With regard to Na+ and K+ equilibrium potentials and the resting and active membrane potentials, write down (a- O the directions of the forces indicated, acting on the ion in the table below under the respective condition(s). NB the examples given. lon Condition Electrical/Chemical force Direction of force Chemical e.g. ....inward. ******** Na ENa+ = + 60 mV Electrical a. Chemical e.g....outward. EK+ =- 90 mV Electrical b. Chemical C. Na Emp = 0 mV Electrical d. Chemical K+ Emp= -20 mV Electrical
With regard to Na+ and K+ equilibrium potentials and the resting and active membrane potentials, write down (a- O the directions of the forces indicated, acting on the ion in the table below under the respective condition(s). NB the examples given. lon Condition Electrical/Chemical force Direction of force Chemical e.g. ....inward. ******** Na ENa+ = + 60 mV Electrical a. Chemical e.g....outward. EK+ =- 90 mV Electrical b. Chemical C. Na Emp = 0 mV Electrical d. Chemical K+ Emp= -20 mV Electrical
Human Physiology: From Cells to Systems (MindTap Course List)
9th Edition
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Lauralee Sherwood
Chapter4: Principles Of Neural And Hormonal Communication
Section: Chapter Questions
Problem 4TAHL: Assume presynaptic excitatory neuron A terminates on a postsynaptic cell near the axon hillock and...
Related questions
Question
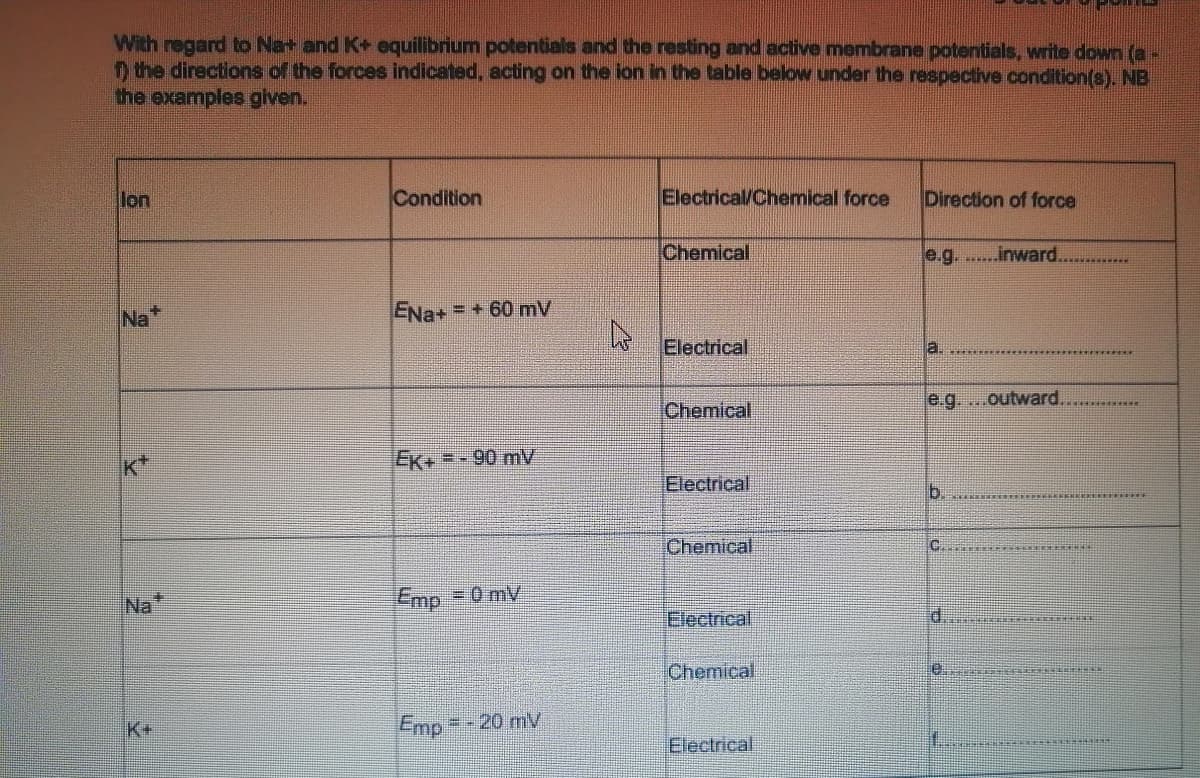
Transcribed Image Text:With regard to Na+ and K+ equilibrium potentials and the resting and active membrane potentials, write down (a-
D the directions of the forces indicated, acting on the ion in the table below under the respective condition(s). NB
the examples given.
lon
Condition
Electrical/Chemical force
Direction of force
Chemical
e.g....inward..
******
Na
ENa+ = + 60 mV
Electrical
a.
Chemical
e.g. ...outward..
EK+ =- 90 mV
Electrical
b.
Chemical
C.
Na"
Emp = 0 mV
Electrical
d.
Chemical
K+
Emp
=-20 mV
Electrical
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Human Physiology: From Cells to Systems (MindTap …
Biology
ISBN:
9781285866932
Author:
Lauralee Sherwood
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College