b. Consider the following molecule and use it to answer the two questions (i. and ii) that follow. H H H H H. H H H i) Classify the molecule as being saturated or unsaturated. Give a reason for your answer. ii) Classify the Carbon (marked with a *) as being 1º, 2º, 3º. State a reason for your classification. c. Arrange the compounds below in order of increasing boiling point. Explain the reasons for your answer and add structures to your worked solutions. (Hint- Draw structures of the compounds to aid your explanation) 2-methylheptane, octane and 2,2,4-trimethylpentane
b. Consider the following molecule and use it to answer the two questions (i. and ii) that follow. H H H H H. H H H i) Classify the molecule as being saturated or unsaturated. Give a reason for your answer. ii) Classify the Carbon (marked with a *) as being 1º, 2º, 3º. State a reason for your classification. c. Arrange the compounds below in order of increasing boiling point. Explain the reasons for your answer and add structures to your worked solutions. (Hint- Draw structures of the compounds to aid your explanation) 2-methylheptane, octane and 2,2,4-trimethylpentane
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter11: Intermolecular Forces And Liquids
Section: Chapter Questions
Problem 14PS: Answer the following questions using Figure 11.12: (a) What is the equilibrium vapor pressure of...
Related questions
Question
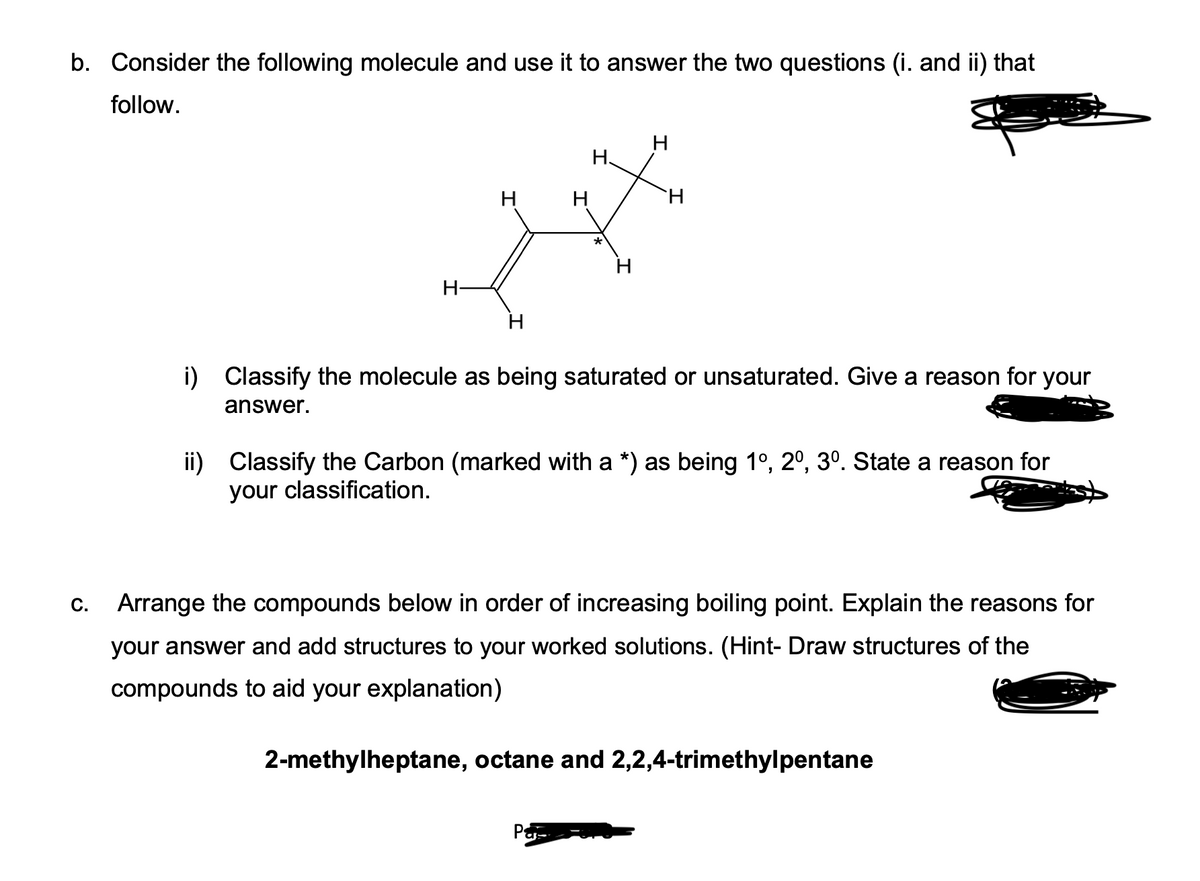
Transcribed Image Text:b. Consider the following molecule and use it to answer the two questions (i. and ii) that
follow.
C.
H
H
H
H
H.
H
H
Pa
H
i) Classify the molecule as being saturated or unsaturated. Give a reason for your
answer.
ii) Classify the Carbon (marked with a *) as being 1º, 2º, 3º. State a reason for
your classification.
Arrange the compounds below in order of increasing boiling point. Explain the reasons for
your answer and add structures to your worked solutions. (Hint- Draw structures of the
compounds to aid your explanation)
2-methylheptane, octane and 2,2,4-trimethylpentane
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning