Q: Does the chemical potential of any constituent of an ideal solution depend on the ______ of the…
A: Chemical potential: In thermodynamics, the chemical potential of a species is the energy that can be…
Q: If 100. mL of 0.400 mol L-1 Na2SO4(aq) is added to 200 mL of 0.600 mol L- NaCl(aq), what is the…
A:
Q: How many distinct monochlorination products are possible when the alkane below undergoes free…
A: in the monochlorination product, one H is substituted by chlorine and one Cl is attached to the…
Q: Chemistry NO2 and NO are in an equilibrium according to the following equation [NO2] + [NO] [N2O3]…
A: Order of the reaction tells about the rate of the reaction and the concentration of all the species…
Q: 4. ( You are performing the titration of a buffer with a pKa of 7.2. You start at the pka, and after…
A: A Buffer solution resist the change of pH,When small amount of acids ar base is added.
Q: What is the hydroxide ion concentration in a solution that containing 0.50 mole of ammonium…
A: Given : No. of moles of Ammonium hydroxide = 0.50 mol No. of moles of Ammonium chloride = 0.05 mol…
Q: Turquoise, CuAl 6 ( P O 4 ) 4 ( OH) 8·5 H 2 O(s), is one of the most valuable non-transparent…
A: Calculate molar mass of both hydrated and anhydrous form of the mineral and then calculate the mass…
Q: The following data are for the decomposition of dinitrogen pentoxide in carbon tetrachloride…
A: The half life of a reaction depend on the initial concentration of compound. It is the time required…
Q: NH₂ х CI НО. дива ОН SH (1 eq) ОН pyridine ОН P205
A:
Q: 2. Write the balanced "net ionic" equations for the following reactions: HNO₂ (aq) KOH (aq) 7 HOX+ +…
A:
Q: A) methylbenzene B) aniline C) phenol D) anisole E) methyltoluene
A:
Q: In a saturated solution of BaCO3 at 298K, [Ba2+(aq)] = 7.07 x 10-5 mol L-1; [CO32- (aq)] =…
A: Ksp is the solubility product constant, is defined as equilibrium constant for a solid substance…
Q: Draw a complete structure for a molecule with the molecular formula C3H8O
A:
Q: The following is correct for a solution of the strong acid, HI HI reacts only with strong bases It…
A:
Q: Explain why sodium chloride, NaCl(s), is insoluble in benzene, C6H6(). In your explanation, refer to…
A:
Q: a-pinene en a-pinene reacts with 1. BH3; 2. H₂O₂, NaOH, H₂O ssible cis,trans isomers can be formed,…
A: According to the question, we need to draw the major product formed when pinene is treated with BH3…
Q: prepare 0.9% potassium Chloride (KCI) in 0.5 liter ?
A:
Q: How many atoms of each element are in a molecule of the amino acid lysine
A: Structure of lysine Chemical formula of lysine C6H14N2O2
Q: belove some reaction mechanism. in wwhich choice shows the necessary to complete the methenism I HBJ…
A: This is SN1 reaction so we will perform the mechanism of SN1 reaction.
Q: The thermochemical equation for the combustion of methanol is CH3OH (g) + 3/2 O2(g) → CO2(g) +…
A: Here we are required to find the heat produced on burning 5 gm of methanol
Q: A mystery element has two main isotopes of 62.9300amu and 64.928amu. Their percent abundances are…
A: The isotopes are the elements having same atomic number but different mass number.
Q: 500 mL of a 1.52 mol/L solution of vinegar (acetic acid) is used to react with a 2.53 g sample of…
A: Given the molarity of acetic acid(CH3COOH) in the vinegar solution, C = 1.52 mol/L Volume of acetic…
Q: Situation: 0.9% (w/v) sodium chloride (MW = 58.44 g/mol) solution (Normal Saline Solution) has an…
A:
Q: Which of the following compound has the highest negative heat of combustion? A. 2-methylheptane…
A:
Q: (1) The final boiling points of gasoline, diesel, atmospheric gas oil (AGO), and lubricating oils…
A: Boiling point order of gasoline,diesel, AGO , lubricating oil
Q: 247 96 Column A 1. 2. 3. Cm →He + 4X What is the atomic mass of the new atom? What is the atomic…
A:
Q: CfY+4X nn A What is the atomic mass of the new atom? What is the atomic number of the new atom? What…
A: In nuclear reaction, atomic number and mass number is balanced. Balanced chemical reaction,…
Q: 5) The combustion of sucrose (C12H22011, molar mass=342.3 g/mol) has a AErxn of -1.40 x 104 kJ/mol.…
A: In thermodynamics, the heat transfer is derived from the technique called as calorimetry. This heat…
Q: Given the reaction: 2 KMnO4 + 10 KI + H₂SO4 → 6 K2SO4 + 2 MnSO4 +5 12 + 8 H₂O How many moles of…
A: Given, 2 KMnO4 + 10 KI + H2SO4 → 6 K2SO4 + 2 MnSO4 + 5 I2 + 8 H2O moles of KMnO4 react = 5 moles…
Q: What is the correct answer to the following calculation? Select all answers that apply. Sig figs…
A: During division of two values the result must contain the number of significant figure equal to the…
Q: What is the mole fraction of CH3COOH in a solution containing 1.50 g CH3COOH (FM= 60.053 g/mol) and…
A: Given -> Weight of CH3COOH = 1.50 gm Molar mass of CH3COOH = 60.053 g/mole Weight of H2O = 1000.0…
Q: elementary reaction rate constant 1 H₂(g) + 2NO(g) t N₂O(g) + H₂O(g) k₁ 2 H₂(g) + N₂O(g) → N₂(g) +…
A:
Q: In a filtration of suspended cells under full vacuum using a 20 cm Büchner funnel, you collect data…
A: To decrease the time require for filtration- of solution of given volume we use a particular type of…
Q: The Ottawa Charter is increasingly used to guide the development of health promotion activities in…
A: The very first international conference on health promotion was held on Ottawa on 1st November,…
Q: What is the IR and HNMRAndCNMR and mass in this example explain
A: Double bond equivalence (DBE) of the molecular formula C2H4Br2 can be calculated by using the…
Q: What is the molar concentration of a solution containing 1.50 g CH3COOH (FM= 60.053 g/mol) and 1000…
A:
Q: pres 10) Which of the following equations demonstrates the formation of NaHCO3 from its respective…
A: In formation reaction one mole of a compound is formed from its constituent elements in their…
Q: A compound is made up of 19.1% Co, 34.4% CI, and 46.6% O. A 2.0 L sample of this compound had a mass…
A:
Q: Pls help on both questions.
A: From the percentage of individual elements we can determine the empirical formula of compound.
Q: 14. Propose a mechanism for following transformation. M₂O 0
A: In this question, we will write the details mechanism steps by using curved arrows for the given…
Q: d. Mg and Mg2+ e. Al and Al3+ f. Pb and Pb²-
A:
Q: Drawing H3O+ -OH Atoms, Bonds and Rings Charges Draw or tap a new bond to see suggestions. ← Undo Ⓒ…
A:
Q: IE₁ = 1012 kJ/mol; IE2 = 1900 kJ/mol; IE3 = 2910 kJ/mol; IE4 = 4960 kJ/mol; IE5 = 6270 kJ/mol; IE6 =…
A:
Q: One of the following is not one of the Minimization of errors. Internal standard method O الاختيارات…
A: Here we have to find which option is not correct I.e. one of them is not one of the minimization of…
Q: 1. CH3OH (shown below) is soluble in water. Draw molecular level pictures representing: (a) a…
A:
Q: 1. Which of the following reactions occurs most rapidly at standard conditions? A. 2Fe(s) + O2(g) →…
A: The correct options are as follows: 1. Option D. 2. Option A. 3. Option C.
Q: A compound is made up of 19.1% Co, 34.4% CI, and 46.6% O. A 2.0 L sample of this compound had a mass…
A: We have to determine the molecular formula of the gas
Q: Which of the following compound has the highest negative heat of combustion? A. 2-methylheptane…
A: Heat of combustion: The heat that is released when one mole of a substance is burned or undergo…
Q: 1) What is the major product for the following reaction? -BuOH, -BuOK A) Of-Bu B) c) D) E) 43
A: -> In presence of base it can abstracted most acidic hydrogen and form alkene the hydrogen which…
Step by step
Solved in 2 steps with 2 images

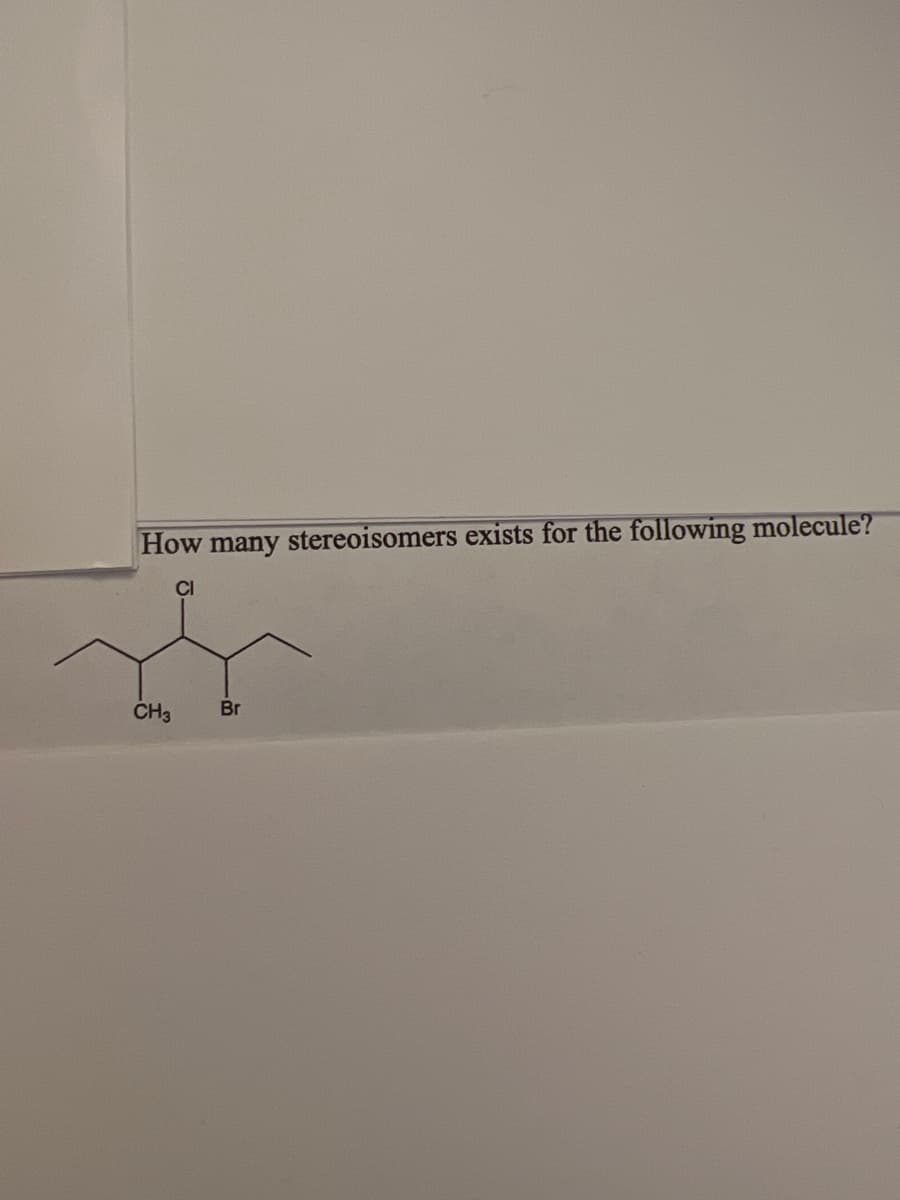
- Which of the following structures represent the same stereoisomer? A. only 1 and 2 B. only 1 and 3 C. only 2 and 3 D. 1, 2 and 3 Please explain.How many possible stereoisomers?Are the following constitutional isomers, conformers, or stereoisomers. If they are stereoisomers, are they enantiomers or diostereoemrs?

