World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter2: Matter
Section: Chapter Questions
Problem 7STP
Related questions
Question
Transcribed Image Text:ho
<CHE154 S20 Ch19 Sec8-10
Ay
Chapter 19 Multiple Choice Question 48
Review I Consta
Part A
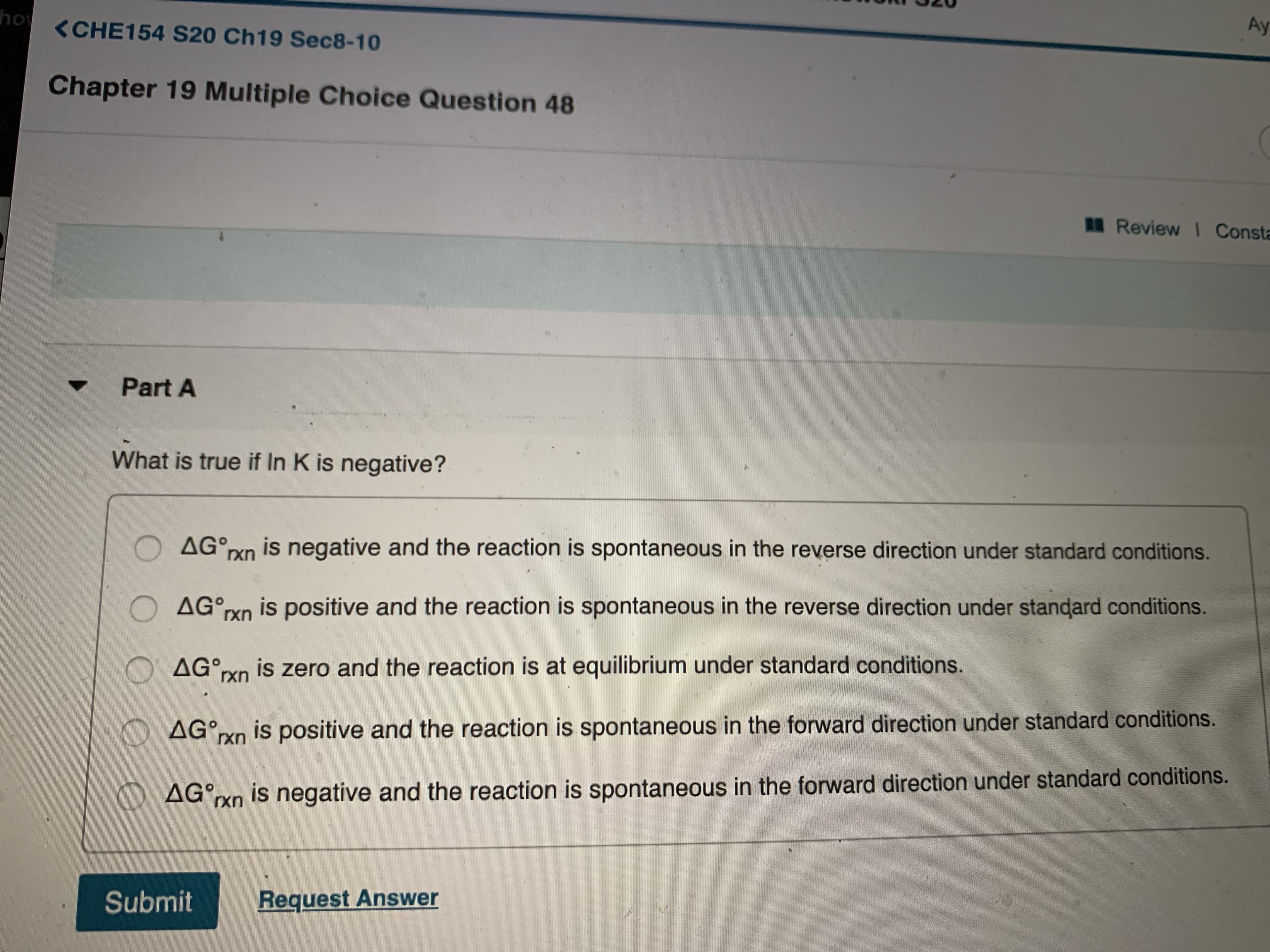
What is true if In K is negative?
O AG°rxn is negative and the reaction is spontaneous in the reverse direction under standard conditions.
O AG°rxn is positive and the reaction is spontaneous in the reverse direction under standard conditions.
AG°rxn is zero and the reaction is at equilibrium under standard conditions.
AG°XN is positive and the reaction is spontaneous in the forward direction under standard conditions.
rxn
AG°
AG°rxn is negative and the reaction is spontaneous in the forward direction under standard conditions.
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning