Q: 180 (a) 160 (b) (c) 140 120 OL 100 PPM 80 60 @ 40 20 0
A:
Q: For a 1.0 x 10-4 M solution of HCIO(aq), arrange the species by their relative molar amounts in…
A: Given: Concentration of HClO = 1.0×10-4 M
Q: 3.40 Draw the Fischer projection for the oxidation and the reduc- tion products of D-ribose. What…
A:
Q: For each system listed in the first column of the table below, decide (if possible) whether the…
A: We have to what happened to the entropy of the system when the following changes are made.…
Q: The K₂ of a monoprotic weak acid is 0.00542. What is the percent ionization of a 0.134 M solution of…
A:
Q: m 24) When 1.75 g of CaCl2 dissolves in 125 g of water in a coffee-cup calorimeter, the temperature…
A:
Q: Predict the products of each reaction below. Indicate regiochemistry and stereochemistry when…
A: In the first reaction reduction of alkene take place.
Q: Assuming equal concentrations, arrange these solutions by pH. Sr(OH)₂ (aq) HF(aq) KOH(aq) Highest pH…
A: Hydronium ion concentration is an important parameter of an aqueous solution. The concentration of…
Q: When 77.6 g of benzamide (C₂H₂NO): are dissolved in 1250. g of a certain mystery liquid X, the…
A: Mass of benzaldehyde = 77.6 gram Mass of solvent = 1250g ∆T = 5.4°C i for KBr = 1.84 To find…
Q: What reaction corresponds to the heat of formation of solid Mg(NO3)2
A: The heat of formation: When one mole of the substance is formed by its constituent elements in its…
Q: 13.36 Identify each of the following as the a or ß isomer: а. CH₂OH b. CH₂OH Н HO Н ОН ОН Н Н ОН Н…
A: Anomers are two sugar that differ in the configuration only at the carbon that was carbonyl carbon…
Q: Provide the correct systematic name for the compound shown here. t
A: Nomenclature of organic compounds.
Q: 13.22 Identify each of the following as the D or L enantiomer: а. Н. 0 b. CH2OH HO H - Н- с -Н OH ОН…
A: D enantiomer is the one in which -OH on last chiral carbon is onto right side and L enantiomer is…
Q: The following chemical equation is balanced. Ca(NO3)2 + Na₂CO3 CaCO3 + NaNO3 O True False
A:
Q: Part A At 20 ∘C and a partial pressure of 76 mmHg, the solubility of CO2 in water is 0.169 g/100mL.…
A: Part A - Given that - Temperature = 20°C Initial pressure P1 = 76 mmHg Final Pressure P2 =…
Q: Balance the following reaction. Place a digit in the order from left to right. If a component has no…
A:
Q: what are compounds in a sentence?
A: A compound clause or sentence is a clause that joins two independent clauses, usually using a…
Q: T Al(s) + AlF3(aq) + NaClO3(aq)- NaCl(aq) + O2(g) ZnS(s) + Zn3P₂(s) + Ag2S(s)→ Ag(s) + S8(s) 10…
A: Balancing chemical equations involves the addition of stoichiometric coefficients to the reactants…
Q: This is the chemical formula for nickel tetracarbonyl (a powerfully poisonous liquid used in nickel…
A:
Q: Re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in…
A: To solve this problem we have to arrange the atoms or ion in decreasing size .
Q: ) What is the pH of water in equilibrium with 0.040 ppm SO2 gas? The total air pressure is 1.02 atm.…
A: #3: (a): Given the concentration of sulfur dioxide, SO2 in air = 0.040 ppm 1 ppm = 106 parts =>…
Q: In a voltaic cell cations go to the The electrons will go to the and anions go to the
A: We have to fill the missing boxes in the given statement
Q: A pure substance is represented in the particle diagram. On the following cooling curve for the…
A: There are three phases of matter. The phase of matter can be interchanged by changing the…
Q: Suppose the galvanic cell sketched below is powered by the following reaction: Mn(s) + Cu(NO3)(aq)…
A:
Q: How many types of bonds are present in ethane? In ethene? In ethyne? What type of isomers is shown…
A: Types of bond Type of isomers in butane Type of isomers in C2H6O
Q: What is the expected hybridization of the central atom in a molecule of Til4? This molecule is…
A: he have to determine the hybridization of TiI4
Q: When a metal at a higher temperature is transferred to water at a lower temperature, heat the value…
A: Heat flows from hot object to cold object until they reach at same temperature. A calorimeter is…
Q: A radioactive element has a half-life of ln(2) weeks. If e³ tons are present at a given time, how…
A:
Q: What is the pH of a 9.1x10-4 M aqueous solution of HNO₂? Answer:
A: Here we are required to find the pH of the solution.
Q: non-standard state of water vapor is AGm(100.0 °C, 1 bar pressure) = -225.16 kJ/mol. What is the…
A: Here non standard state of water vapour have free energy formation-225.16Kj/mol at 100°C and 1bar…
Q: Can you explain the IUPAC name that best helps understand Chloroacetophenone structure?
A: We have to explain the IUPAC name of the chloroacetophenone. Introduction: Initially we have to…
Q: A given molecule has 2 degenerate states that are 700 cm³¹ (wavenumbers, 1/2) above the lowest…
A: (a) It is given that a molecule has 2 degenerate state that are 700 cm-1 above the lowest energy…
Q: A piece of plastic at 182 °C was put in a beaker with 135.6 g ethanol at 20.3 °C. The temperature of…
A: When plastic at higher temperatures was put in ethanol ar lower temperature , then plastic will lose…
Q: Please explain how you could determine whether a particular molecular compound was polar if you were…
A: The covalent bond is formed by the sharing of electrons between the atoms.
Q: The pressure in Denver, Colorado averages about 632 mm Hg. How many atmospheres is this?
A:
Q: which has more calories : table sugar or aspartame ? what does synthetic gas (water gas) consist of…
A: Aspartame is an artificial non saccharide sweetener, while table sugar is a natural disaccharide…
Q: For a particular reaction, AH = -111.4 kJ and AS = -25.0 J/K. Calculate AG for this reaction at 298…
A:
Q: A student requires 60.0 mL of a solution of potassium permanganate with concentration 2.45 M for an…
A: Molarity (M) is a concentration term for a solution. The molarity of a solution is defined as the…
Q: Assign oxidation states to all of the species in the following redox reaction. For the reactants,…
A: Reducing agent is the species that reduces the other substance and itself gets oxidized. Oxidizing…
Q: approximate values of bond angles marked "a", "b", and "c", respectively? H :O: HIC C H-N: H b…
A: S character is directly proportional to bond angle
Q: In the process of fractional distillation, a mixture of hydrocarbons is separated into its fractions…
A: To solve this problem we have to know about fractional distillation process .
Q: A student mixes 41.8 g of ethanol at 15.8°C with 50.7 g of water at 49.2°C. If the resulting…
A: To find the answer, just compare the heat that ethanol gains to the heat that water loses. Water…
Q: Workshop 10: Stoichiometry and Limiting Reagent In chemistry we represent chemical reactions using…
A: The reactant which is present in lesser amount in the chemical reaction and which guides the amount…
Q: 3 % Transmittance 100 50- 0- 4000 singlet, 3H (a) triplet, 2H. (b) 3000 2000 2 1500 Wavenumber…
A: Molecular formula - C7H14O
Q: Substance CO(g) O₂(g) CO₂(g) c. d. 1.22 x 1045 8.23 x 10-46 e. 7.48 x 1068 AHfᵒ (kJ/mol) ? 0 -393.5…
A: Given reaction: CO(g) + 12O2(g) ⇌ CO2(g) We have to calculate the value of K for the production of…
Q: Using the following data at 528 K: (Points are equally distributed) (1) A(s) + B(g) ×10² (2) 4 C(g)…
A: Value of equallibirium constant can be given as:
Q: Sulfur dioxide (SO2) is a by-product of burning coal and contributes to the formation of acid rain.…
A:
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A:
Q: What is the solubility of AgCN(s) (Ksp = 2.2 x 10-16) in a solution containing 0.19 M H? (Ka for HCN…
A: Given: Ksp = 2.2×10-16 H+=0.19 M Ka for HCN = 6.2×10-10
Q: Re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in…
A: Size decrease as move across period from left to right. Size increases as we move down the group.…
Draw the major product of the following reaction sequence.
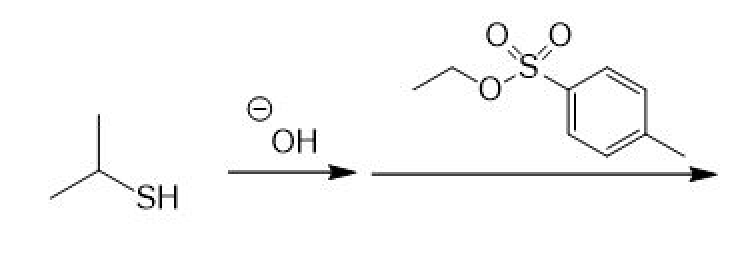
Step by step
Solved in 3 steps with 2 images


