http://openvellum.ecollege.com/course.html?courseld-154322748HepID-2b3e48e6520860bfd5591538a4a5a27b#10001 Search... ne y w any kilojoules are releas... USAonline: EMS-440-801 Fall2... n.com- Online Sh... TripAdvisor Ses 21 of 40 ents Review | Constants | Periodic Table A sample containing 8.00 g 02 gas has a volume of 31.0 L. Pressure and temperature remain constant Part A Eings What is the new volume if 0.800 mole O2 gas is added? ? EXEW PVA V = 1.18 L Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Part B PPearson 2019 Pearson Education Inc. All rights reserved. Terms of Use Copyright Privacy Policy Permissions Contact Us hp a 1:41 PM 12/4/2019 hp Stocio Sournd ins prt sc f11 f6 f12 f8 f9 144 * + $ & 3 6 7 2 P U T Y (6 L K D F 00 I Ln CR S ge.com/course.html?courseld= 15432274&HepID=2b3e48e6520860bfd5591538a4a5a27b#10001 Search. How many kilojoules are releas... S USAonline: EMS-440-801 Fall 2... TripAdvisor 21 of 40 Review | Constants | Periodic Table A sample containing 8.00 g O2 gas has a volume of 31.0 L. Pressure and temperature remain Oxygen is released until the volume is 10.5L. How many moles of O, are removed? constant Iνα ΑΣφ mol Submit Request Answer Part C What is the volume after 6.00 g He is added to the O, gas already in the container? Πνα ΑΣΦ P Pearson Copyright 2019 Pearson Education Inc. All rights reserved. Terms of Use Privacy Policy Contact Us Permissions hp a 1:41 PM 12/4/2019 hp Odts Studio Sound f6 f11 fg 1A4 1o ins prt sc f12 delete %23 & 7. 4. 8. 6. backspace { H. L. enter pause B %24
http://openvellum.ecollege.com/course.html?courseld-154322748HepID-2b3e48e6520860bfd5591538a4a5a27b#10001 Search... ne y w any kilojoules are releas... USAonline: EMS-440-801 Fall2... n.com- Online Sh... TripAdvisor Ses 21 of 40 ents Review | Constants | Periodic Table A sample containing 8.00 g 02 gas has a volume of 31.0 L. Pressure and temperature remain constant Part A Eings What is the new volume if 0.800 mole O2 gas is added? ? EXEW PVA V = 1.18 L Submit Previous Answers Request Answer X Incorrect; Try Again; 5 attempts remaining Part B PPearson 2019 Pearson Education Inc. All rights reserved. Terms of Use Copyright Privacy Policy Permissions Contact Us hp a 1:41 PM 12/4/2019 hp Stocio Sournd ins prt sc f11 f6 f12 f8 f9 144 * + $ & 3 6 7 2 P U T Y (6 L K D F 00 I Ln CR S ge.com/course.html?courseld= 15432274&HepID=2b3e48e6520860bfd5591538a4a5a27b#10001 Search. How many kilojoules are releas... S USAonline: EMS-440-801 Fall 2... TripAdvisor 21 of 40 Review | Constants | Periodic Table A sample containing 8.00 g O2 gas has a volume of 31.0 L. Pressure and temperature remain Oxygen is released until the volume is 10.5L. How many moles of O, are removed? constant Iνα ΑΣφ mol Submit Request Answer Part C What is the volume after 6.00 g He is added to the O, gas already in the container? Πνα ΑΣΦ P Pearson Copyright 2019 Pearson Education Inc. All rights reserved. Terms of Use Privacy Policy Contact Us Permissions hp a 1:41 PM 12/4/2019 hp Odts Studio Sound f6 f11 fg 1A4 1o ins prt sc f12 delete %23 & 7. 4. 8. 6. backspace { H. L. enter pause B %24
Chapter30: Kinetic Methods Of Analysis
Section: Chapter Questions
Problem 30.12QAP
Related questions
Question
Transcribed Image Text:http://openvellum.ecollege.com/course.html?courseld-154322748HepID-2b3e48e6520860bfd5591538a4a5a27b#10001
Search...
ne
y
w any kilojoules are releas...
USAonline: EMS-440-801 Fall2...
n.com- Online Sh...
TripAdvisor
Ses
<MC 13 on Temperature, Changes in Energy, and Gases
ome
Item 21
>
21 of 40
ents
Review | Constants | Periodic Table
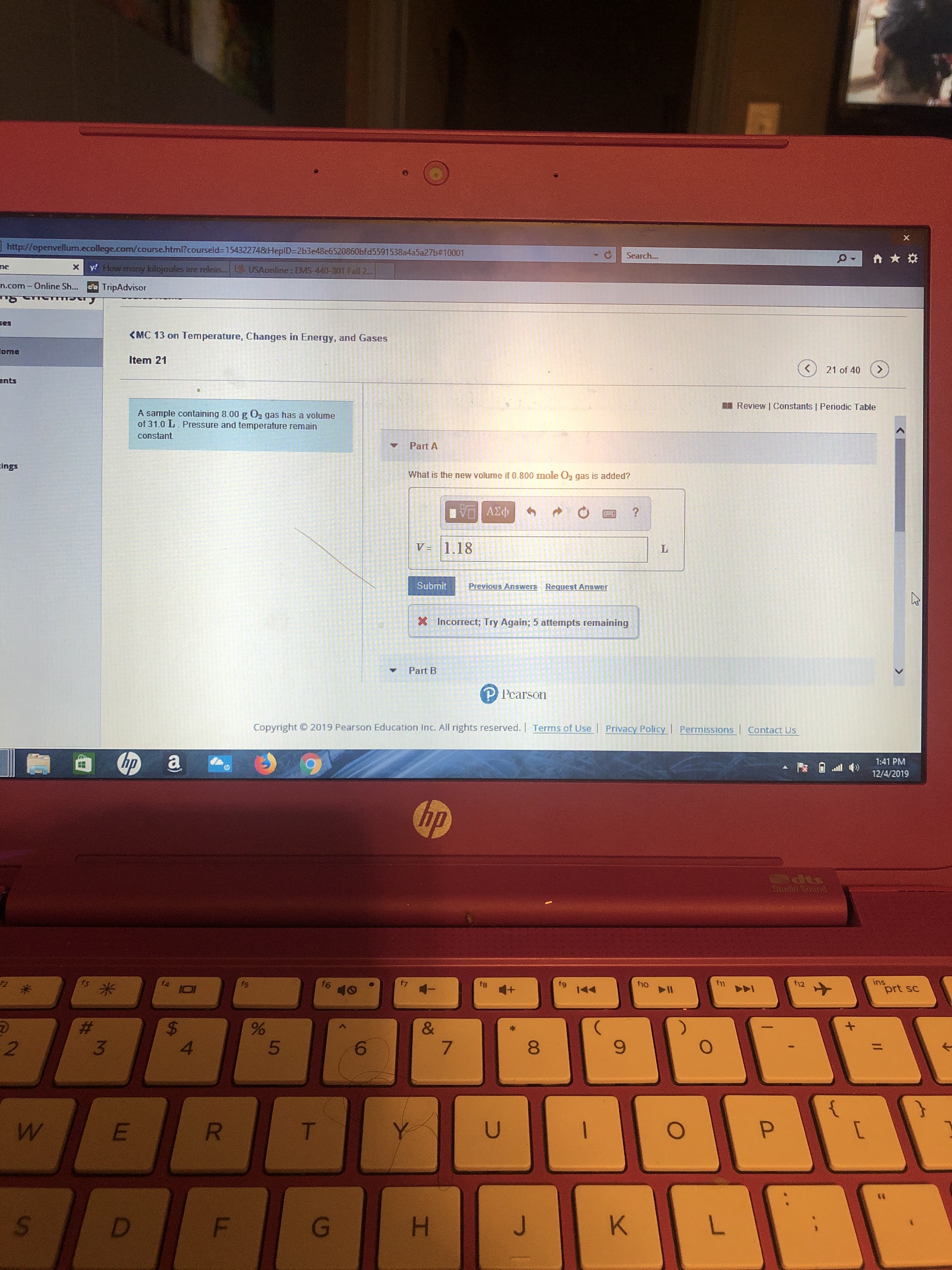
A sample containing 8.00 g 02 gas has a volume
of 31.0 L. Pressure and temperature remain
constant
Part A
Eings
What is the new volume if 0.800 mole O2 gas is added?
?
EXEW
PVA
V = 1.18
L
Submit
Previous Answers Request Answer
X Incorrect; Try Again; 5 attempts remaining
Part B
PPearson
2019 Pearson Education Inc. All rights reserved. Terms of Use
Copyright
Privacy Policy
Permissions
Contact Us
hp
a
1:41 PM
12/4/2019
hp
Stocio Sournd
ins
prt sc
f11
f6
f12
f8
f9
144
*
+
$
&
3
6
7
2
P
U
T
Y
(6
L
K
D
F
00
I
Ln
CR
S
Transcribed Image Text:ge.com/course.html?courseld= 15432274&HepID=2b3e48e6520860bfd5591538a4a5a27b#10001
Search.
How many kilojoules are releas... S USAonline: EMS-440-801 Fall 2...
TripAdvisor
<MC 13 on Temperature, Changes in Energy, and Gases
Item 21
<>
21 of 40
Review | Constants | Periodic Table
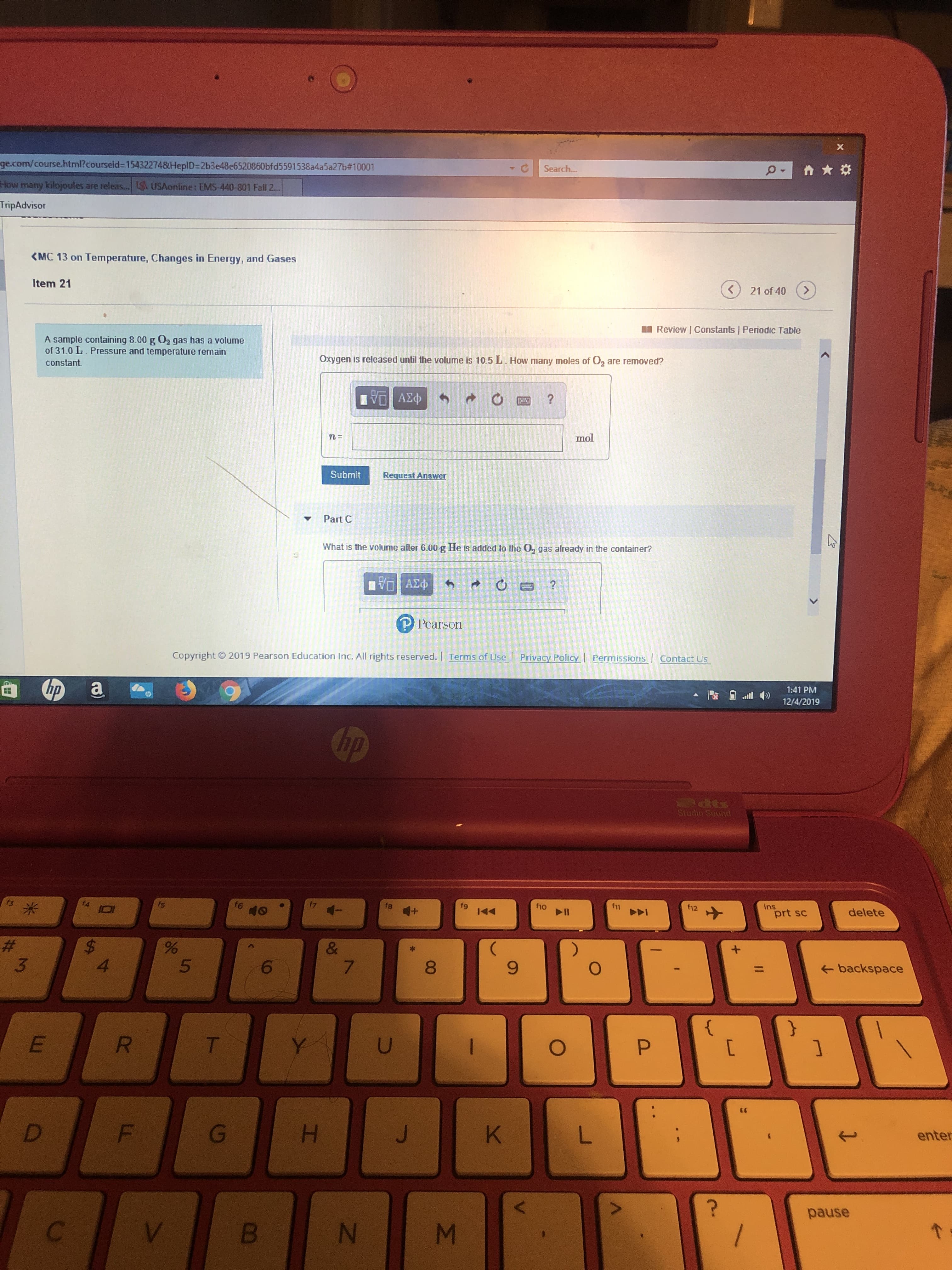
A sample containing 8.00 g O2 gas has a volume
of 31.0 L. Pressure and temperature remain
Oxygen is released until the volume is 10.5L. How many moles of O, are removed?
constant
Iνα ΑΣφ
mol
Submit
Request Answer
Part C
What is the volume after 6.00 g He is added to the O, gas already in the container?
Πνα ΑΣΦ
P Pearson
Copyright
2019 Pearson Education Inc. All rights reserved.
Terms of Use
Privacy Policy
Contact Us
Permissions
hp
a
1:41 PM
12/4/2019
hp
Odts
Studio Sound
f6
f11
fg
1A4
1o
ins
prt sc
f12
delete
%23
&
7.
4.
8.
6.
backspace
{
H.
L.
enter
pause
B
%24
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 3 images

Recommended textbooks for you

