Q: What 1° amine and carbonyl compound are needed to prepare each imine?
A: a. The given compound is, 1° amine and carbonyl compound are needed to prepare the above compound…
Q: Draw the products formed when each amine is treated with [1] CH3I (excess); [2] Ag2O; [3] Δ.…
A: Solution: a) part Product formed is given below when treated with [1] CH3I (excess); [2] Ag2O; [3]…
Q: What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was…
A: Phenacetin is an analgesic drug that reduces pain and fever. Furthermore, chemical name of…
Q: Which amine below is an primary aromatic amine? a. N-methyl-2-phenylethanamine b. All of the given…
A: We have to choose the following given amine below is an primary aromatic amine as follows in step 2:
Q: Be sure to answer all parts. Be sure to include the ion charges. What products are formed when the…
A: A neutralization reaction occur when an acid and base react
Q: Draw the major product formed from Hofmann elimination of the following amine. CH3 [1] CH3I (excess)…
A: Hofmann elimination: Elimination of group to form alkene is known as hofmann elimination. Only…
Q: Which of the following methods could be used to synthesize propylamine? 1. Gabriel amine synthesis…
A: Propyl amine can be prepared by using all the four methods given in question.
Q: CH3 NH2
A: Gabriel synthesis is the best method to prepare primary amines. In this reaction, potassium…
Q: What reaction involving amines is required to produce N-Propylcycloheptanamine? Propose a synthesis…
A: Since you have asked multiple questions, we will solve first question for you. Kindly post the…
Q: 1.answer the following questions A. What is reagent used to convert secondary amine to tertiary…
A: A. Secondary and Tertiary amines are those in which N atoms are bonded with two and three alkyl…
Q: 2) Predict the products formed when the following amides are treated with bromine and sodium…
A: Hoffman reaction
Q: 1. Rank the following nitrogens or amines in order of decreasing basicity (strongest to weakest,…
A: In case of basicity only to check the availability of loan pair of electron on nitrogen atom when…
Q: 2) What amine can be used to produce the following imine from a ketone or aldehyde? NH2 А. В. `NH2…
A:
Q: What is the name of the major product of the following synthetic scheme? OH 1. CH,CH,NH, 275 C 2.…
A: In this reaction acid amine coupling takes place which results in formation of amide bond which…
Q: 15. How is an amide converted to an anhydride? Water with strong acid → PCl5 → Carboxylic Acid PCl5…
A: Conversion of amide to anhydride:
Q: 3. For the following amines, propose a stepwise synthesis for each compound. ? NH2 a) OH b)
A: Step 1 The given amines can be formed using the alkenes as one of the reactants and with the use of…
Q: The first step in the mechanism for the reaction of an amine with a carboxylic acid to produce an…
A: The first step involved in the reaction between amine and carboxylic acid is given below
Q: What carbonyl compound and amine or alcohol are needed to prepare each product?
A: Given compounds,
Q: NH₂ The following reaction will give 3 different organic products. Sketch the three products. CH3Br…
A: The Gabriel synthesis is a chemical reaction that transforms primary alkyl halides into primary…
Q: Provide a synthesis for the following amine using a Gabriel synthesis. CH3 NH2
A: We have to provide a synthesis for the following amine using a Gabriel synthesis as follows in step…
Q: 8. We recently learned a few ways to prepare amines. Show how to synthesize the following secondary…
A: A. First Route: Reductive amination:
Q: Draw the products formed when each carbonyl compound reacts with the following amines: [1]…
A: Primary and secondary amines reacts with different carbonyl compounds to give different products.…
Q: N. NH2 a b N N. d I Z
A: The basicity of Amine depend upon various factors such as . Delocalization of lone pair effect.…
Q: What starting materials are needed to prepare each drug using reductive amination? Give all possible…
A: a) Reductive amination is a two-venture strategy that changes over aldehydes and ketones into…
Q: What starting materials are needed to prepare each drug using reductive amination? Give all possible…
A: Reductive amination involves the formation of a primary, secondary or tertiary amine by the reaction…
Q: Which of the following statements is correct regarding aliphatic amines? Select one: O a. aliphatic…
A: Amine are the compounds having -NH2 as a functional group in them.
Q: How could you prepare the following amides using an acid chloride and an amine or ammonia? (a) CH…
A: We have to synthesize the given amides using an acid chloride and an amine or ammonia. The given…
Q: Rank the compounds in order of electrophilicity. (X > Y means X is more electrophilic than Y). O…
A: An electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron…
Q: N. carbonyl compound amine Select one: NH2 a. „NH2 O b. HN O d. + ZI
A: When a ketone or Aldehyde (carbonyl group containing Compound) is treated with amine , then imine is…
Q: Which of the following statements is correct regarding aliphatic amines? Select one: a. aliphatic…
A: The true statement regarding aliphatic amines has to be given,
Q: What starting materials are needed to prepare each drug using reductive amination? Give all possible…
A: Reductive amination involves the formation of a primary, secondary or tertiary amine by the reaction…
Q: What is the product of the following reductive amination? CH, CH, NHz mild acd Nafity or Na BH3CN…
A: This is an typical example of reductive amination. The details solution for this reaction is…
Q: Draw a stepwise mechanism for the following imine hydrolysis.
A: The hydrolysis of imine leads to the formation of corresponding amine and a carbonyl-containing…
Q: Rank the labeled nitrogen atoms in each compound in order of increasing basicity. Histamine causes…
A: The basicity of the nitrogen is due to the presence of lone pair on it which it can donate to other…
Q: Show how you would use appropriate acyl chlorides and amines to synthesize thefollowing amides.(a)…
A: (a) Amines are undergoing a condensation reaction with acyl chlorides to give amide. The…
Q: What are the products of reactions of aldhydes and ketones with (I) chloroform (ii) primary amines?
A:
Q: Give the expected product when N-methylethanamine reacts with propanal followed by treatment with…
A: Structure of N-methylethanamine is as follows. N-methylethanamine is a secondary amine. Structure…
Q: What amides will form each amine on treatment with LiAlH4?
A:
Q: Rank the following amines in order of basicity from hig
A: Electron pair donar is called " Lewis base " Example : amines In amines N atom has lone pairs…
Q: What alkyl bromide would you use in a Gabriel synthesis to prepare each of the following amines? a.…
A: a.
Q: Q1- From a-haloketone how can you prepared flowing compounds : Imidazole Oxazole…
A:
Q: How could you convert N-methylbenzamide to the following compounds? a. N-methylbenzylamine b.…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: Which of these amine will react fastest in a reductive amination reaction?
A: Reductive amination is the successive conversion of primary amine into secondary amine and tertiary…
Q: a) What are the difficulties with the following reaction to synthesize an amine. + HBr Br + NH NH2…
A: Given reaction is :
Q: 3. For the following amines, propose a stepwise synthesis for each compound. ? NH2 a) ÓH ? b) ZI
A: Alkene converts to epoxide in presence of mcpba. Epoxide can be transformed into the target material…
Q: How could you prepare the following amides using an acid chloride and an amine or ammonia? | (a)…
A: To find: The preparation method of the following amide from acid chloride and an amine or ammonia a)…
Q: (a) Explain why phentermine [PhCH2C(CH3)2NH2] can’t be made by a reductive amination reaction.(b)…
A: (a)The structure of phentermine is given below.
Q: What 1° amine and carbonyl compound are needed to prepare each imine?
A: In the reaction of primary amine with carbonyl compounds in presence of acid, the C=O gets replaced…
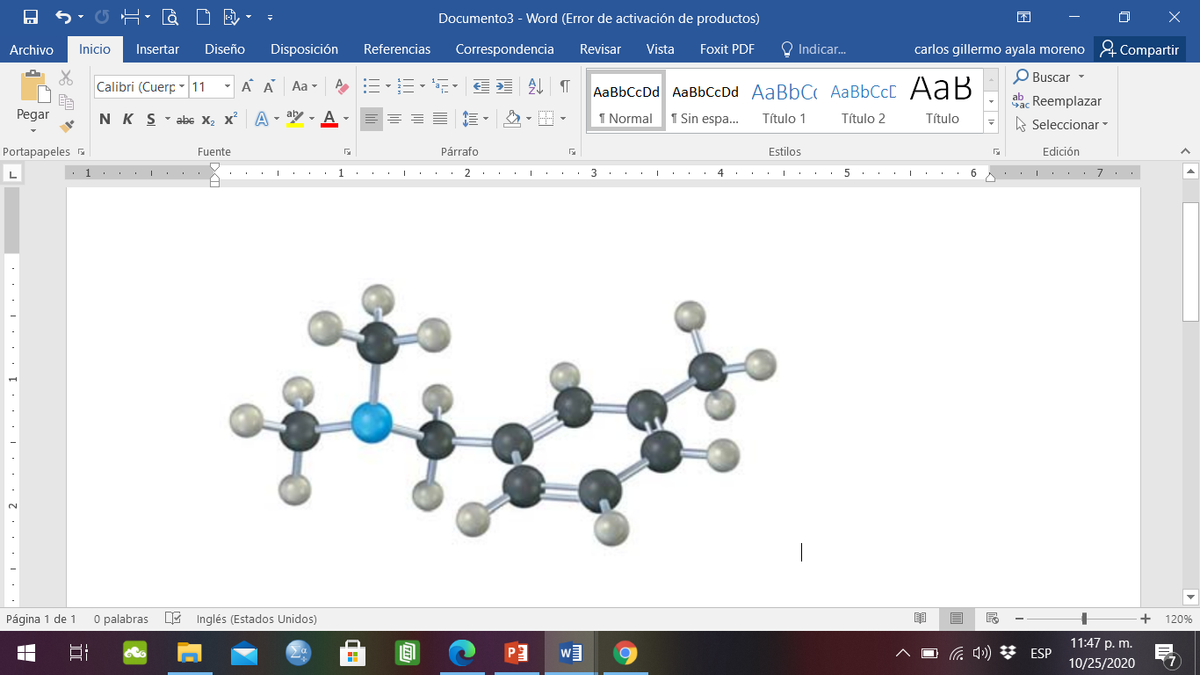
How can you prepare the following
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- In the synthesis of hydrocarbons, the carbon source is carbon dioxide. Although the CO2 concentra?on in the atmosphere raises at a drama?c speed, point sources are probably the easier sources for a PtX process. Iden?fy 3 possible point sources, explain why CO2 is formed and what challenges each of the three CO2 streams presentsI this calculation of the enthalpy of a reaction as measured in a constant pressure telemetry container, I totally get everything except for the exothermic part. I understand that hcl at 25 C mixed with NaOH at 25 degrees gives off h20 and heat, thus exothermic. But what puzzles me is why the formula q = s m delta T yields a positive number indicating an endothermic process. ( final temp higher than initial)Carbonic anhydrase of erythrocytes (Mr 30,000) has one of the highest turnover numbers known. It catalyzes the reversible hydration of CO2:This is an important process in the transport of CO2 from the tissues to the lungs. If 10.0 μg of pure carbonic anhydrase catalyzes the hydration of 0.30 g of CO2 in 1 min at 37 °C at Vmax , what is the turnover number (kcat) of carbonic anhydrase (in units of min −1 )?
- If qrxn =-2000 J, the equation has 1:1 mole ratio, and their are 0.00125 moles of limiting reagent. What is deltaH, when deltaH = q/n?What is ΔSsurr for a reaction at 28.6 °C with ΔHsys = 38.9 kJ mol-1 ? Express your answer in J mol-1 K-1 to at least two significant figures.A pellet of benzoic acid standard was combusted in a bomb calorimeter to determine the experimental calorimeter constant. After 6 mins of monitoring the temperature, the bomb was fired. The following data were obtained. mole of benzoic acid: 0.0049 mol mass of burnt fuse wire: 0.0125 g qwire,surr: 5858 J/g ΔHcombustion of benzoic acid: -3228.0 kJ/mol Δng (combustion of benzoic acid): -0.5 mol R: 8.314 J/mol•K r1: 0ºC/min r2: 0ºC/min Tf: 30.4ºC Ti: 28.4ºC Determine the calorimeter constant (in J/ºC). Report your answer in 2 decimal places.
- For the reaction A + B <=> AB, kassoc = 2.0 sec-1 M-1 and kdissoc = 0.5 sec-1. This means that Kassoc is equal to ____ M-1. (Enter your answer to the nearest 0.1; do not include units)1. CaCO3(s) <--> CaO(s) + CO2(g) ΔH° = +178 kJ exo or endo - thermic remove mass(CaO) --> shift: __ (increase) P(CO2) shift? __ ... two ways to lose mass(CaCO3)? __________ higher T shift? __ 2. SO2(g) <--> (1/8)S8(s) + O2(g) ΔH° = +297 kJ exo or endo - thermic Δν(gas)= _, (increase)Vtot --> P(gas)_ > on _hs shift? __ (decrease)P(SO2) shift? __ ... two ways to make mass(S8)? ___________ lower T shift? __ 3. C(s) + 2H2(g) <--> CH4(g) ΔH° = –75 kJ exo or endo - thermic Δν(gas) = _, (decrease)Vtot -->P(gas)_ > on _hs shift? __ (increase)P(CH4) shift? __ ...two ways to to lose mass(C)? ___________ higher T shift? __Assume no acid-base catalysis occurs and the following data: Under biological conditions (310 K) 1. dGb = -20.00 kJ/mol. 2. The dG of E +S binding to ES (dGs) is -3.00 kJ/mol. 3. dGd mismatch of energy in ES = +2.00 kJ/mol. What is the catalytic power (kcat/k_uncat) at 360 K? Use 1 decimal place.
- Thermodynamics Quantities for Selected Substances at 298.15 K (25⁰C) Substance ∆H⁰f (kJ/mol) ∆G⁰f (kJ/mol) S (J/K-mol) Carbon C2H2(g) 226.7 209.2 200.8 CO2(g) -393.5 -394.4 213.6 Hydrogen H2(g) 0 0 130.58 Oxygen O2(g) 0 0 205.0 H2O(l) -285.83 -237.13 69.91 What is the ∆S⁰ in the combustion of ethane in the presence of excess oxygen yields carbon dioxide and water: 2C2H2(g) + 7O2(g) → 4CO2(g) + 6H2O(l) in J/K?By the use of Henderson Hasselbalch equation; pH = pKa + log{[acetate ion]/[acetic acid]} 4.5 = 4.75 + log{[0.10 M]/[acetic acid]} -0.25 = log{[0.10 M]/[acetic acid]} [Acetic acid] = 0.10 M/ 10-0.25 [Acetic acid] = 0.10 M/0.56 [Acetic acid] = 0.1786 M Moles of sodium acetate dissolved in 250 mL buffer solution = 0.10 M× (250mL/1000mL) × 1L = 0.025 mol Weight (w) of sodium acetate (purity 100%) dissolved to prepare 250 mL of solution with buffer concentration of 0.10 M is calculate as follow; w100% = 0.025 mol × 82.0343 g/mol = 2.051 g Weight (w) of sodium acetate (purity 99%) is calculate as follow; w99% = 2.051 g× (100/99) = 2.072 g What was the volume of 6.12 M acetic acid HC2H3O2 needed to prepare the 250 mL acetic acid/acetate ion buffer solution required in this part? Show your calculations.Compound Δ?∘f (kJ/mol)ΔGf∘ (kJ/mol) A +387.7+387.7 B +600.4+600.4 C +402.0+402.0 Use the data given here to calculate the values of Δ?∘rxnΔGrxn∘ at 25 ∘25 ∘C for the reaction described by the equation A+B↽−−⇀C A+B↽−−⇀CA+B↽−−⇀C Δ?∘rxn=ΔGrxn∘= kJkJ If Δ?∘rxnΔHrxn∘ and Δ?∘rxnΔSrxn∘ are both positive values, what drives the spontaneous reaction and in what direction at standard conditions? The spontaneous reaction is enthalpy-driven to the left. enthalpy-driven to the right. entropy-driven to the right. entropy-driven to the left.

