Q: K, for benzoic acid, C,H¿COOH, is 6.30×105. K, for nitrous acid, HNO, is 4.50x10-4. K, for…
A: In this question, we will see the strongest conjugate base. You can see the details solution below.…
Q: Identify the conjugate bases corresponding to the following acids: [Co(NH3)5(OH2)]3+, HSO−4 ,…
A: In Bronsted-Lowry Theory, Bronsted acid is one which donates a proton to another compound present in…
Q: For the following reaction write the identify which compound is the Acid and Base on the reactant…
A:
Q: Write the formula of the conjugate base of C.H.SH.
A: When an Acid donates H+ , it forms conjugate base. When base accepts H+ , it forms conjugate acid.
Q: Identify the pair of species that is not a conjugate acid-base pair. H,SO,; HSO, H,CO3; CO; -…
A:
Q: Fill in the missing chemical formulae in the tables below. base conjugate acid H,0 Hpo, 2- НРО 4 OH
A:
Q: The following aqueous species constitute two conjugate acid-base pairs. Use them to write one…
A: The idea of conjugate acid-base theory is of Bronsted-Lowry acid-base theory. Stronger the acid…
Q: 2. Write the name and formula for the conjugate bases of the following: name of the conjugate…
A:
Q: base or acid
A:
Q: Draw the conjugate base for the acid (CH3)2C=OH+. Remember to include charges and non-bonding…
A: According to Bronsted-Lowry theory of acid and base deprotonation of molecule leads to the formation…
Q: For each conjugate acid-base pair, identify the first species as an acid or a base and the second…
A:
Q: Consider the following reaction, CH3COOH + HBr CH3COOH2+ + Br - Identify two acid-base conjugate…
A: Applying concept of conjugate acid and conjugate base.
Q: The following aqueous species constitute two conjugateacid-base pairs. Use them to write one…
A:
Q: Write the formula of the conjugate base of each acid. H2SO4 HI NH4+ HPO42- HCO3-
A: The conjugate base for acid is determined by removing an H+ from the formula of the acid in other…
Q: Write the formula of the conjugate base of each acid: HCI, HPO,, and CH,CH,CH2OH. Be sure to answer…
A: Conjugate base of acid :- There will be one less H in formula of conjugate base of acid formula.…
Q: A. Write the formula of the conjugate acid or base of the following substances: 1. Acids a. HC1O b.…
A:
Q: Write the formula for the conjugate base of each acid. (a) H2SO4 (b) H3BO3 (c) Hl (d) H3O+ (e) NH4+…
A: When an acidic molecule or ion donates H+ ions and accepts OH- ions then we get the conjugate base…
Q: According to the Brønsted-Lowry theory, an acid is any substance (molecule or ion) that can transfer…
A: Given: acids : HX, HY, HZ bases : X- Y- Z- in Question they given strongest base is Y- and…
Q: B. For the following bases, give their conjugate acid and rank the conjugate acids from strong to…
A: A question based on the concept of acid-base, which is to be accomplished.
Q: Write the formula for the conjugate base of each acid.a. HCl b. H2SO3 c. HCHO2 d. HF
A: A conjugate base is formed when a proton is removed from an acid.
Q: The formula for the conjugate acid of HPO42- is? The formula for the conjugate base of…
A: When the base is added with a hydrogen ion, H+ ion it gives the conjugate acid of that base.…
Q: Write the formula of the conjugate base of each acid: HF, HCO3 , and CH3OH. Be sure to answer all…
A: Bronsted-Lowry concept of acids and bases :- According to this theory , Acid is a substance which…
Q: Which of the following is not a conjugate acid-base pair? O H;SO4, SO, O HPO4, HPO O HNO3, NO; O…
A: For any pair to be conjugate acid-base pair, there should be difference of 1 proton i.e H+ between…
Q: Write the formula of the conjugate base of each acid: HCI, HPO,?-, and CH3CH,OH. Be sure to answer…
A: Conjugate base is formed by lose of one proton from an acid.
Q: Which two species act as the acids in the following reaction? HF CH3CO0 2F+CH3COOH OA CH3COO, F¯ O…
A:
Q: Compare the conjugate bases of these three acids. Acid 1: carbonic acid , H2C03 Acid 2: sulfurous…
A: In this question, first we will compare the conjugate bases of these given acids and then Identify…
Q: Which of the following pairs of species is not a conjugate acid-base pair? HOCI, OCI HNO2, NO2* O…
A: Conjugate acid-base pair is a pair of substances which only differ in terms of proton. When H+ is…
Q: What is the formula for the conjugate base of C5H¬N ?
A: Given :- acid = C5H7N To write :- formula of conjugate base
Q: Draw the conjugate base for the acid (CH,),C=OH*. Remember to include charges and non-bonding…
A: Acids are the species that have a tendency to donate a proton whereas base accepts a proton. In…
Q: Which one of the following pairs is a conjugate acid-base pair? H2CO3, CO32- H2CO3, HCO3- HCl, Cl…
A: Since conjugate acid-base pairs are those in which the difference between the 2 substance is of only…
Q: 1. The formula for the conjugate base of CH3COOH is . 2. The formula for the conjugate acid of…
A: Applying concept of conjugate acid/base.
Q: Which species is the conjugate base of HPO4 2-? Write the formula net charge
A: A base is any substance that can accept proton from any other substances or substance that ionize in…
Q: Identify acid, base, conjugate acid, and conjugate base in the following reactio a. H20 + H2CO3 =…
A: We know that acid is a substance that releases H+ ions in an aqueous solution. A base is a substance…
Q: Draw the conjugate base for the acid (CH,),C=OH*. Remember to include charges and non-bonding…
A: An acid is the one which loses proton (H+ ion) in aqueous solution to form its conjugate base. For…
Q: Write the conjugate base for each of the following acids: a) HS- b) H2SO3 c) NH3 d) HBrO4
A: Conjugate base of any acid is the substance formed after the acid donates 1 proton i.e H+ ion.
Q: The conjugate acid and conjugate base of bicarbonate ion, HCO 3 are, respectively: OA H30* and C032-…
A: According to the Bronsted-Lowry concept, the proton donating species are termed as acids while the…
Q: Identify the pair of species that is not a conjugate acid-base pair. CH,NH; CH,NH, H,CO3; CO; - H,…
A: conjugate acid base pair- acid have add H+ ion and base remove H+ ions by one H+ but in case of 2nd-…
Q: Write the formula of the conjugate base of CH$SH.
A: Conjugate base: The chemical species that results from the proton removal from acid is called the…
Q: 2. Write the formula for the conjugate base for each of the following acids: a. HCI b. HI С. HNO3 d.…
A: conjugate base:-the species that accepts a proton or hydrogen is the conjugate base.
Q: What is the conjugate base of HClO4, H2S, PH4 +, HCO3 - ?
A:
Q: Which of the following is a conjugate acid/base pair? A) H.PO4, PO.- B) H2PO., PO. C) HPO.2, P0.- D)…
A: Conjugate acid of any base is formed by adding 1 H+ ion to the base. And Conjugate base of any acid…
Q: Write the formula of the conjugate acid or base of the following substances: 1. Acids 2. Bases a.…
A: 1) Acids = The conjugate base of the given acids are as follows: a) HClO = HClO + OH- ---> ClO-…
Q: Draw the structures of the conjugate bases of the following acids:
A: To draw the structures of the conjugate bases of the following acids.
Q: Give the formula of the conjugate base:(a) HPO₄²⁻(b) NH₄⁺(c) HS⁻
A: Conjugate base of an acid is produced by loosing a proton from acid. HA ⇔ H+…
Q: 3. Designate the Lewis acid and Lewis base in each of the following reactions: ÇI CH, CH,-C-OH, ČH,…
A: Anything that occupies space and has mass is called matter. The matter may be classified into acids…
Q: Draw the structures of the conjugate bases of the following acids:
A: According to Bronsted-Lowry concept, the acidic and basic character of a compound can be interpreted…
Q: 4. Write the formula of the conjugate base of each acid. H2SO4 HI NH4+ HPO42- HCO3-
A:
Q: conjugate acid conjugate base Ka K, formula formula b. H,CO, - 7 4.5 X 10 CH,NH, - 4 4.4 X 10…
A: Conjugate acid base pair will always have a difference of 1 H+ ion. Hence we can say Conjugate acid…
Q: (a) Give the conjugate base of the following Brønsted– Lowry acids: (i) HCOOH, (ii) HPO4 2-.
A: The conjugate base of given Bronsted-Lowry acids has to be given.
| Write the formula of the conjugate base of each acid: HI, HPO42−, and CH3CH2CH2OH. Be sure to answer all parts |
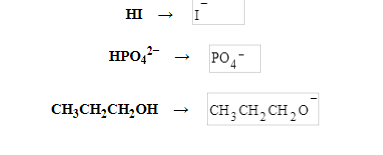
Step by step
Solved in 2 steps

- (a) How does the structure of diborane (B2H6) differ fromthat of ethane (C2H6)? (b) Explain why diborane adoptsthe geometry that it does. (c) What is the significance ofthe statement that the hydrogen atoms in diborane aredescribed as “hydridic”?what is the reasonance structure of this?Explain- Conversion of RCOOH to RCOOR' ?
- In the methoxide anion (CH3O-), is it possible for a double bond to exist between C and O, given that the negative chargeresides on O? Explain why or why not.Write the molecular diagram (HOMO LUMO) of indigo C16H10N2O2 and explainHow would I draw all the important resonance structures for cyanate (NCO1-)?

