Q: From the practical standpoint, is it better to use a "good" solvent or a "poor" solvent when…
A: Polymer molecular weight is an important characteristic that influences various properties such as…
Q: 4. Find the temperature at which water boils on a day in the mountains when the barometric pressure…
A: we have to calculate the boiling point of water at the mountains where pressure is 593 mmHg
Q: 1. Find the AT_b, and a AT_f, of the solvent in a solution containing 4.62 grams of glycerol,…
A: (1) Given that - Mass of glycerol = 4.62 g Mass of ethanol = 211 g Now, molality of the…
Q: A solution of urea (MW = 60 g/mole) in water has a concentration of 2.41 m (where m = molality,…
A: Molality of urea solution = 2.41 m Density of solution = 2.0 g/mL Molarity of solution = ?
Q: 3. Answer the following questions concerning sulfathiazole, below. :N S: sulfathiazole a. What is…
A:
Q: 2) Lowry's assay requires the sample protein concentration under the range of
A: Introduction The Lowry protein assay is a widely used technique that enables the quantification of…
Q: Where did these values come from
A: These values came from the calculation of the theoretical oxygen demand (ThOD) of each compound.…
Q: An experimental method of determining solubility parameters of polymers involves taking lightly…
A: Determining the solubility parameters of polymers is an important aspect of polymer science and…
Q: Which main theory did Compton use to explain electron scattering from the metal target?
A: We need to answer Which main theory did Compton use to explain electron scattering from the metal…
Q: A student has an acid (A) and base (B) solution that they mix together. The acid solution has 10 ml…
A: According to the question, The initial concentration of acid = C1 = 2 MThe final concentration of…
Q: 4. MATCH a structure or term from the following list with each description below. Place the letter…
A: Electrophilic substitution reaction are the reactions in which an electron rich group attacks on the…
Q: 3. a. Draw the structures of both of the major products for the following reaction. + H-CI b. Draw…
A:
Q: Calculate Mn, Mw and the polydispersity index for a hypothetical polymer sample that contains equal…
A: Given :- Equal number of molecules i.e. n1 = n , n2 = n , n3 = n m1 = 30000 m2 = 60000 m3 =…
Q: 3. Fill in the empty boxes with the correct information. Cation Ca2+ NH₂ Anion CI- CIO3 Formula K₂S…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Calculate the mass of potassium hydrogen phthalate (KHP), KHC8H4O4 needed to react completely with…
A:
Q: 2-4 please
A:
Q: I still don't get the following two questions, can you assist me?
A:
Q: CbzHN OHI DMAP activator RC(O)O- CbzHN H₂NCH₂CO₂Me H CO₂Me
A: Organic reactions are the chemical reactions that are takes place between organic reactants. There…
Q: Hydrazine, N2H4, reacts with oxygen to form nitrogen gas and water. N2H4(aq)+O2(g)⟶N2(g)+2H2O(l)…
A:
Q: determine the number of protons, neutrons, and electrons present in each of the atoms
A: Here, we have to calculate the number of protons, neutrons, and electrons for 34S and 67Zn.
Q: 12. a) Consider Ozone, Os. 1) Draw the Lewis structure. Include all resonance structures and formal…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 5. The equilibrium constant Kp for the following reaction is 0.403 at 1000 °C. FeO(s) + CO(g) Fe(s)…
A: Answer: Whether a system is at equilibrium or not, it can be checked by calculating the value of…
Q: Solid ammonium chloride, NH4Cl,NH4Cl, is formed by the reaction of gaseous ammonia, NH3,NH3, and…
A:
Q: Arrange the molecules in decreasing strength of intermolecular forces. (Strongest = 1; Weakest = 4).…
A: In general intermolecular forces depends on the types of compound.
Q: NaNO2(aq) + H2NSO3H(s) → NaHSO4(aq) + N2(g) + H2O(l) If you start with 1.627 g of H2NSO3H and an…
A:
Q: 5. A piece of metal weighing 59.047 g was heated to 100.0 °C and then put it into 100.0 mL of water…
A:
Q: Unknown solid Benzamide Ethyl-p-aminobenzoate D-toluic acid (Identification code (Manufacturer…
A: The mixture has a low melting point and a wide range of melting points than both the two pure…
Q: Given the exact mass of a 207 Pb atom is 3.4369171x10-25 kg, calculate the total nuclear binding…
A:
Q: 36 S u > Q2/ A) ethyl propyl ether (M=88) that has the following absorption peaks in mass spectrum…
A: Mass spectrometry is mainly used for identification of molecular weight of the compound. Also from…
Q: 2. Show how the following synthesis can be performed. Make sure to show all relevant intermediates…
A: The above transformation can be carried out by using different sets of reactants in a single step or…
Q: 6. Show that the angle between any two of the lines (bonds) joining a site of the diamond lattice to…
A: Show that the angle between any two of the lines (bonds) joining a site of the diamond lattice to…
Q: When a bond breaks, what is happening with the energy?
A: For breaking a bond energy is required. That is bond breaking is an endothermic process.
Q: N₁ N
A:
Q: Find AS for the system, surroundings, and universe when 20.6 g of liquid H₂O is evapourated at 100.0…
A: Given data., Mass of water given = 20.6 gram Molar mass of water = 18.015 g/mol Moles of water =…
Q: Calculate the standard potential, Eº, for this reaction from its AG value. 4+ X(s) + Y¹+ (aq) →→→…
A: Given ΔG° = 42 kJ = 42000 J
Q: A current of 3.69 A is passed through a Fe(NO3)2 solution. How long, in hours, would this current…
A:
Q: 1. 10 mL of 5.0x10 M HCl solution is mixed with 150 mL of water to make a new HCI solution. What is…
A:
Q: A biochemist has 100 mL of a 0.1 M solution of a weak acid with a p?a of 6.3. She adds 6 mL of 1.0 M…
A:
Q: A mixture is initially found to contain 0.47M H2 and 3.59M HCL at 648°C H2 + Cl2 (in equilibrium)…
A: The given equilibrium reaction is as follows: H2 + Cl2 ⇌ 2HCl The initial concentration of H2 = 0.47…
Q: Draw four resonance structures of the following ion. H₂C CH₂ CH₂ CH₂
A: Resonance structure:- Resonance structure represents same molecule. They are formed due to…
Q: ▼ Part A Calculate the osmotic pressure (in atmospheres) of a solution containing 2.60 g ethylene…
A:
Q: hat is the best way to draw a molecular orbital diagram for 1,3-cyclopentadiene, identifying the…
A: Number of molecular orbitals is equal to number of sp2 carbons present in the compound. In…
Q: 7 An unknown compound, insoluble in water and soluble in 10% aqueous HCl, has the molecular formula…
A: double bond equivalent (DBE) = 12[2 × n4 + n3-n1+2] where n4 = number of four valency carbon atoms…
Q: A fictitious element X has an average atomic mass of 122.397 u. Element X has two naturally occuring…
A: According to the question, The average atomic mass of the element X = 122.397 uThe mass of an…
Q: For the reaction below, N40, is a first order reactant. At 333 K the rate constant is 2.9 x 10-3 s1,…
A: Answer: In the question first order decomposition reaction of N2O5 is mentioned and we have to find…
Q: calculate the mass of CoCl 2 ·6H2 O required to obtain 50.00 mL of a 0.2500 M solution of CoCl 2…
A:
Q: 1- IR spectroscopy is most useful for identifying. 2- H₂ doesn't give an IR spectrum because 3- The…
A: Complete the following blank: Dear learner, as per our guidelines i can solve only three subparts of…
Q: Which is the best approach for the analysis of study from paracetamol in a pharmacokinetic the…
A: Full form of LC-MS is liquid chromatography-mass spectrometry GC-MS is gas chromatography-mass…
Q: How many atoms of all kinds are contained in 42.1 grams of calcium carbide (Ca2C)? Ans: 8.25 x 10^23…
A: Given : Mass of Ca2C = 42.1 gm Molar mass of Ca2C = 92 gm mol-1…
Q: Give typed solution A compound has two carboxyls and one aminium group. The pKas of the groups are…
A: A compound has two carboxyls and one aminium group. The pKas of the groups are 2.1, 4.5, and 9.1,…
please do with detail explanation and mechanism in chem draw
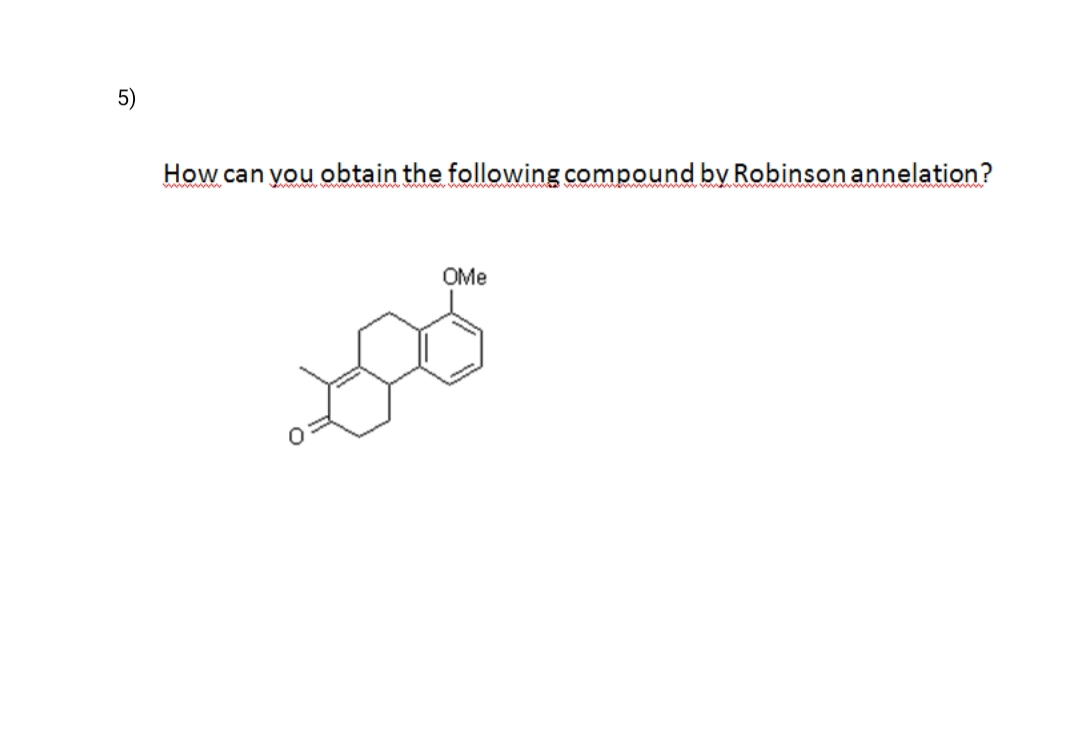
Step by step
Solved in 2 steps with 2 images

- How can the following synthesis be accomplished?please"The following reactions DO NOT afford the product that is shwon. Concisely explain why this is so."Attached compounds undergoes E2 elimination with strongbase? For compounds that undergo elimination, draw the product. Forcompounds that do not undergo elimination, explain why they areunreactive.
- What two carbonyl compounds are needed to synthesize each of the following compounds, using a Robinson annulation?Please deaw structures of the resonance structures of "enolate" of the following compounds with "a-H's" . Each has more than one kind, so choose the a-H marked (*). Thank you!How To Synthesize a Compound Using the Robinson Annulation ?

