How many milliliters of water have to be added to 122.5 mL of 0.50 M HCl to reduce the concentration to 0.10 M HCI? Vwater mL %3D eTextbook and Media
How many milliliters of water have to be added to 122.5 mL of 0.50 M HCl to reduce the concentration to 0.10 M HCI? Vwater mL %3D eTextbook and Media
Chapter11: Solving Equilibrium Problems For Complex Systems
Section: Chapter Questions
Problem 11.1QAP
Related questions
Question
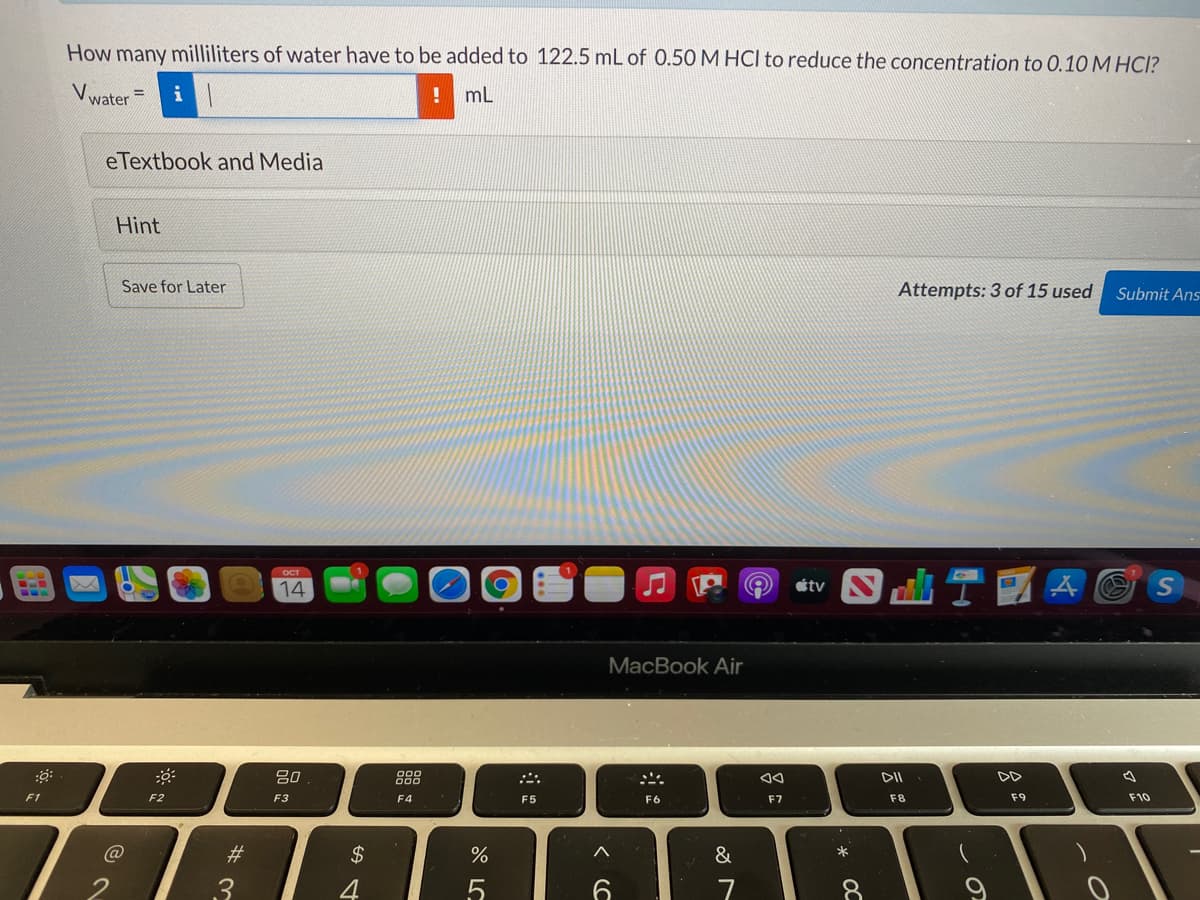
Transcribed Image Text:How many milliliters of water have to be added to 122.5 mL of 0.50 M HCl to reduce the concentration to 0.10 M HCI?
Vwater
mL
%3D
eTextbook and Media
Hint
Save for Later
Attempts: 3 of 15 used
Submit Ans
ост
14
tv
MacBook Air
80
888
DD
F1
F2
F3
F4
F5
F6
F7
F8
F9
F10
@
23
2$
4
7
OC
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning



Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning