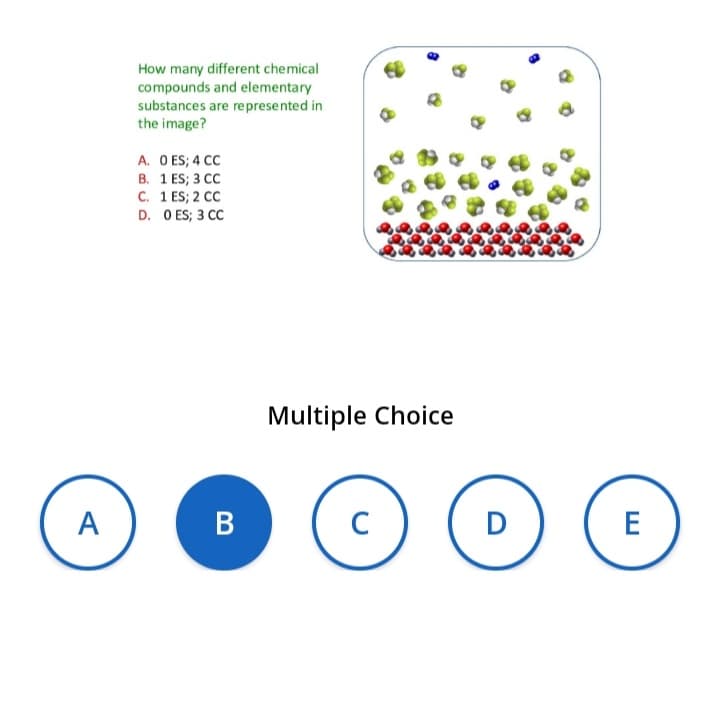
How many different chemical compounds and elementary substances are represented in the image? A. O ES; 4 CC B. 1 ES; 3 CC C. 1 ES; 2 CC D. O ES; 3 CC
Q: 201 80 Но- е — Complete the reaction 0 write the molecular mass followed by the atomic number…
A: Electron capture is a type of radioactive decay where the nucleus of an atom absorbs a K or L shell…
Q: Use the References to access important values if needed for this question. 1A 8A H 2A 3A 4A SA 6A 7A…
A: Two species are iso electronic when they have same number of electron thus have electronic…
Q: D. _KCIO3(S)---> _KCI(aq)+ _02(g) E. Fe203 (s) + _H2O(I)---> _Fe(OH)3 (s) F. Ca(OH)2 (aq) +_HNO3…
A: In a balanced chemical equation, the number of moles of each atoms on both sides of the equation is…
Q: PBCI, Cr+ O C?- Cr*+ C14- Pb4+ Cr+ O CI- 03- O PBCI 02- O Pb3+ Na, PO, Al(CIO,), PO, O CIo}- Na, P2…
A: Given, Cr2O3 Ions are => Cr3+ and O2- PbCl4 Ions are => Pb4+ and Cl- Na3PO4 Ions are…
Q: I, J and K are metals on the periodic table of elements. An experiment was carried out with all…
A:
Q: Write the cell notation for the voltaic cell that incorporates each of the following redox…
A: To find: The cell notation for the given reaction
Q: CH«g) + O3(g) → CO2(g) + 2H2O(g) AH? (kJ/mol) -249.2 Substance Substance AHf (kJ/mol) H2O1) H2Og)…
A: Calculate change in enthalpy of the given reaction---
Q: A mixture of 130.5 g of P and 134.8 g of O, reacts completely to form P¸O, and PO10. Find the masses…
A: Given - Grams of P = 130.5 g Grams of O = 134.8 g This means that 8 moles of P reacts with 8 moles…
Q: When magnesium hydroxide reacts with nitric acid, it produces magnesium nitrate and water.…
A:
Q: 3. H,SO, (aq) + Sr(OH), (aq) (aq)
A: The given un balanced reaction is : H2SO4(aq)+Sr(OH)2(aq)→? +?
Q: 17. 75.21 °C, Ca(s) + 2 H,SO, () → CaSO,(s) + SO2 (g) + 2 H20((0) 18. 982 K, C¿H2 (9) + 5 N,0(g) → 2…
A: Given Reaction C2H2(g) +5N2O(g) →2CO2(g) +H2O(g) +5N2(g)To calculate ∆G° at 982 k∆G° = ∆H°-T∆S°∆H° =…
Q: Identify the oxidizing and reducing agents in the following:
A: The oxidizing agent and reducing agent in the chemical reactions has to be identified. (a) The given…
Q: HO. 36 o and 5 rT O 21 o and 5 t 50 o and 5 n
A: The question is based on the concept of chemical bonding. we have to calculate how many Sigma and Pi…
Q: O 2Li(s)+Cl2(g)→2LİCI(s) O NHẠCI(s)→NH3(g)+HCI(g) O Rn(g)+F2(g) →RNF2(g) O…
A:
Q: Which is true about the chemical equation for photosynthesis? A.carbon dioxide is produced…
A:
Q: Solar energy is: O the energy stored within the structural units of chemical substances. O the…
A: To define: Solar energy.
Q: 4NH,() + 50,()= 4NO() + 6H,O(g) Procice Exercises 6.1 4NH,(2) + 50.(e) 4NO() + 6H,OI b. H,CO,(ag) =…
A: Equilibrium constant can be expressed in terms of concentration ( Kc ) or pressure ( Kp). Kc is the…
Q: T1,O pyridine BH в A C Cul, Pd(PPh,), Br Br Pd(PPh,) NaOEt Pd(OAC), PPh,. NEt, E D
A:
Q: 12:35 Th 7 21 thg 5 a bai tap c1-đã gộp-đã nén - میں ویلی OH 0%+0² [a OH oo-oo OH Br oo-ob In $ H…
A: Given, Grignard reagent ( RMgX) used for conversion of carbonyl compounds such as ketones Or…
Q: التاريخ NOZ HADZ HzSou what is me Canisum
A: Quinoline is a heterocyclic aromatic compound, It can undergoes Nitration 5th and 8th position.
Q: Please help don’t understand
A: A chemical reaction is symbolic representation of the conversion of substances to new substances.
Q: (a) CaCO,(s)²Ca²*(aq) + CO,²-(aq) (b) Mg,(PO,);(9) =3 Mg²"(aq) + 2 PO,' (aq) (c) CO(g) +…
A:
Q: A sample of propane gas, C3H8, was combusted to produce energy according to the equation: C3H8…
A: Given : Molecules of CO2 produced = 1.35 x 1024 Density of water = 1 g/cm3 Also, molar mass of…
Q: 2KCIO,(s) 2KCI(s) + 302g) If 17.8 g of potassium chlorate is heated and decomposes, find the mass of…
A:
Q: A cable making company produces a metal wire, with a diameter of 1.00 mm. What will be the length of…
A: Introduction: Given data: • The diameter of metal wire is 1 mm •The number of moles of metal is 1.00…
Q: C3H8(g) + 5 02(g)-> 3 CO2(g) + 4 H2O(1) AG (kJ/mol) -23.49 Substance CaHe(g) O2(g) Co2(g) H2O(1)…
A: Formula : ∆Gr° = ∑∆Gf°products-∑∆Gf°reactants ∆Gr° = (- 394.4 x 3) + ( - 237.2 x 4 ) - ( - 23.49 )…
Q: Draw the products of the following reactions:
A: Given,
Q: a 9.48 kg sample of steel contains 0.08058kg og carbon and 0.1896kg of manganese. How many kg of…
A: A 9.48 kg sample of steel contains 0.08058 kg of carbon and 0.1896 kg of manganese. The mass of…
Q: - Write the chemical equation(s) for each part of the experiment, as outlined in Equation 1. (0)…
A: (a) This reaction actually occurs in 2 steps. In the first step, the copper (red-brown) is oxidized…
Q: Chemical equations are balanced with numbers called: a. subscripts b. exponents c. coefficients d.…
A: Chemical equation is a way to symbolically represent chemical reaction using atomic symbols or…
Q: You have a 11.5 mg sample of blood that contains various proteins. Hemoglobin is the only protein in…
A: Molar mass of heme (C₃₄H₃₂FeN₄O₄) = 616.49 g/mol Molar mass of Fe = 55.85 g/mol Each mole of heme…
Q: Given these reactions, where X represents a generic metal or metalloid 1)…
A: There are some errors in the given reactions, here the corrected reactions are,
Q: Automobile airbags inflate due to the formation of nitrogen gas from the chemical reaction 2 NaN,…
A: The given chemical reaction is represented as follows:
Q: Use the References to access important values if needed for this question. 1A 8A H 2A 3A 4A SA 6A 7A…
A: Isoelectronic species are the species having same electrons. Electron of Cl- = (17+1)= 18e-…
Q: a. Al (s) + HCl (aq) → AlCl3 (aq) + H2 (g) b. C2H4 (s) + O2 (g) → CO2 (g) + H2O (g) NH3 (g) + 02 (g)…
A: Following are the balanced chemical equations.
Q: of a Hydrate 23.43 29.24 26.31 1. Mass of crucible 2. Mass of crucible and hydrate 3. Mass of…
A: The question is based on the concept of molecular formula determination. we have been given…
Q: g. CdCd?a)||Sbag)|Sb(s) h. Pt|Cutag), Curaq)||Mg?aq)|M9(s) i. Pt|Cran), Cr20Ta)||CLO5(aq), Clag)|Pt…
A: Answer:- This question is answered by using the simple concept of writing the balanced chemical…
Q: Provide a Balanced equation for the hydration of each of the following compounds
A: Sodium sulfate (Na2SO4) is a strong electrolyte that dissociates completely into its ions when they…
Q: A cable making company produces a metal wire, with a diameter of 1.00 mm. What will be the length of…
A:
Q: Given these reactions, where X represents a generic metal or metalloid 1)…
A: There are some errors in the given reactions, here the corrected reactions are, Required…
Q: 22) HO. + H,C=C=0 ?
A: A ketene is an organic compound of the form R'RC=C=O where R and R' are two arbitrary chemical…
Q: (c) 174Hf – 170yb + ? chemPad Help Greek - (d) 192pt - 1880s + ? chemPad Help Greek - 1L
A: Loss of 1 Alpha particle causes decrease in mass number (A) by 4 units and decrease in atomic number…
Q: Cलकावैक he य रिचक इन्कोपारक क कुनेज्ञ प्रनंje \৪০ 184 188 74 १५ 74 74 व) पकांकी nueiबिर र काकम्मे…
A: Radioactivity is a nuclear phenomenon where a heavy unstable nucleus undergoes spontaneous decay and…
Q: 3A+6B→3D AH - 403 kJ/ тol E + 2F→ A AH, = – ΔΗ 105.2 kJ/mol C→E+3D AH3 = +64.8 kJ/mol 6B+4E+6F→C AH…
A: Given equations are:
Q: What is -30°F in K? Зок O 30K O 303 K O-30 K O 239 K 04K
A: To convert F to K, Subtract 32 from the Fahrenheit temperature. Multiply this number by 5. Divide…
Q: Types of Chemical Reactions Determine what types of chemical reactions are the equation undergo.…
A: The most basic types of chemical reactions are combination, decomposition, single-replacement,…
Q: A number of atoms on the reactants side: Na: Cl: A number of atoms on the products…
A: 2Na + Cl2 -----> 2NaCl (reaction is ) The number of atom on the reactant side is Na: 2 Cl : 2
Q: Puniverse n substance: O2(g) → NİSS) 318 -88.1 SO2(9) + O2(g) 68 NiOs) at 25 °C SO2(g) 412 -296.8…
A: According to the question, we need to determine the value of ∆Suniverse. The chemical reaction:…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps


