Q: Which group in each pair is assigned the higher priority? a. – CH3, – CH2CH3 b. – I, – Br c. – H, –…
A: According to CIP rule, priority of atoms or groups depends on atomic number of atoms. In other…
Q: Reagents Available a. CH2=CHCH,CI, AICI3 b. CН-Br2, ОН c. CH3CH2COCI, AICI3 d. H2, Pt e. NBS, CCI,…
A:
Q: Q3: Give 4ive the Product of fellowing ( 5x2=10) of fellowing (5x2=10) CH2-COH CH CH,-C-OH PCls +…
A:
Q: (a) (b) (c) ОН -OH OH ? -CH₂OH ? (d) О CHOH CH₂OH ? ? moilosot not in ее 2010 о о (e) H (f) -CH₂CI ?…
A:
Q: Tell whether each stereogenic isomer is either Ror S. Assign the correct priority of each group…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Be sure to answer all parts. Rank the following groups in order of decreasing priority: A. CH CH2 В.…
A: According to IUPAC the priority order should be: Hydrogen<Alkane<Alkyne<Alkene
Q: CH3-CH-CH-CH3 Br Br 2,3-dibromorobutane How many stereoisomers are possible for 2,3-dibromorobutane?
A:
Q: Which structure is the enantiomer of the original molecule? F H3C- H original structure H F F CH3…
A: the two pairs of molecules can be said to be enantiomers if they are mirror images of each other and…
Q: For the given ee values, calculate the percentage of each enantiomer present.a. 90% eeb. 99% eec.…
A: Constitutional isomers can be defined as the molecules which have the same molecular formula and…
Q: || CH2-С-NH 2 KOBr → (A); Product (A) is : C-0- CH3 (a) (b) (c) NH (d) NH 3.
A:
Q: Which group in each pair is assigned the higher priority in R,S nomenclature? а. — CDз, — СH b. -…
A: a.
Q: Which compound has eight stereoisomers? a) CH2-CH -сно ОН ОН b) CH2-CH-CH2 ОН ОН ОН c) CH2-CH-CH…
A: Carbon with four different ligands are called chiral centre. If n no. of chiral centre are present…
Q: 2. H₂C CH₂ مله CH3 + H₂C + AICI3 → (f) (major product only) CH₂ + Cl₂ (in uv light) → (g) (major…
A:
Q: Draw the structure of cis 1-bromo-3-methylcyclohexane and trans 1-bromo-3-methylcyclohexane (w/ all…
A: Alkanes can be defined as the organic compounds that contain single bond between carbon atoms in a…
Q: 5.47 Show how to convert methylenecyclohexane into each of these compounds. CH2OH (a) OH (b) =CH2…
A: a) The addition of water to an alkene in the presence of a catalytic amount of strong acid leads to…
Q: a.) Classify the following carbocations (1º, 2º, or 3º). b.) Encircle the carbocation/s that can…
A:
Q: one Group 2 two Compound A Compound B Compound C three H CH,CH, H CH,CH, H,CH,C Br Brm F I H,CH,C…
A: Enantiomers : are the non-superimposable mirror images. If the total inversion of all the chiral…
Q: B. Draw the structure of the missing reactant, reaction intermediate, or organic product/s. Use…
A: The answer to the following question is-
Q: From HzSO4 conc. 2) MCPBA + EN 3) Hz + EN
A: Using concept of reaction mechanism, stereochemistry of molecules and selectivity of reagents.
Q: Locate the tetrahedral stereogenic center(s) in each compound. A molecule may have one or more…
A: To identify: Tetrahedral stereogenic centres in the molecules.
Q: Part A Of all the possible cyclooctanes that have one chloro substituent and one methyl substituen…
A:
Q: CH2-CH3 HNO3 A (Para Product Only)- H2/Pt or Cl2 B H2SO4 Fe/HCI FeCl3
A: A chemical reaction is a process that leads to the chemical transformation of reactant to product.…
Q: a. O3, CH;Cly, -78 °C b. Me,S b OH + enantiomer OH но H.
A: Given reaction is : Product of the reaction = ? Options are :
Q: A. Name the following: CHa CH3-C-CH2 a. CH3CH2CH2-C-CH2CH2CH2CH2 CH3 CH-C-CH3 c.…
A: IUPAC RULES : Find and name longest continuous chain(Parent chain) with functional group if…
Q: H2 OH H2SO, Pd/C K 7. Major product only Only one stereoisomer Stereochemstry required NaBHa, MEOH…
A:
Q: What are the relative positions of the substituents in the following compound. A. ipso B. ortho…
A: Relative positions on benzene ring
Q: Rank the following groups in order of decreasing priority. a. – COOH, – H, – NH2, – OH b. – H, –…
A: The following groups are to be ranked in order of decreasing priority: – COOH, – H, – NH2, – OH –…
Q: 8. Br CH₂ CH₂CH3 XWV O VWX e AZY OYZA O O OH NaOCH₂ SN2 CrO₂, H₂SO4 acetone 10. * НСІ CH3 CH3-CH-OH…
A:
Q: .Br CH3 CH3 1S,2S)-1-bromo-1,2-dimethylcyclohexane Rx de B- Eliminación CH 1,2-dimethylcyclohex-1…
A: β-Elimination is a chemical reaction where atoms or groups are lost from adjacent atoms and it…
Q: CH CH, CH H Br Br CH CH, CH, CH, A C a) Which one of the above (A, B.C or D) is…
A:
Q: Br K* OC(CH)3 K* OC(CH); Br cis trans
A:
Q: What is each compound’s systematic name?
A: Systematic Name: It is a standardized name given for a chemical compound in systematic manner. Any…
Q: Draw the following compounds: a) 6-[1-chloropropyl]-5-methyl-1-decyne b)…
A:
Q: 15.45 Circle and name the functional group(s) in each compound: (a) CH3-CH=CH-CH2-OH (b) CI–CH,-…
A: Functional group :- The atom or group of atoms present in organic compound which shows the…
Q: No.e the following etrers 22 (a) H3C-0-CH3 (c) CH3-CH2-CH2-CH2-0-CH3 CH3 (b) H;C-0–CH CH3
A: a) Dimethyl ether b) Butyl methyl ether
Q: Be sure to answer all parts. Rank the following groups in order of decreasing priority: A. CH CH, B.…
A:
Q: 6 f) -Br CI CI H3C- H3C- KI acetone CH3 OK CH3 CH3 -OH CH3 CH3CH₂ONa CH3CH₂OH
A:
Q: [References) Rank the set of substituents below in order of priority according to the…
A: Rank the set of substituent according to CIP rule---
Q: How many stereoisomers of 3-bromo-butan-2-ol exist? 01 02 O5 4.
A: The structure of 3-Bromo-butan-2-ol is:
Q: 10. Assigh E or Z configuration for each alkene shown below. EN H3CH2C. `CH2NH2 H3C HO CI
A: According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority. 2) If first atom…
Q: Explain with appropriate chair structures why cis-decalin (ie. cis-bicycle[4.4.0]decane) is only…
A:
Q: (a) Draw all stereoisomers formed by monobromination of the cis and trans isomers of…
A: A question based on alkanes that is to be accomplished.
Q: Arrange the following groups in the order of increasing priority (lowest priority to highest…
A: Polar covalent bond:The covalent bond is formed by the sharing of electrons between the atoms. If…
Q: 1. a) Draw the most stable chair conformation for each of the compounds shown below: Br X…
A: 1. We know, a conformation of a compound is the spatial arrangements of atoms which can be easily…
Q: CH, CH3 CH2 - CH2 - CH-CH CH3. CH3 CH3 CH3 CH3 CH Least stable elipsed configuration Most Stable H…
A: Yes you are true Eclipsed form is if dihedral angle is 0° between group If dieheadral angle is 60°…
Q: Increasing reactivity towards SN2 CI CI CI CI CH3 CH3 .C- ーH CH3 CH3 CH3 A В C Answer:
A: We have to predict the reactivity order of given alkyl halides in SN2 reaction.
Q: किष वैhह लनहलीवाकड ले शेण्डक बन्कमवmdेड कएकीवालड ले भ्रोण्क टनतकककादड the की निष्ट चैनए तलाव su…
A: -> In amine there are nitrogen which has loan pair .It can acts base and give acid base reaction…
Q: Rep Two stereoisomers of 1-bromo-4-methylcyclohexane are formed when trans 4-methylcyclohexanol…
A: Reaction of trans-4-methylcyclohexanol with HBr
Q: What is each compound’s systematic name?
A: Systematic Name: It is a standardized name given for a chemical compound in systematic manner. Any…
Q: 2 H ( A lily X IF Br
A:
imagine attached for question
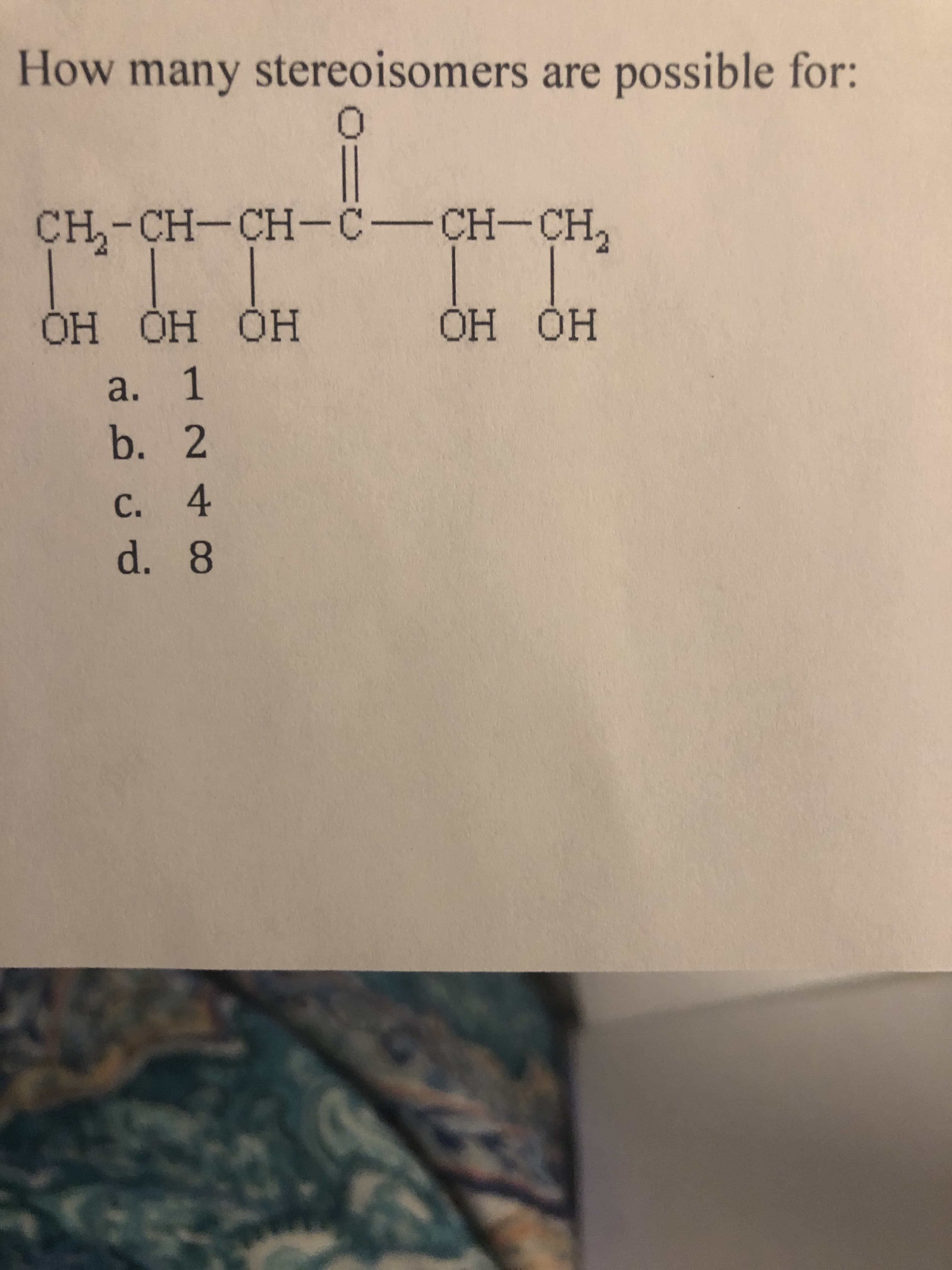
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- up an example (not appearing in this ChemActivity) of a pair of molecules that are a)constitutional isomers, b) conformers. c) configurational stereoisomers.If I chlorinate 2-bromo-1-chloropropane under light, how many products do I obtain? Are any chiral?Explain the observation with hydroxylation of cis-2-butene with OsO4 yields a different product than hydroxylation of trans-2-butene. First draw the structure and show the stereo chemistry each product and then molecular models.
- When HBr adds across the double bond of 1,2-dimethylcyclopentene, the product is a mixture of the cis and trans isomers. Show why this addition is not stereospecific.How many stereoisomers are possible for 1,4,7-octatriene? a) 2 b) 4 c) 8 d) 16How many unique stereoisomers exist for 1,2,3,4,5-pentachlorocyclohexane?
- Rank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2ClCalculate the difference in energy between the two conformation of trans_4_tert_butyl_1_methylcyclohexaneRings + Unsaturation --- Hydrogenation If compound A C51H81BrN5O3P3 is hydrogenated to give compound B C51H101BrN5O3P3. How many rings does compound A have? Assume that P has a valency of 5. Would the answer be 4 rings? Formula -> unsat + rings = 1+C +N/2 - H/2 - X/2
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.Consider 1-bromopropane, CH3CH2CH2Br. (a) Draw a Newman projection for the conformation in which CH3 and -Br are anti (dihedral angle 180°). (b) Draw Newman projections for the conformations in which - CH3 and -Br are gauche (dihedral angles 60° and 300°). (c) Which of these is the lowest energy conformation? (d) Which of these conformations, if any, are related by reflection?

