HW1 - U1M1 Homework. Due in 2 days O 1/15 answered Energy Exchanged Homework Unanswered Click on the region of these energy transfer (AE) versus temperature (T) graphs that corresponds to a phase change from gas to liquid. ΔΕ AE O Targets placed: 0/1 Delete selected Remove All Undo 3 Fullscreen You can place up to 1 targets
HW1 - U1M1 Homework. Due in 2 days O 1/15 answered Energy Exchanged Homework Unanswered Click on the region of these energy transfer (AE) versus temperature (T) graphs that corresponds to a phase change from gas to liquid. ΔΕ AE O Targets placed: 0/1 Delete selected Remove All Undo 3 Fullscreen You can place up to 1 targets
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter2: The First Law Of Thermodynamics
Section: Chapter Questions
Problem 2.14E: A 5-mmdiameter hailstone has a terminal velocity of10.0m/s. Assuming its mass is6.031025 kg and all...
Related questions
Question
Transcribed Image Text:HW1 - U1M1
1/15 answered
Homework . Due in 2 days
Energy Exchanged
Homework• Unanswered
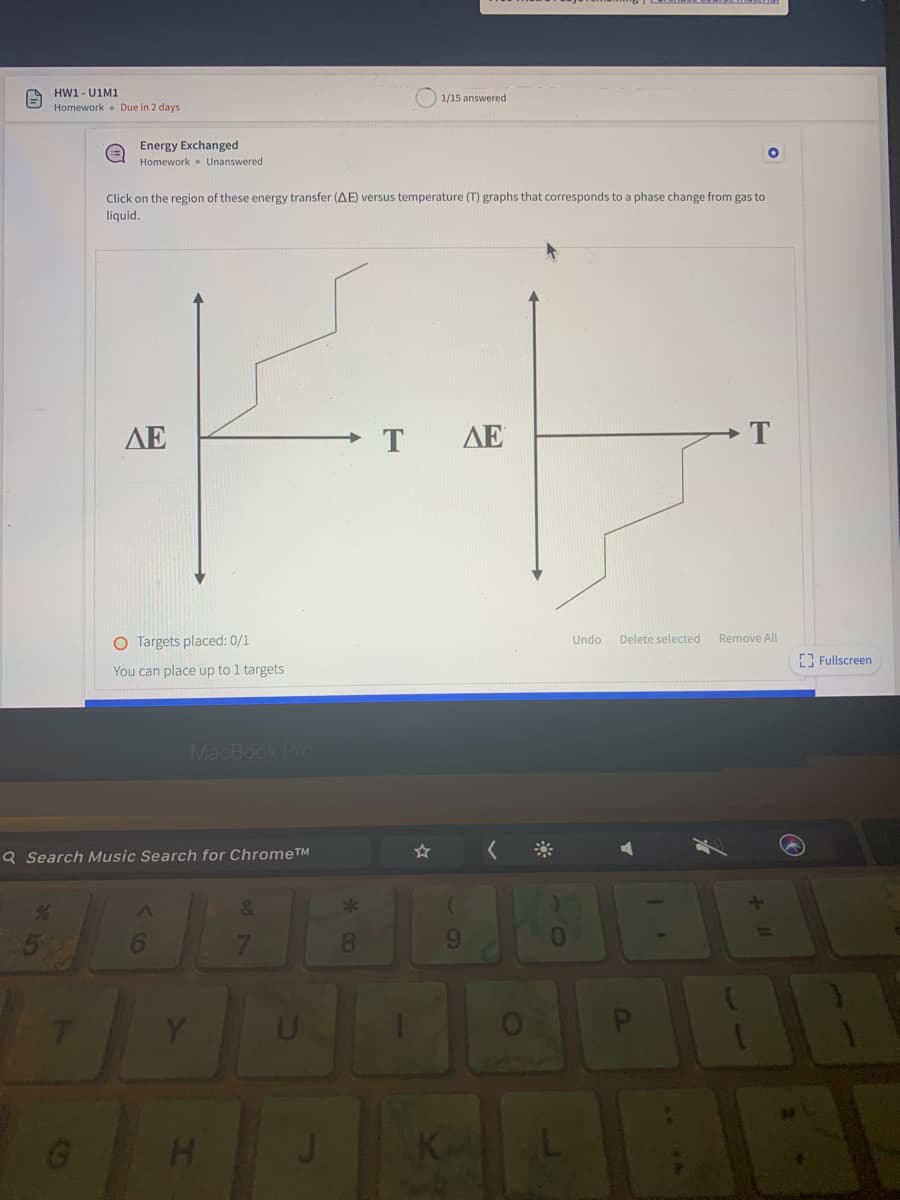
Click on the region of these energy transfer (AE) versus temperature (T) graphs that corresponds to a phase change from gas to
liquid.
AE
T
AE
O Targets placed: 0/1
Delete selected
Remove All
Undo
Fullscreen
You can place up to 1 targets
MacBook Pro
Q Search Music Search for ChromeTM
8.
9.
116
K
Expert Solution
Step 1
The diagram shows phase change of a substance energy vs Temp phase change diagram.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning