Classes Of Functional Groups
Organic Chemistry deals mostly with carbon and hydrogens, also called hydrocarbons, but those groups which replace hydrogen and bonds with carbon to give a characteristic nature, unique of their own, to the hydrocarbon they are attached to, are called functional groups. All the compounds belonging to a functional group undergo reactions in a similar pattern and are known to have similar physical and chemical properties.
Characteristics Of Functional Groups
In organic chemistry, we encounter a number of special substituent groups which are attached to the hydrocarbon backbone. These groups impart certain characteristics to the molecule of which it is a part of and thus, become the highlight of that particular molecule.
IUPAC Nomenclature
In Chemistry, IUPAC stands for International Union of Pure and Applied Chemistry which suggested a systematic naming approach for the organic and inorganic compounds, as in the beginning stage of nomenclature one single chemical compound was named in many ways by which lead to confusion. The need for this approach aroused as the number of chemical compounds newly discovered were increasing (approximately 32 million compounds) and the basic concept of nomenclature i.e. the trivial nomenclature and the derived system of nomenclature failed to overcome the challenge. It is an important task to name a chemical compound systematically and unambiguously which reduces lots of confusion about the newly reported compounds.
Write the IUPAC and, where possible, a common name for each compound.
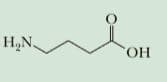
A systematic name is given to an organic compound by IUPAC nomenclature. The set of rules which are used to name any compound are:
- Select the longest carbon chain containing the functional group or the unsaturated bond. This chain is known as parent chain.
- Identify all the substituents which are attached to the parent chain. These form the side chains.
- Number the carbon atoms of the parent chain such that each of the substituent gets the lowest possible number. When two or more side chains have same positions than the lower number is assigned according to the lowest sum rule. The positions numbers are added to get the lowest possible sum.
- When two functional groups are present than priority order is considered. The group which is present at the top forms the suffix and bottom group is taken as the prefix.
- When the same type of substituent is present more than one in number, the prefixes di-, tri-, tetra-, penta- are used.
- When more than one substituent is present, the names of these substituents are written in the alphabetical order.
- Commas are used in between numbers and dashes are used in between the letters and numbers. There is no space in writing the name of a compound.
- In general, the names of substituents are written in alphabetical order followed by the base name of the parent chain.
Step by step
Solved in 2 steps




