Q: Starting with an appropriate alkyl halide and base, outline syntheses that would yield each of the…
A: In this question, we will synthesized the these given alkenes products from the starting material (…
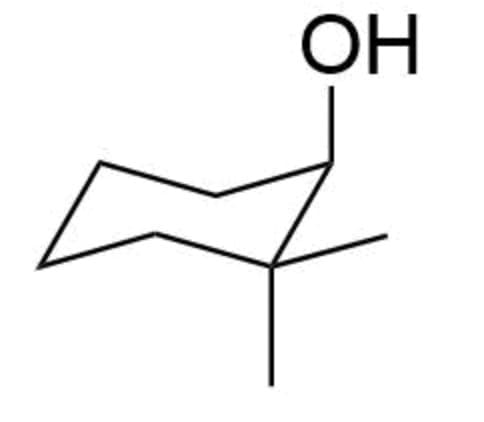
Q: Question 18 Name the following compound: nem Question 19 Name the following compound:
A: The given molecules are esters and to write the name of a ester first of all we number the carbon at…
Q: b. Which is a stronger base?
A: Bronsted acid-base theory: Bronsted acid: proton (H+ ) ion donors are called Bronsted acid Bronsted…
Q: ОН (i) HC (її) ОН HC (iii) ОН
A: Acid is a compound which gives H+ ion.Generally, carboxylic acids are more acidic than phenol. The…
Q: J How many different kinds of protons are present in each of the following compounds? OH Multiple…
A:
Q: 4. Out of the following metallic cations in solution, which would precipitate last when Na2S(s) is…
A: The solubility product is an important parameter to determine the stability of an insoluble…
Q: Which of the following elements has no unpaired electrons in its ground state? Select one or more:…
A: Hund's rule states that degenerate orbitals (same energy orbitals) are each occupied by one electron…
Q: Classify the Carbon (marked with a *) as being 1o, 20, 30. State a reason for your classification.
A: In this question we have given an organic compound and we have to mark primary, secondary and…
Q: Given: Colloid Type CEC cmolc/kg pH= 7 Organic matter 200 Vermiculite 150 Smectite 100 Mica…
A: To calculate the total cation exchange capacity (CEC) of the soil, we need to consider the…
Q: Stereoisomers of α-bromocinnamic acid: draw the reaction mechiansim with arows for the…
A: A question based on reaction mechanisms. Reactants and products involving dehydrobromination is…
Q: Oxygen gas can be produced by the decomposition of solid NaNO3. The equation for this reaction is:…
A: Mass of O2 produced = 124 gMass of NaNO3 required = ?
Q: a. As b. Cr c. Br d. Sr Write the following full and noble gas electron configurations:
A: To solve this problem we have writte the full and Nobel gas electronic configuration.
Q: What is the product of the following sequence of reactions? Cl. I A) I NaOH 11 B) II C) III NaNH,…
A: When 1,2-dichloroethane is treated with alcoholic NaOH then dehalogenation takes place and we get…
Q: For the reaction: C₂H₂(g) + 50₂(g) → 3CO₂(g) + 4H₂O(g) Part 1 of 2 Predict the enthalpy of reaction…
A:
Q: When 3.49 g of a nonelectrolyte solute is dissolved in water to make 935 mL of solution at 23 °C,…
A: mass of nonelectrolyte solute = 3.49 gVolume of the solution = 935 mL = 0.935 LOsmotic pressure (π)…
Q: (19) НО (20) с 0 ОН OCH 3 1. 2. H3O+ CI Pyridine ОН одо
A: In the first reaction, alcohol is given. As a reagent pyridine base is given.The given alcohol will…
Q: The synthesis of maleic acid anhydride (CH:O) can be accomplished by reacting benzene (C.H.) and…
A: 2 C₆H₆(l) + 9 O₂(g) → 2 C₄H₂O₃(s) + 4 CO₂(g) + 4 H₂O(g)mass of benzene (C₆H₆) reacts = 45.8 gmass of…
Q: b. Consider the following molecule and use it to answer the two questions (i. and ii) that follow. H…
A: Unsaturated molecules are those which contains atleast one double or triple bond and saturated…
Q: ) Indicate the number of bonds in the shown molecule which would would release energy when…
A: i) there are two bonds in the molecule which would would release energy when hydrolysed.ii) the…
Q: Convert 8.9 g/mL to its equivalent in kg/m^3. Round the answer to the nearest unit.
A: Using unit conversion method we can convert 8.9 g/mL to kg/m3 unit.
Q: 6. Write the long form electron configuration for phosphorous
A:
Q: I got -1366.7 but I don't think that's right.
A: The standard enthalpy of a reaction is given by:It is the difference between the sum of standard…
Q: a) b) c) H.C d) H2C H3C CH3 and CH₂ CH3 and CH3 CH3 and CH3 H3C H3C -CH3 CH3 and CH H3C. CH3 H3C CH3…
A:
Q: Synthesis. Design a synthetic route to perform the transformations shown below. You may use any…
A: The reaction scheme involves the conversion of a carbohydrate called erythrose to form erythronic…
Q: Which of the following molecules is the major product of the reaction shown in the box? C
A: The reaction of benzene with acetyl chloride (CH3COCl), aluminum chloride (AlCl3), and water (H2O)…
Q: Provide the correct IUPAC names for the following compounds and state the function group present: i.…
A:
Q: How do beryllium hydroxide and barium hydroxide each compare to the Group II metal hydroxides
A: 1. Ionic Size:Beryllium hydroxide has a smaller ionic size compared to magnesium hydroxide, calcium…
Q: The SN2 reaction of (S)-2-brombutan with cyanide (-CN) with the others from the server.
A:
Q: 11. Draw the dinucleotide containing two molecules of 5'- monophosphate-guanosine in the space…
A: Here, we have been asked to draw a dinucleotide two molecules of 5'-guanosine monophosphate First of…
Q: Consider the reaction. A(aq)↽−−⇀2B(aq) ?c=7.07×10−6 at 500 K If a 4.10 M sample of A is heated to…
A:
Q: For a generic acid, HA, Ka = 1.0 × 10-8. Calculate the pH of a solution made by mixing 31.3 mL of…
A: Given, Molarity of NaA = 0.340 MVolume of NaA = 31.3 mLMolarity of HCl = 0.100 MVolume of HCl = 25.1…
Q: The boiling point of an aqueous solution is 101.18 ∘C. What is the freezing point?
A:
Q: Caffeine is the active ingredient in coffee, tea, and some carbonated beverages. Add lone pairs, as…
A:
Q: Question 1 Drain cleaner was created by mixing 16 g of sodium hydroxide in 600 mL of water. The pH…
A:
Q: Name the following compound: OH CH H2C - OH CH₂ ОН
A:
Q: 3B. Draw the mechanism for the addition reaction of HBr and 3-methyl-2-butene. What is the major…
A: Addition of HBr to 3-methyl-2-butene (alkene) is an electrophilic addition reaction. In this…
Q: Devise a synthetic sequence for the reaction using the list of reagents available. Select the best…
A: An alkyne is prepared from an alkyl dihalide (either geminal or vicinal) using two equivalents of…
Q: c. Draw the major protonation state of cysteine at pH = 9.2. pKa values given. 8.37 HS. 3. OH H₂N…
A: As we know that if PH is equal to 7 , less than 7 and above 7 the medium will be neutral, acidic and…
Q: Four liquids are described in the table below. Use the second column of the table to explain the…
A: When a non-volatile solute is added to the pure solvent, the freezing point of the solution will…
Q: Lewis Structures Lewis Structures are used to describe the covalent bonding in molecules land ions.…
A:
Q: How many atmospheres of pressure are there in 836 mmHg?
A: The relation between the pressure in atmosphere(atm) and mmHg is 1 atm equals to 760 mmHg
Q: [HI] (M) 1.0 0.8 0.6 0.4 0.2 0 0 1 2 3 4
A: The graph shows the concentration of hydrogen Iodide versus time.From the reaction equation2 moles…
Q: Silver (1) ions form a precipitate with chromate ions. Ksp = 9.0 x 10-¹2. a. Write the balanced…
A: Silver Chromate precipitates out when silver ions reacts with chromate ions in aqueous solution
Q: Refer to the chemical equation Cl₂(g) + 2 HBr (aq) --> 2 HCI (aq) + Br₂ (I) to match each item below…
A: This problem is based on redox reaction. The detailed solution is given below.
Q: What is the formula of the ionic compound expected to form between the elements O and Al? Al2O3
A: An ionic compound is a type of chemical compound composed of positively charged ions (cations) and…
Q: What is the name of the compound with the formula P4010? Phosphorus pentoxide What is the name of…
A: Here the formula is given P4O10 we have to predict its name... So the name of this compound will…
Q: In a 4.00 L pressure cooker, water is brought to a boil. If the final temperature is 115.0 °C at…
A:
Q: When the following molecular equation is balanced using the smallest possible integer coefficients,…
A:
Q: One method of experimentally determining whether a species is paramagnetic is to weigh it in an…
A: Introduction:The magnetic susceptibility balance, also known as a Gouy balance, is a laboratory…
Q: How many lone pairs are found on Xe in XeF4 (don't let this freak you out - yes, Xe is a noble gas…
A: XeF4, also known as xenon tetrafluoride, is an inorganic compound composed of xenon (Xe) and…
Step by step
Solved in 3 steps with 1 images


