Q: Draw the structure of ethylenediaminetetraacetate ion (EDTA“) is reacted with Mn" forming an octahed...
A: Mn4+ reacts with EDTA4- to form complex as per the below reaction Mn4+ + EDTA4- --------> MnEDTA...
Q: Determine the number of moles (of molecules or formula units) in sample. 0.1187 g C8H18
A: The expression for determining the number of moles of any substance is as follows, Molar mass of a ...
Q: Which one of the following statements is False? The rate constant depends on the activatio...
A: Rate constant, in chemical kinetics define the rate of reaction of a chemical reaction. The factors ...
Q: What is the name of the unit that equals (b) 10-6 second
A: A unit is an individual which is used in measurements. it tells about the quantity of something. For...
Q: Determine the pH of the solution that results from the addition of 5mL of a .1M HCl to 50mL of pure ...
A: Moles of HCl = Molarity × Volume = 0.1 M × 5 mL = 0.5 mmol Total volume of the...
Q: propose a mechanism that shows why p-chlorotoluene reacts with sodium hydroxide to give a mixture of...
A: When p-chlorotoulene reacts with NaOH, it gives benzyne as an intermediate. The Para substituted chl...
Q: If 0.10 M aqueous solutions are prepared of each of the following acids, which produces the solution...
A: The concentration of all the acids given are same. Also, all the given acids are monoprotic acids. S...
Q: Apply molecular orbital theory to determine the bond order of NO. (Use the energy ordering of N2.) I...
A: The bond order of a chemical bond between two atoms in a diatomic molecule is the number of bonds th...
Q: Please fill in the blanks and answer the questions! Thank you!
A: 1. increasing the concentration of a reactant causes the equilibrium to shift right side. 2. decre...
Q: Solve the attached Problem.
A: Given reaction,
Q: (a) Which is the acid and which is the base in the reaction? Explain using the appropriate acid / ba...
A: (a) In the above reaction NH3 is giving a H+ ion so NH3 is a acid here and Si9 4- is a base. (b) Ac...
Q: A rigid, sealed container that can hold 28 L of gas is filled to a pressure of 5.93 atm at 385 °C. T...
A: The gaseous state is the state of matter with least intermolecular force of attraction between molec...
Q: Write an expression for molarity and molality of a solution.
A: Molarity and molality both are the concentration terms. Molarity is seen in terms of volume whereas ...
Q: Draw structural formulas for the major organic product reaction
A: Given:
Q: Show how to convert propene to this compound , using any inorganic reagents as necessary. Q.) 1-Prop...
A:
Q: Why is 1-butanol, CH₃CH₂CH₂CH-OH, soluble in water, while heptane, CH₃(CH₂)₅CH₃, is not?
A: Why is 1-butanol, CH₃CH₂CH₂CH-OH, soluble in water, while heptane, CH₃(CH₂)₅CH₃, is not?
Q: The lattice energy of CsF is -744 kJ/mol, whereas that of BaO is -3029 kJ/mol. Explain this large di...
A: Given information: Lattice energy of CsF = -744 kJ/mol Lattice energy of BaO = -3029 kJ/mol
Q: What products are formed when each glycol is treated with HIO4?
A:
Q: 7. Solubility of PbCl2 in water is 0.0150 g/L at 25 oC. Determine standard free energy change for th...
A: The solubility of PbCl2 in g/L is given = 0.015 g/L Since solubility in g/L = molar solubility X mol...
Q: An industrial-scale process for making acetylene consists of the following sequence of operations: C...
A:
Q: Calculate the value of Q if mixture contains 0.21 M SO,, 0.19 M 02, and 0.42 M SO,
A: The reaction is: 2SO2 + O2 -----> 2SO3 [SO3]= 0.42 M [SO2] = 0.21 M [O2] = 0.19 M
Q: Fabiola, who lives in Mexico City, fills her car with gas, paying357 pesos for 40.0 L. What is her f...
A: Given, cost of 40.0 L of fuel = 357 pesos Now, let us convert the price of fuel in pesos to dollars ...
Q: notations or notations from structures, f
A: The notations from structure of tripeptides can be drawn as
Q: How many grams of Acetic acid are needed in 10L solution of 0.1M concentration?
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: The equilibrium constant (K.) is 0.0025 at 2127°C. If an equilibrium mixture at this temperature con...
A: The reaction in which reactant and product concentration do not change with time is called equilibri...
Q: The following is the balanced equation for the formation of ammonia. What is the rate of reaction w...
A: Given reaction, Ammonia formation reaction from nitrogen and hydrogen.
Q: How many molecules are in sample? 6.5 g H2O
A: Since the molar mass of H2O is 18 and one mole of H2O contains Avogadro number of molecules, the num...
Q: Using appropriate chemical reactions for illustration, show how calcium present as the dissolved HCO...
A: Bicarbonate, chloride salts of calcium causes hardness of water. Out of both, bicarbonate causes tem...
Q: Show how to convert the given starting material into the desired product. Note that some syntheses r...
A: Given reaction,
Q: A solution is prepared by mixing 631 mL of methanol with 501 mL of water. The molarity of methanol i...
A:
Q: What is a photon? How is the energy of a photon related to its wavelength? Its frequency?
A: The smallest particle which is able to represent the quantum of visible light and any electromagneti...
Q: 44 B only
A: 44. b. a nuclear isotope is genereally represented as where A = mass number z =atomic number 1.ato...
Q: Show reagents and experimental conditions to bring about the following transformations. H,C C=C H3C ...
A: Given reactions:
Q: What are alloys?
A: Alloys: Combination of two or more metals in order to enhance the chemical and physical properties f...
Q: During kinetic studies, the following reaction was determined to be second order in respect to NO an...
A: The given reaction is, 2 NO (g) + O2 (g) ----> 2 NO2 (g) The rate ...
Q: For the [Co(ox)3] -3complex What's the name of the complex? Draw the structure of the complex, does...
A: . trioxalate cobaltate(III) Yes it has one optical isomer/ enantiomer
Q: An unknown weak acid with a concentration of 0.190 M has a pH of 5.600. What is the Ka of the weak a...
A: Assuming the unknown weak acid is HA Hence the dissociation reaction of acid is HA ------> H+ + ...
Q: How much heat would need to be removed to cool 192.7 g of water from 25.6°C to -10.7°C? Csolid = Cli...
A: Heat released = 24.954 kJ
Q: Based on the provide elementary steps below, determine the net reaction. NO,CI (g) – NO2 (g) + CI (g...
A: In chemical kinetics, we study about the rate at which a chemical reaction proceeds and the mechanis...
Q: The reaction of ammonia decomposition on a patina surface is: 2NH3(g) àN2(g)+3H2(g) Is a zero-order ...
A: Since the reaction is given 0th order reaction. Hence the rate law of reaction is Rate = K = rate c...
Q: 7.Find the Kcor Kp values for each of the reactions
A: The reaction given is N2 (g) + H2 (g) --------> NH3 (g) Balancing : Since we have 2 N in LHS. He...
Q: Can you show me the right answer on this question? I don't know how to put the right the convert fac...
A: The reaction given is 2 Na (s) + Cl2 (g) --------> 2 NaCl
Q: Draw the product of treating alcohol Cyclohexanolwith chromic acid
A: The given alcohol is a secondary alcohol which undergoes oxidation with chromic acid (strong oxidizi...
Q: For which of the following reactions is S° > 0.Choose all that apply. S(s,rhombic) + 2CO(g) SO...
A: The entropy change of any reaction i.e ΔSo is +ve when the products have more gaseous components tha...
Q: Draw structural formulas for the major organic product reaction
A: Given: To draw the major organic product of the given reaction,
Q: Determining Density and Using Density to Determine Volume or Mass Calculate the density of mercury i...
A:
Q: An analytical chemist is titrating 177.8 mL of a 0.8600 M solution of ethylamine (C,H,NH,) with a 0....
A: We have, Let's calculate the number of moles of both, the acid and the base:
Q: How many carbon atoms are there in 16.3 mol of CO2
A: Given : Molecule is CO2 And moles of CO2 = 16.3 Since 1 molecule of CO2 has 1) 1 atom of C 2) 2 a...
Q: Draw structural formulas for the major organic product reaction
A: Given,
Write IUPAC names for these alcohols.
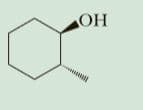
In the given compound, hydroxide group is the functional group and -CH3 is present as the side chain.
Step by step
Solved in 2 steps with 1 images


