Q: Which of the following is capable of producing longer alkyne products? a Internal alkyne with…
A: We have to tell among the given options, which one is capable of producing longer chain alkyne…
Q: In a closed vessel, 3.2 grams of carbon monoxide reacted with 14.6 grams of 02 Based on the given…
A: For the reaction - CO + 1/2O2 ---> CO2 Mass of CO = 3.2 g Mass of O2 = 14.6 g Thus , 3.2 g CO…
Q: What is the energy change when the temperature of 12.4 grams of solid silver is decreased from 37.3…
A: Given: Mass of solid silver = 12.4 g Initial temperature = 37.3oC Final temperature = 21.7oC Known:…
Q: Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are…
A:
Q: A 210-gram aluminum block at a temperature of 55°C is placed into an insulated container with 360…
A: The final temperature of the metal and ice is needed to be calculated if 210 g of aluminum block at…
Q: Balance the following chemical equation. Show yc Fe2O3(6) + CO → Fe) + CO2(g)
A:
Q: Given is teh mass spectogram of an unknown compoound. This unstable cyclic compund is a colorless…
A: Note : The compound name is cyclohexene. Cyclohexene on retro Diels Alder reaction produces the m/z…
Q: The age of wine can be determined by measuring the trace amount of radioactive tritium, 3H, present…
A: N(t)=N0 exp[-k t]N(t) is the number of radioactive nuclei remaining at time t, N0 is the initial…
Q: At 725 K, AG° = -21.6 kJ/mol for the reaction A (g) + 2 B (g)3 C (g). If the partial pressures of A,…
A: Given temperature is 725 k
Q: 2. Touprofen, the anit-inflammatory ingredient in Advil, has the formula C13HIBO2. a. How many…
A: We are authorised to solve only first three subparts of the question. Please post rest of the…
Q: A student performed titration of an unknown reducing substance (molar mass 200. g/mol) in a sample…
A: Answer: This question is based on the calculation of molar concentration of unknown sample with the…
Q: Consider the reaction: 2H,0,(1)-2H,0(1) + 0,(g) Using standard absolute entropies at 298K, calculate…
A: Number of moles of H2O2 = 2.30 mole
Q: Answer 1,2,3,4,5. Answer ad Show solution. No need to discuss each step of the solution. Keep it…
A: (1) PbO2 + HBr ---> PbBr2 + O2 + H2O Here Oxidation state of Pb decreases from +4 to +2 , thus…
Q: e allowed to react in a vessel maintained at a temperature of 300 K and a pressure of 10ʻPa. ) Write…
A:
Q: At a certain temperature, K = 0.914 for the reaction NO2(3) + NO(5) N20(s) + O2(s) Amixture was…
A: Given : Kc = 0.914 Moles of each gas = 0.200 mol Volume of vessel = 5.00 L
Q: What is the major driving force in chromatographic separation? difference in affinity with…
A: Please find your solution below : Chromatography is an analytical technique used for the separation…
Q: Which of the following set of reagents will yield a vicinal diol when reacted with an alkene? a…
A: ->Vicinal diol is Compound in which -OH group attached at adjacent carbons.
Q: 1. Calculate the pH of a pyridine buffer that contains 500.0 mL of 0.4214 M pyridine and 500.0 mL of…
A: Here we have to determine the pH of 500 ml solution containing 0.4214M and 0.4082M pyridinium . Kb…
Q: Molecular vibration happens when an atom or molecule absorb IR radiation.
A:
Q: Pulmonary surfactant, a substance critical for allowing the lungs to fully inflate, is made of this…
A: To answer for question 2
Q: For the gas phase decomposition of nitrogen dioxide at 383 °C 2 NO,2 NO + 02 the average rate of…
A:
Q: Which of the following describes a gradient elution process? O stationary phase has varying…
A: We have to describe a gradient elution process. We know that in a gradient elution process, the…
Q: 6. For each numbered step in the qualitative analysis scheme in number 5 above, type all net ionic…
A: When we are constructing net ionic equations, we have to keep few points in our mind: Only salts in…
Q: Question 27 Use the following chromatogram of standard amino acids: 24 11 16 19 21 12 19 ,8 9 10 23…
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: Question 31 Use the following chromatogram of standard amino acids: 24 11 16 19 21 3 12 15 23 68 9…
A: A multiple choice question based on analytical separations that is to be accomplished.
Q: Which of the following conditions DOES NOT show an improvement in the elution process? O increased…
A: Elution is a separation method that uses a stream of gas or liquid moving in the opposite direction…
Q: Which of the following factors DOES NOT affect retention time of a sample undergoing normal phase…
A: Retention time is the time period where a sample spend on column. Here we have to tell which factor…
Q: OH А. HOʻ С. D. H B. A.
A: Since we know that Tollen's test given by aldehyde... Tollen's reagent is known as silver ammonical…
Q: What volume of each of the following acids will react completely with 25.00 mL of 0.200 M HCl? a.…
A: We have to find volume of base that react with 25.00 mL of 0.200 M HCl a. 0.100 M NaOH b. 0.0500 M…
Q: please answer all 3 questions asked
A:
Q: An infrared spectrophotometer contains a grating. O True O False
A:
Q: The table shows the data obtained for the separation of the components Methyl acetate (A), methyl…
A: To calculate number of plates for each compound.
Q: Vhat kind of reaction should you subject it to in order to become 2-methylpentan-3-ol? A Hydration B…
A:
Q: Question 3 how do you call the analysis technique done by identifying the fragments corresponding to…
A: Mass spectrometry is the branch of chemistry in which we deal with deal with molecular ions,…
Q: Which of the following statements is NOT true about halohydrin formation from alkenes? A It adds-X…
A: * In halohydrine reaction alkene act as reactant . * It results in formation of halohydrin i.e a…
Q: What is the class of organic compound that is produced when a carboxylic acio derivative undergoes…
A: The class of organic compounds that is produced when a carboxylic acid derivatives undergoes…
Q: Arrange the following sets of organic compounds according to decreasing solubility in water 1.…
A: Alcohol can be defined as organic compound in which carbon atom is attached with OH group.
Q: What is the IHD of a chemical compound with a formula of C6H5NO2? a 4 b 5 c 6 d 7
A: IHD stands for Index of Hydrogen Deficiency. In a hydrocarbon with no double bond or rings, the no…
Q: Question 16 In a reverse phase chromatography setup, which of the following compounds will have the…
A: Highest retention time is nothing but time spent by an eluent in column
Q: Question 2 Which amomg the fragments below will be dectected by mass Spectrophotometer? i. [CH3CH31*…
A: Given that :- [ CH3CH3 ]+ [ CH3CH3 ] [ •CH2CH3 ] [ CH3CH4 ]-
Q: 28-31. Supply the missing value. n 1.55 1.65 1.70 1.80 ??? 1.62 Conc. 10% 15% 25% 35% 22% ???…
A: A question base on analytical process that is to be accomplished.
Q: How does the common-ion effect influence the pH of the following solution? Please explain in detail.…
A:
Q: Table sugar granules were dissolved in a glass of water. The solid sugar granules have no measurable…
A: Given that - Mole fraction of water, solvent = 0.75 Mole fraction of Solute = 1-0.75 = 0.25…
Q: 1. What volume of cefoxitin (25 mg/ml) must be added to 400 ml of Mueller-Hinton broth to yield a…
A:
Q: At a certain temperature, the reaction 2HF(g) 2 H2(g) + F2(g) has K. = 1.2 × 10-13. Does this…
A:
Q: Please give a solution to number 2-5 suppose the pharmacist was tasked again obtained an analytical…
A: the solution is shown below.
Q: 62. A 0.25g of a certain substance is dissolved in 60g of water. The freezing point of the solution…
A: Answer: This question is based on the colligative property of the solution where when a non-volatile…
Q: What would be the concentration of X 2- if a 10 mL of a 1 M solution of KOH was added to 50 mL of a…
A: The chemical reaction for the neutralization of a diprotic acid H2X with KOH is needed to be…
Q: Given is teh mass spectogram of an unknown compoound. This unstable cyclic compund is a colorless…
A: The compound name is cyclohexene. Cyclohexene goes retro aldol produces the m/z = 54.
Q: How many grams of NH3 can be produced from 40.0 grams of CaCN2? CACN2 + 3H20 CACO3 + 2 NH3
A:
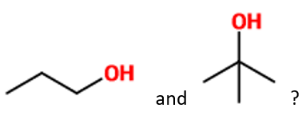
Which of the following tests would best differentiate
| a |
Lucas Test |
|
| b |
2,4-DNPH Test |
|
| c |
Iodoform Test |
|
| d |
Fehling's Test |
Step by step
Solved in 2 steps with 1 images


