How many mL of 0.668 M HBr are needed to dissolve 7.69 g of MgCO,? 2HB1(aq) + MgCO3(s) MgBrz(aq) + H50(1) + CO2(g) mL 9 more group attempts remaining Retry Entire Group Submit Answer hp ins prt sc dele f12 f11 トト f8 f9 f10 f6 17 L. 80
How many mL of 0.668 M HBr are needed to dissolve 7.69 g of MgCO,? 2HB1(aq) + MgCO3(s) MgBrz(aq) + H50(1) + CO2(g) mL 9 more group attempts remaining Retry Entire Group Submit Answer hp ins prt sc dele f12 f11 トト f8 f9 f10 f6 17 L. 80
Chapter13: Titrations In Analytical Chemistry
Section: Chapter Questions
Problem 13.29QAP
Related questions
Question
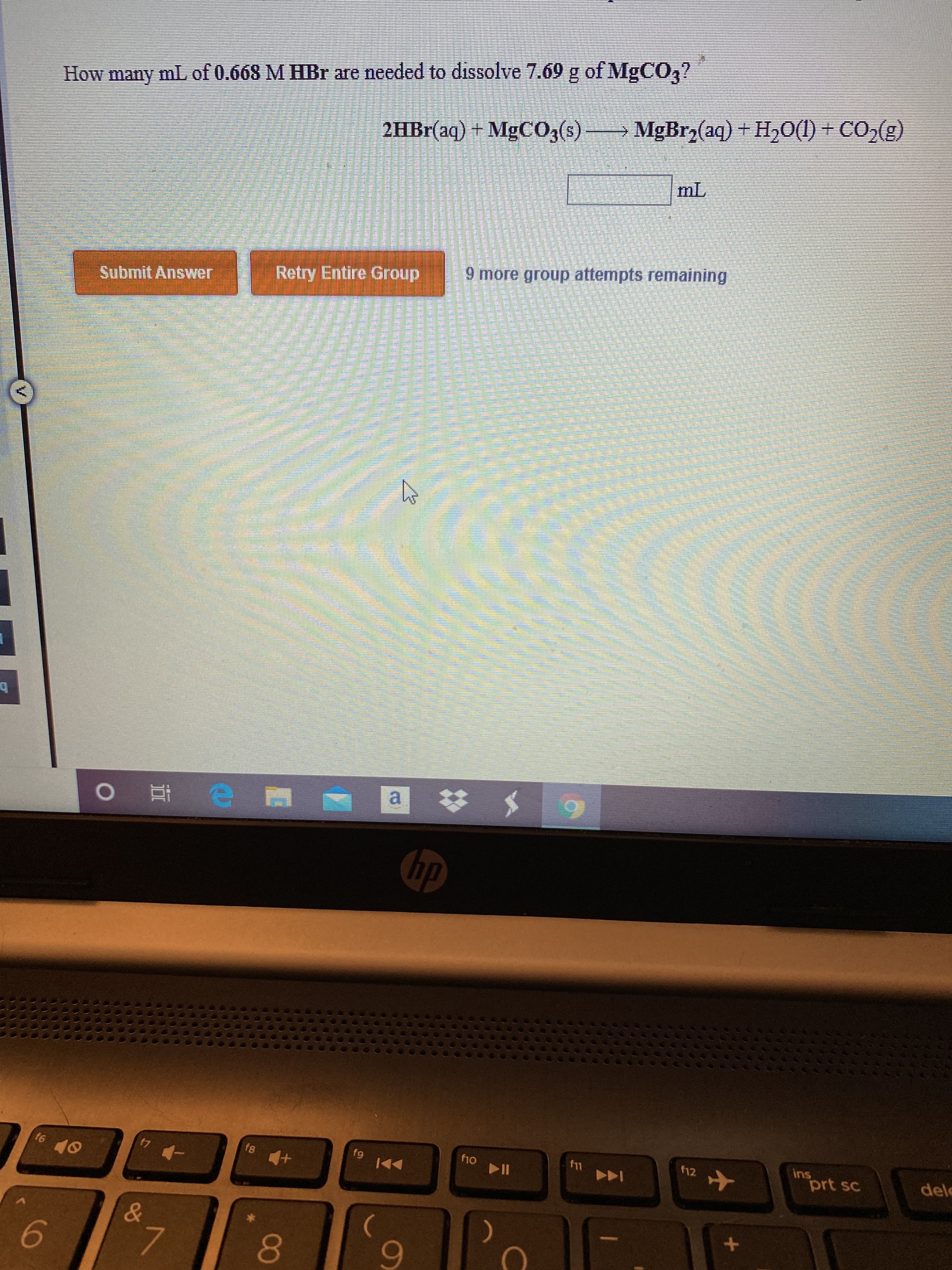
Transcribed Image Text:How many mL of 0.668 M HBr are needed to dissolve 7.69 g of MgCO,?
2HB1(aq) + MgCO3(s) MgBrz(aq) + H50(1) + CO2(g)
mL
9 more group attempts remaining
Retry Entire Group
Submit Answer
hp
ins
prt sc
dele
f12
f11
トト
f8
f9
f10
f6
17
L.
80
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning



Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning