Q: Which equilibrium system will show an increase in product formation when the volume is decreased? A.…
A: Le Chatelier's principle: According to the Le Chatelier's principle when a change in concentration…
Q: Desribe the reaction product. (q) CI ОН ОН (r) (s) 1 H ОН ОН
A:
Q: 11. A weak acid (Ka = 2.5 x 104) is neutralized with a weak base (Kb = 5.0 x 108). What is the pH of…
A: 11. option: a. Acidic 12. option: c. Ka = Kb 13. option: c. NH4ClO4 (aq) 14. option: a.…
Q: The reaction 5 CI (aq) + CIO3(aq) + 6 H*(aq) → 3 Cl₂(g) + 3 H₂O(l) is expected to obey the following…
A: The slowest step of the reaction decides the rate of the reaction. So it is also called rate…
Q: Support it with the correct solution 1. What mass of aluminum oxide must be electrolyzed with…
A: In Hall's process for the industrial production of Al(s), the below half-reactions takes place in…
Q: At 300 K the rate constant for a reaction is 1.0 x 1010 times greater in the presence of an enzyme…
A: Given: The rate constant for a reaction is 1.0×1010 times greater in presence of an enzyme compared…
Q: Arrange the following sets of systems in order of increasing entropy. Assume one mole of each…
A: Entropy is a measure of randomness of a system. It is denoted by capital letter 'S' and its value…
Q: (7) A . B C D What is the major product of the following reaction? Br CH3O+Na CH3OH J (A) OCH 31 (B)…
A:
Q: The reaction shown below has Kc = 9.0. 2 A(g) ⇒ B(g) + C(g) If you start with 1.0 M A, what is the…
A:
Q: (3.9: Similar to For More Practice 3.15) To find the mass of potassium in 5.6 g potassium hydroxide…
A:
Q: 4. Determine the formula mass for each of Br2: A. 133.34 amu B. 238.0 amu C. 18.02 amu D. 159.8…
A: Molecule is made up of atoms. An atom consist of nucleus that contain protons and neutrons.
Q: If a system releases 80 kJ of heat while having 50 kJ of work done on it, the AEuniverse is positive…
A: According to 1st Law of Thermodynamics, dE= q+ও The heat gain by the system (q)= -80kj,…
Q: A 29mL sample of chromium-51 contains 1.06 mCimCi. If a patient is to receive a 50 μCiμCi dose to…
A:
Q: Given the reaction mechanism shown below Step 1 Ce4+ + Mn2+ → 3+ Ce³+ + Mn³+ 3+ Step 2 Ce4+ + Mn³+ →…
A:
Q: Convert the following transmittance data to absorbance. a) 27.2% b) 0.579% c) 30.6%
A: The relation between transmittance and absorbance -> A = 2- log(T%) A = absorbance T% = percent…
Q: XIDIZED Cr+ + Sn4+ 72+ our answer
A: The given reaction, Cr + Sn4+ -----> Cr3+ + Sn2+ Half reaction splitting Cr ----> Cr3+ + 3e…
Q: 5. Draw line structures to correspond with the following names a. 3-isopropylpentane b.…
A:
Q: 2 → HOCI Activity 2: Complete the reaction by drawing or writing the correct structure to match the…
A:
Q: 3.Complete the following Reaction. Include the IUPAC name of the product. OH + H₂C-CH-CH₂-NH₂ H50,…
A:
Q: The completion of the synthesis of the first compound is provided below. B Br Using the list below,…
A: The alkene react with hydrogen halide in presence of peroxide to give the addition product in which…
Q: The completion of the synthesis of the second compound is provided below. B Br Using the list below,…
A:
Q: . Household sugar, sucrose, has the molecular formula C12H22011. What is the %, by mass, of carbon…
A:
Q: Use the following data to calculate the reaction rate law and the rate law constant for the system.…
A: Given reaction: NO (g) + H2 (g) → HNO2 (g) Given data: We have to find the rate law and the rate…
Q: A compound containing chromium and silicon contains 73.52 mass percent chromium. Determine its…
A:
Q: Assign E or Z configuration (a) H3C CH₂OH C=C CH3CH₂ Cl (c) CH3 C=C CO₂H CH₂OH to the following…
A: Rules for assigning E or Z configuration
Q: At a certain temperature, the equilibrium constant K for the following reaction is 0.0041: NO3(g) +…
A: Here we are required to find the equilibrium constant.
Q: 19. Calculate the number of moles oh hydrogen atoms in 17.4 g of (NH4)2SO4. A. 2.13 mol H atoms B.…
A:
Q: Kinetic data were obtained on the aqueous reaction of hydrogen peroxide with iodide ion and H+.…
A:
Q: In a 0.50 M solution of Li3PO4 the theoretical value of i is 3 none of the answers are correct 02 04
A: Colligative properties: The colligative properties are those properties of solutions that depend on…
Q: HO |||| A CH 3 # H NH/₂ 0
A: the following steps to be followed : Give each atom connected to the chiral center a priority…
Q: How many peaks (13C) would be evident in the decoupled spectrum of a. methylcyclohexane b.…
A: Given compounds: a. methylcyclohexane b. cyclohexene c. 1-methylcyclohexene We have to find the…
Q: Identify the following reaction if it is redox or not, HNO3 + SO2 ----> H2SO4 + NO2.
A: Redox reaction is type of reaction in which oxidation and reduction takes place.
Q: 3.Complete the following Reaction. Include the IUPAC name of the product. [ OH + H₂C-CH₂-CH₂-NH₂ a.…
A: This question is related to condensation reaction in which two species combined with the removal of…
Q: 1. Calculate the number of moles of cyclohexanol (d = 0.96g/ml) if 20 mL was used in the reaction?
A: Dear student, The informtion for question two is not complete. Please provide the information on…
Q: Write the complete IUPAC name of the molecule. Consider the stereochemistry of the molecule in…
A: given writing the IUPAC name
Q: Hydrogen is manufactured on an industrial scale by this sequence of reactions: CH4 (g) + H₂O(g) = CO…
A:
Q: 3. Which structural formula below represents an unsaturated hydrocarbon? Explain. H H H H H-C-H…
A:
Q: For large, hydrophobic molecules the dispersion force is usually dominant. Which of the two…
A: Melting point depends on the packing of the molecules in a solid, and among trans and cis alkenes,…
Q: (2.8)An element occurs as three isotopes with atomic masses 19.99 amu (abundance = 90.51%), 20.99…
A: We have to calculate the atomic mass the given element
Q: An electrolytic cell for depositing copper from copper sulfate solutions is placed in series with…
A: We use the formula below to find the weight of a metal(W) deposited in an electrode during…
Q: Kinetic data were obtained on the aqueous reaction of hydrogen peroxide with iodide ion and H+.…
A:
Q: Which of the following is true for chemical equilibrium? I. Forward and reverse reaction rates are…
A:
Q: Which structure of SCO¹ is the most stable? 1. :0=C=S: II. :O-CES: III. :o=c-s: BIL Ô) II. D) II.…
A: we have to select the most stable structure
Q: An electrolytic cell was used to cover a cupper cathode with a thin layer of Silver, AgNO3 is the…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: If a 1 g/mL solution of a mixture of compounds F and G in a 1 decimeter polarimetry cell rotates…
A: Don't worry, I am providing the detailed solution of this question, so that you may understand it…
Q: Research and explain the equilibrium relationship the following system: Human blood A…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Consider the following equilibrium: PCl3(g) + Cl2(g) ⇄ PCl5(g) When 0.80 moles of PCl3 and 0.80…
A:
Q: 5. Calculate the number of molecules in 5.00 mol of NH3. A. 3.01 x 1024 molecules of NH3 B. 1.03 x…
A: Given, Moles of NH3 = 5.00 mol. The number of molecules of NH3 in 5.00 mole of NH3 is:
Q: 1. When 18.2g of sugar, a nonelectrolyte, is dissolved in exactly 600.0g of water, the solution has…
A:
Q: The specific heat capacity of lead is 0.13 J/g∙oC. How much heat (in J) is required to raise the…
A:
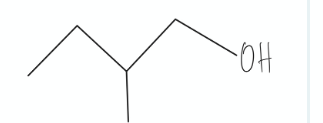
Determined if the molecule is achiral or chiral
Step by step
Solved in 2 steps with 1 images


