Q: Represent by a Lewis structure the ammonium ion, NH4+. Then determine the molecular shape.
A: Given -> NH4+ ->Lewis structure is structural formula for molecule and polyatomic ions that…
Q: 6. The pH of a solution is calculated by using the formula pH = -log[H], [H*] where is the…
A:
Q: 3. Three ways to make 2-ethoxyoctane from 2-octanol outlined below. Predict the product, A B, and C…
A: During SN2 reaction involving breaking of bond at chiral center leads to the inversion in…
Q: b Milk contains 1.65 µg of riboflavin per milliliter. If you drink one quart (946 mL) of milk, how…
A: The concentration of riboflavin in milk is = 1.65 μg The volume of milk is = 946 mL The mass of…
Q: Match the following aqueous solutions with the appropriate letter from the column on the right..…
A: When non volatile solute are dissolved in solvent then freezing point of resultant solution…
Q: San Diego Bay is polluted due to leaching of hydrocarbons from old creosote pilings. If there are…
A: We have find out the steady state hydrocarbon level expected in the bay.
Q: Provide the missing curved arrow(s) to show the last step in the mechanism. H,C. CH3 H CH3 H CH3…
A:
Q: 5. The synthesis of aspirin (acetylsalicylic acid) is shown below. OH OH salicylic acid ("SA") ● H3C…
A:
Q: Draw several molecules of methanamide and show the intermolecular hydrogen bonding that account for…
A: In this question we have to draw the structure of methanamide which shows H bonding between…
Q: What is the ratio of ionized:unionized amine of glycine (pKa 9.60) in a solution at pH 8.0?
A: Amines are weak base. Amine like RNH2 and ionized amine like RNH3+. Mixture of amine and ionized…
Q: 1) Write structural formulas for each of the following. a) 3,4-Octadiene c) (Z,Z)-1,3-Cyclooctadiene…
A:
Q: 395. mg of an unknown protein are dissolved in enough solvent to make 5.00 mL of solution. The…
A: The osmotic pressure maybe calculated as follows:
Q: Complete the following statements. Word bank: a reaction coordinate lowest slower highest a lower…
A: Answer: All the terms mentioned in the above problem are used to characterize and study the reaction…
Q: what is the ionic equation and net ionic for caloric acid and barium hydroxide HClO3 + Ba(OH)2 =…
A:
Q: MISSED THIS? Read Section 15.6 (Pages 656 - 660); Watch KCV 15.6, IWE 15.9. Consider the following…
A: A chemical reaction can be added or subtracted like an algebraic equation. The given reaction…
Q: Historically, naphthalene was used as the main ingredient in mothballs and is easily recognized by…
A:
Q: What type(s) of bonding would be expected for each of the following materials: (Write on your…
A: Types of bonds are a)Ionic bonds b) Covalent bonds c) Metallic bonds d) van der Waals forces of…
Q: What temperature change results from the loss of 255 kJ in a 10.0 kg water sample? (c = 4.184 J/g…
A: The specific heat of water is 4.184 J/goC, which states that 4.184 joules of heat energy will be…
Q: Draw the Lewis structure of water, acetonitrile, ethanol, bromine and n-hexane show all lone pairs…
A: We have to draw the Lewis structures of water, acetonitrile, ethanol, bromine and n-hexane. We also…
Q: Allenes are a class of compounds that have two adjacent double bonds. The simplest allene…
A: We have been given an allene and we have been asked to predict hybridisations of two carbon atoms…
Q: ctrolyte, write out an ionic equation as NaCl(s)→ Na* (aq) + Cl(aq) f. CaCl₂ (soluble salt) strong…
A:
Q: See Figure 2-6. The MOST correct formula for the structure drawn is C9H14CINO2 CIO₂C8NH12…
A: We have to identify the correct formula for the above structure .
Q: IW a) b) CA +43 +30+ →→P (net) write the right equation Calaulate & Reaction A B 1.8.3.5 2 1.6.3.5…
A: A rate law shows how the rate of a chemical reaction depends on reactant concentration. For a…
Q: 15.59 Two methods convert an alkyl halide to a carboxylic acid having one more carbon atom. b. C. HO…
A: The first reaction proceeds via SN2 reaction mechanism where the CN- nucleophile attacks from…
Q: 3. Draw the orbital structure of hydrogen cyanide. H-CEN: 1. a-bond (sigma) bonding orbital…
A: A sigma bond is a covalent bond produced by collinear or coaxial overlapping of an atomic orbital in…
Q: A sample of neon gas occupies a volume of 750 ml at 25°C. What will the gas occupy at 50°C if the…
A: This question is related to gas laws. Gas laws relate the pressure, volume, and temperature of a…
Q: A doctor prescribes 500 mg of Pediazole to be taken every six hours. The pharmacy supplies a…
A:
Q: If an object has a density of 1.67g/mL and à volume of 3.00mL, what is the mass of the object?
A:
Q: [A] Slope = -0.42 t In [A] t 1 [A] t
A: Concentration = [A] in (M) Time = time in (s) Rate law = ?
Q: Write the two half-reactions of the following redox reaction. Calculates the difference between the…
A:
Q: 3. Consider the following pairs of compounds. (CLA) a. Br₂ and 12- Pick the one with the higher…
A: We are given a pair of compounds in the question. We need to identify the one with higher vapour…
Q: If an object has a mass of 3.80g and its volume by displacement changed a volume of water from…
A:
Q: A pure solid sample of Substance X is put into an evacuated flask. The flask is heated at a steady…
A: In a heating curve, the first plateau ( horozontal line ) corresponds to the melting point. At this…
Q: 8. Perform the following synthesis. cyclohexene trans-cyclohexene glycol
A: Epoxidation is the reaction where the C=C double bond is converted into oxiranes (epoxides) using…
Q: 2. Draw the structure of the major organic product for each of the following reactions. Be sure to…
A: Diels-Alder Reactions involves the diene and dienophile. When dienophile remain electron with…
Q: Which one of the following compounds has the highest UV absorption wavelength? a) Od O C о a b) c)…
A: Energy gap between π and π* is inversely proportional with wavelength that is with increasing the…
Q: CH₂=CH-CH=CH-CH=CH-CH₂-CH3 TOPAC Nance
A: Given compound -
Q: What is a key difference between alpha helices and beta sheets? An alpha helix is composed of two or…
A: Alpha-helix and beta-sheets are the types of secondary structure of protein.
Q: Find the solubility of lead(II) sulfate in 0.39 M Na2SO4 (aq). The solubility product of lead (II)…
A: Given , Solubility product of lead (II) sulfate 1.6×10-8
Q: Find the best plot for data graph and slope, and value of K for first order reaction: t (x) min 5 10…
A:
Q: 4. Mechanism. Provide a complete, arrow-pushing mechanism for each of the following reaction 1. 2…
A:
Q: A certain species of grasshopper secretes an allenic substance of molecular formula C13H2003 that…
A: In the given molecular formula of compound is C13H20O3 . Also the compound is have allene…
Q: The half-life of carbon-14, used in rad iocarbon dating, is 5730 years. What i s the decay constant…
A: Given --> Half life (t1/2)= 5730 years = 1.81 x 1011 s Decay constant (k) = ?
Q: When 5.12 g of a salt dissolves in 190. mL of water (density = 1.00 g/mL) in a coffee-cup…
A:
Q: See Figure 2-5. C-1 has a formal charge of _. C-2 has a formal charge of _. O has a formal…
A:
Q: The bond angle of H-O-C₁ is The bond angle of O-C₁-N is [Select] [ Select] The bond angle of C₁-N-C₂…
A: First let us consider the hybridisation of different atoms in isocyanomethanol. O atom= sp3…
Q: 3. Sulfur is applied to the skin for dandruff and itchy skin infection caused by mites (scabies). It…
A: The four quantum numbers for an electron are: 1. Principal quantum number (n) 2. Azimuthal quantum…
Q: Which of the following wave functions are eigenfunctions of the operator d²/dx²? Check all that…
A:
Q: a) What are the possible values of ml if n = 4 and l = 2? b) What kind of orbital is described by…
A: The set of numbers used to describe the position and energy of the electron in an atom are called…
Q: Give answer to all parts? For the system CH3COOH+ answer the following questions ?
A: 1)The total number of electrons would depend on the specific compound or species being referred to.…
Ee.41.
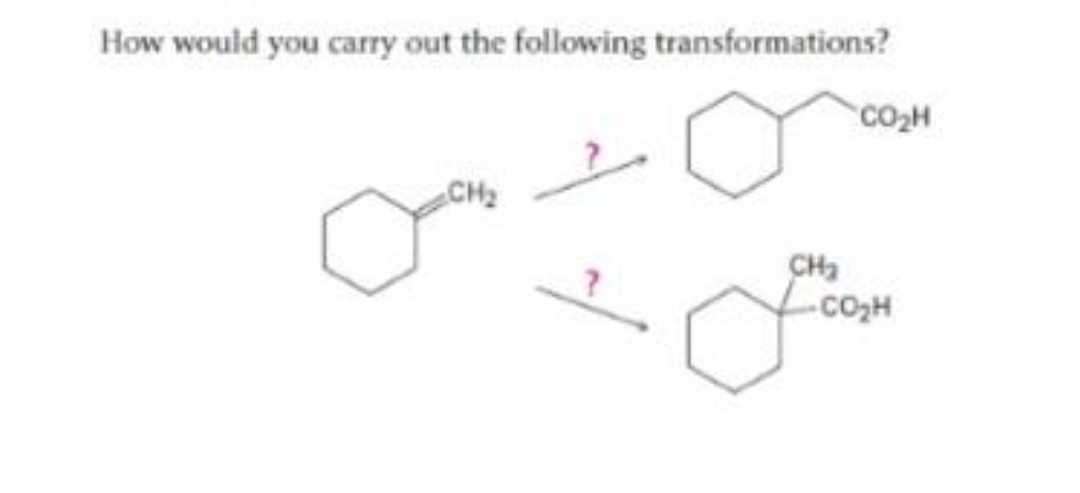
Step by step
Solved in 2 steps with 1 images

- What products would you expect from the reaction of 1-bromopropane with each of the following? (a) NaNH2 (b) KOC(CH3)3 (c) NaI (d) NaCN (e) NaCCH (f) Mg, then H2OWhat product do you expect from the reaction of the following etherswith HBr? a. CH3CH2OCH2CH3 b. cyclohexyl ethyl etherPropose mechanisms and show the expected products of the following reactions. p@nitrobromobenzene + methylamine (CH3 ¬NH2)
- Using bromocyclohexane as a starting material, how could you synthesize the following compounds?What is the major product obtained from the addition of HBr to the following compound?Propose a mechanism for each reaction, showing explicitly how the observed mixtures of products are formed.(a) 3@methylbut@2@en@1@ol + HBr S 1@bromo@3@methylbut@2@ene + 3@bromo@3@methylbut@1@ene

