Home 101 Chem 101 X (274) (Letra) A Mi Me Esta D X X х app.101edu.co Unofficial Transcript... S Document Require... Oregon Scholarship.... Welcome to the OS... myClackamas Login Apps WLogon Home FAFSA on t... The National Societ... > Submit Question 4 of 9 100.0 mL of a 0.635 M solution of KBr is diluted to 500.0 mL. What is the new concentration of the solution? М 1 2 3 х 4 6 с 7 8 х 100 +- 0 4:42 PM Type here to search ENG 11/21/2019 LO
Home 101 Chem 101 X (274) (Letra) A Mi Me Esta D X X х app.101edu.co Unofficial Transcript... S Document Require... Oregon Scholarship.... Welcome to the OS... myClackamas Login Apps WLogon Home FAFSA on t... The National Societ... > Submit Question 4 of 9 100.0 mL of a 0.635 M solution of KBr is diluted to 500.0 mL. What is the new concentration of the solution? М 1 2 3 х 4 6 с 7 8 х 100 +- 0 4:42 PM Type here to search ENG 11/21/2019 LO
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.26QAP
Related questions
Question
Transcribed Image Text:Home
101 Chem 101
X
(274) (Letra) A Mi Me Esta D
X
X
х
app.101edu.co
Unofficial Transcript...
S Document Require...
Oregon Scholarship....
Welcome to the OS...
myClackamas Login
Apps WLogon
Home FAFSA on t...
The National Societ...
>
Submit
Question 4 of 9
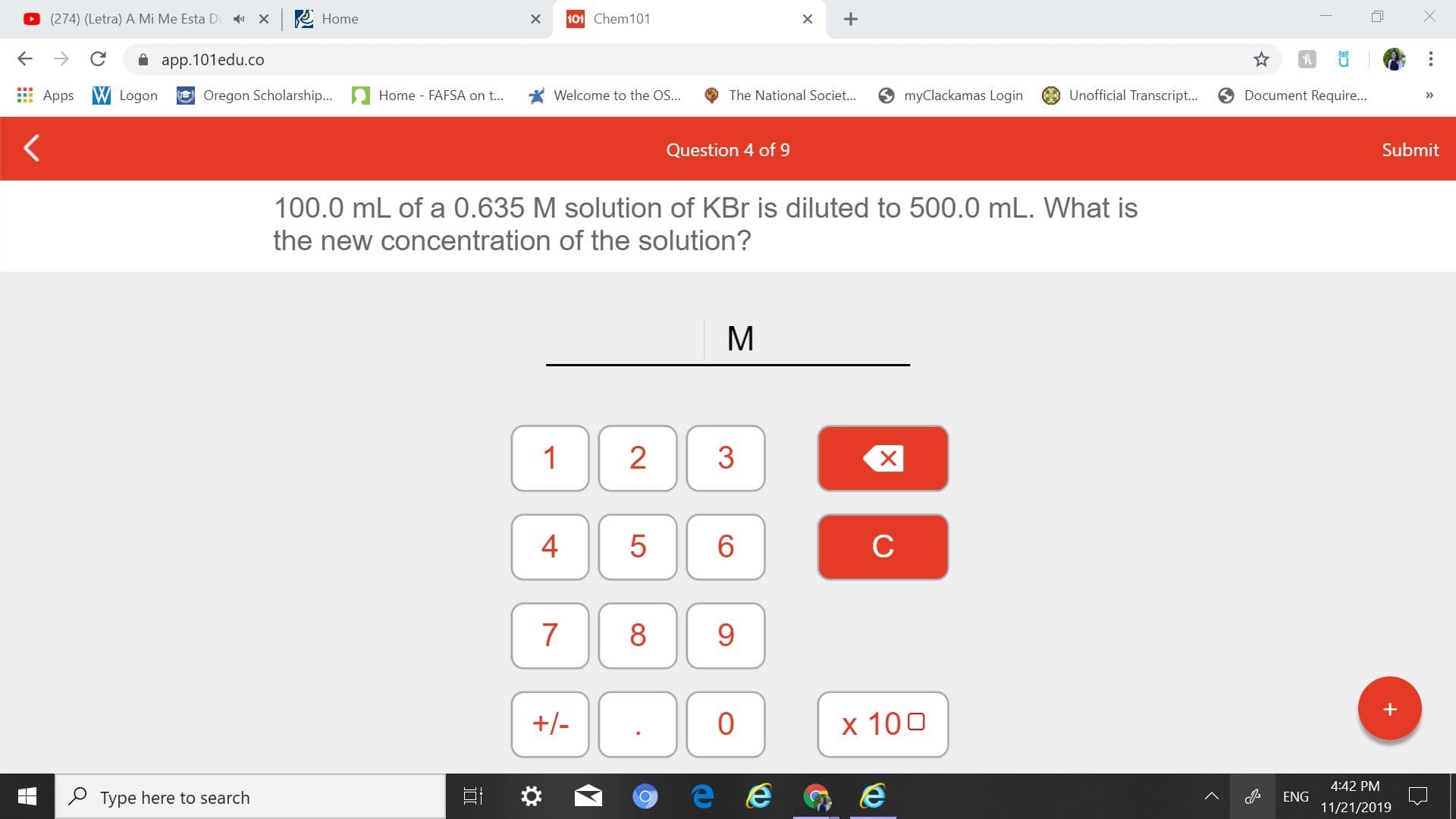
100.0 mL of a 0.635 M solution of KBr is diluted to 500.0 mL. What is
the new concentration of the solution?
М
1
2
3
х
4
6
с
7
8
х 100
+-
0
4:42 PM
Type here to search
ENG
11/21/2019
LO
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning