How many grams of AgCI will be formed when 60 ml. of 0.500 MAGNO, is completely reacted according to the balanced chemical reaction: FeCl,(aq) 3 AGNO,(aq) - 3 AGCI(s) + Fe(NO,),(aq)
How many grams of AgCI will be formed when 60 ml. of 0.500 MAGNO, is completely reacted according to the balanced chemical reaction: FeCl,(aq) 3 AGNO,(aq) - 3 AGCI(s) + Fe(NO,),(aq)
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter3: Chemical Reactions
Section: Chapter Questions
Problem 37QRT
Related questions
Question
Help me with this factor
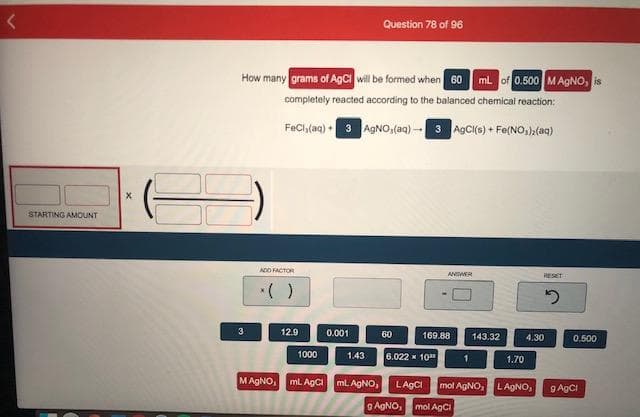
Transcribed Image Text:How many grams of AgCI will be formed when 60 ml. of 0.500 MAGNO, is
completely reacted according to the balanced chemical reaction:
FeCl,(aq) 3 AGNO,(aq) - 3 AGCI(s) + Fe(NO,),(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning
Expert Answers to Latest Homework Questions
Q: Good Day,
Q: 2
Q: Good Day,
Q: Consider these two entries from a fictional table of standard reduction potentials.linlrnax
3+ + 3è…
Q: Good Day
Q: Determine the radius of convergence and the interval of convergence for the power series:
Q: Please Write Step by Step Calculation
I gave Thumb up
Q: Good Day
Q: 10) The dimerization of butadiene was studied at 500. K:
2C4H6 (g) →C8H12 (g)
The following rate…
Q: Good Day,
Kindly assist with this query
Q: QUESTION TWO [30]2.1 Examine the format of the first digital adverts. (15)2.2 Determine how ad…
Q: 1.4 Discuss what are some key types of video content that can be used for marketing purposes to…
Q: 1.3 Determine how can video content be used to build trust with an online audience, and why is trust…
Q: 1.2 Examine what are some common mistakes that can impact conversion rates in online marketing, and…
Q: 1.1 Elaborate on why video content is considered a valuable tool for increasing conversion rates in…
Q: What happens when an employee records a fictitious refund of goods at his cash register?
Question…
Q: after gathering information about a clients risk tolerance and having knowledge of the client’s…
Q: is there an ethical issue that must be considered between the financial advisor and the client in…
Q: Vikram bhai
Q: PRICE (Dollars per bike)
500
450
400
350
ATC
300
250
200
150
100
50
MC
MR
Demand
0
0 50 100 150 200…
Q: None