Glycine D. Ethyl Ether
A: Out of given compounds, ethyl ether is less soluble in water Reason:- It is because it has oxygen…
Q: Use the information provided to determine H°rxn for the following reaction: CH4(g) + 4Cl2(g) —>…
A:
Q: Classify these alcohols as primary (1°), secondary (2°), or tertiary (3⁰). 1° 2° CH₂CH3 OH CH,CCH₂OH…
A:
Q: What is the name of the structure shown below? 0 1,1-dimethylcyclobutane 2,3-dimethylcyclobutane O…
A: In order to write iupac nomenclature we have to follow some rules of iupac 1) first select longest…
Q: Identify primary / secondary / tertiary alcohols H. H Н ОНН TI T H-C-C-C-H II I Η Η Η B C но HH H…
A: We have to identify primary, secondary, and tertiary alcohol.
Q: OH 1) xs LIAIH4, ether 2) H+, H2O b) H,C H3C CI pyridine a) H3C NH2 H,C OH OH 1) xs CH3MgBr,ether +…
A: LiAlH4 reagent is reducing agent which can reduce amide to amine.
Step by step
Solved in 2 steps with 2 images

- Acid catalyzed hydration of alkenes except ethene that leads to the formation of which among the following alcohols? I secondary and tertiary alcoholsII Mixture of secondary and tertiary alcoholsIII Secondary or tertiary alcoholIV Primary alcoholWhich of the following compounds will be most soluble in toluene(C7H8)? A) ethylene glycol (HOCH2CH2OH) B) hexane (CH3CH2CH2CH2CH2CH3) C) acetone (CH3COCH3) D) trimethylamine (N(CH3)3) E) A and D are soluble in toluene.HURRY ASAP I WILL RATE NO NEED FOR EXPLANATION WHICH OPTIN Which of the following statement(s) about the alcohols of which their structures/IUPAC names are given below is/are true? I. CH3-CH(OH)-CH2-CH2-CH(CH3)-CH2-CH3 II. CH3-CH(OH)-CH2-CH2-CH2-CH3 III. Cyclohexanol IV. (CH3)2-C(OH)-CH2-CH2-CH3 V. CH3-CH2-OH The IUPAC name of compound I is 3-methyl-6-heptanol. Compound V is a secondary alcohol Compound III is primary alcohol. Compound IV is tertiary alcohol Compounds I and II are primary alcohols
- In which of the reactions given below is the product pyrimmer alcohol? A. I and III B. Solo II C. II and III D. I and II E. Solo IChoose the correct steps in the dehydration of secondary alcohol; then, arrange inchronological order.I - Formation of AlkeneII - Formation of AldehydeIII - Formation of Protonated alcoholIV - Undergoes E2 mechanismV-Undergoes E1 mechanismA. V-III-IB. IV-III-IIC. III-V-ID.III-IV-II Ethanol is heated with an acidified potassium manganate (VII) solution. What would be theproduct of this reaction?A. EthanalB. EtheneC. Ethanoic acidD. Ethane ALL of the following Ether can be prepared by Williamson's method, EXCEPT:A. Ditertbutyl etherB. Ethyl-tert-butyl etherC. anisoleD. Both A & B. Which of the following is not true of SN1 reactions?A. They are favored by nonpolar solvents.B. The concentration of nucleophiles does not affect the rate of reaction.C. Tertiary alkyl halides generally react through this mechanism.D. They occur through a 2-step concerted reaction. Butan-1-ol is formed from which of the following reactant-reaction pairs?A. Butanal - reduction reactionB. Butanone -…#B: Methyl acetate has methoxy, -OCH3 as the remaining of the alcohol part in the ester. Isopropyl acetate has isopropoxy, -OCH(CH3)2 as the remaining of the alcohol part in the ester. -OCH(CH3)2 is more electron donating than the methoxy, -OCH3 group due to the presence of two electron-donating -CH3 group in the former. Hence saponification reaction of Isopropyl acetate is much slower than methyl acetate. Hence the rate of saponification of methyl acetate, CH3CCO2CH3 is 50 times greater than that for isopropyl acetate.
- What products would you expect to obtain from the oxidation of thefollowing alcohols with CrO3? a. Cyclohexanol b. 1-hexanolAlcohols A and B formed layers with Lucas reagent within 10 minutes. However, compound A formed brown precipitate with KMnO4 while compound B did not. Which are the possible identities of compounds A and B? a. isopropanol and tert-butanol b. ethanol and methanol c. propanol and tert-butanol d. isopentyl alcohol and isopropanolAbsolute ethanol (100% ethanol, with no water) cannot be obtained through simple fractionaldistillation because of the lower boiling azeotrope that forms with water. Propose a method by which onemight prepare absolute ethanol
- IV. In every chemical reaction, include the IUPAC name and draw the structure of ALL organiccompounds present.A. Give the reacton on how to convert the following organic compounds. 1. N-Phenylpropionamide to alcohol 2. N-ethylbutylamide to alcohol1. (a) Which compound would have the highest boiling point? benzophenone toluene acetophenone benzaldehyde (b) Which of the reagents listed below can convert a primary alcohol to an aldehyde? H2CrO4 DMSO, (COCl)2, Et3N AgNO3 KMnO4-, NaOH (c) Oxidation of alcohols involving the use of DMSO and (COCl)2 is also known as which of the following? Jones Tollens Baeyer-Villiger SwernNaphthalene, C10H8 is insoluble in water but sublimes easily when heated. How could you separate a mixture of naphthalene and NaCl? Suggest two different methods. asap

