Which is likely to be more soluble in benzene C,H,? a) Circle one: NH, or CH,CH,CH,CH,CH,CH,Br b) Circle one: H,0 or CH,CH,CH,CH,CH,CH,CH,CH,OH c) Circle one: NaCl or CCI, d) Circle one: or CH,OH
Which is likely to be more soluble in benzene C,H,? a) Circle one: NH, or CH,CH,CH,CH,CH,CH,Br b) Circle one: H,0 or CH,CH,CH,CH,CH,CH,CH,CH,OH c) Circle one: NaCl or CCI, d) Circle one: or CH,OH
Chemistry for Today: General, Organic, and Biochemistry
9th Edition
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Chapter13: Alcohols, Phenols, And Ethers
Section: Chapter Questions
Problem 13.19E
Related questions
Question
Help 2
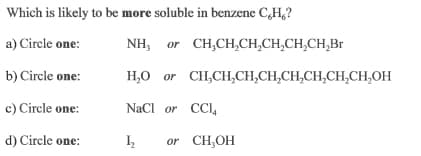
Transcribed Image Text:Which is likely to be more soluble in benzene C,H,?
a) Circle one:
NH, or CH,CH,CH,CH,CH,CH,Br
b) Circle one:
H,0 or CH,CH,CH,CH,CH,CH,CH,CH,OH
c) Circle one:
NaCl or CCI,
d) Circle one:
or CH,OH
Expert Solution
Step 1
Answer) The solubility of compound depends on the nature of bond present in the compound . Principle of solubility states that "Like dissolve Like" Means polar compound dissolve in polar compound and non polar solvent dissolve in non polar solvent.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning